Abstract
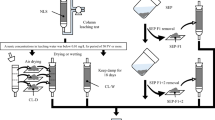
Massive quantities of marine sedimentary rock are excavated from urban coastal areas. The excavated rock often releases arsenic with concurrent oxidation of framboidal pyrite, but the arsenic release is naturally suppressed with subsequent atmospheric exposure. The present study evaluated the re-release of arsenic from excavated rock in which arsenic release has been naturally suppressed by the atmospheric exposure in the presence of sulfate ions under various redox conditions using the biological reduction method. The atmospheric exposure and subsequent batch leaching test revealed that the amount of arsenic release that was naturally suppressed corresponded to 1.2% of the total arsenic content. The sequential extraction analysis also showed that the arsenic in the exposed rock was altered to insoluble phases. We observed a re-release of 6.0–18.2% of the total arsenic content under reductive conditions (< + 70 mV of Eh), exceeding the amount of arsenic that was naturally suppressed, even in the presence of sulfate ions. The correlation in the amount of arsenic and iron re-released demonstrates that arsenic re-release under reductive conditions is mainly regulated by the iron dissolution up to 10 mg kg−1 even in the presence of sulfate ion. Further reduction and dissolution of iron did not cause further increase in the arsenic re-release. Therefore, excavated marine sedimentary rock should be reused under redox conditions in which iron is not reduced. Otherwise, treatments such as chemical immobilization should be performed.







Similar content being viewed by others
References
Alam, R., & McPhedran, K. (2019). Application of biological sulfate reduction for remediation of arsenic – A review. Chemosphere, 222, 932–944. https://doi.org/10.1016/j.chemosphere.2019.01.194
Buschmann, J., Kappeler, A., Lindauer, U., Kistler, D., Berg, M., & Sigg, L. (2006). Arsenite and arsenate binding to dissolved humic acids: Influence of pH, type of humic acid, and aluminum. Environmental Science and Technology., 40, 6015–6020. https://doi.org/10.1021/es061057+
Carbonell-Barrachina, A. A., Jugsujinda, A., Burlo, F., Delaune, R. D., & Patrick, W. H., Jr. (1999). Arsenic chemistry in municipal sewage sludge as affected by redox potential and pH. Water Research., 34, 216–224. https://doi.org/10.1016/S0043-1354(99)00127-X
Chandra, A. P., & Gerson, A. R. (2010). The mechanisms of pyrite oxidation and leaching: A fundamental perspective. Surface Science Reports., 65, 293–315. https://doi.org/10.1016/j.surfrep.2010.08.003
Choppala, G., Bush, R., Moon, E., Ward, N., Wang, Z., Bolan, N., & Sullivan, L. (2017). Oxidative transformation of iron monosulfides and pyrite in estuarine sediments: Implications for trace metals mobilization. Journal of Environmental Management., 186, 158–166. https://doi.org/10.1016/j.jenvman.2016.06.062
Dixit, S., & Hering, J. G. (2003). Comparison of arsenic(V) and arsenic(III) sorption onto iron oxide minerals: implications for arsenic mobility. Environmental ScienceTechnology., 37, 4182–4189. https://doi.org/10.1021/es030309t
Drahota, P., Rohovec, J., Filippi, M., Mihaljevič, M., Rychlovský, P., Červený, V., & Pertold, Z. (2009). Mineralogical and geochemical controls of arsenic speciation and mobilityunder different redox conditions in soil, sediment and water at the Mokrsko – West gold deposit, Czech Republic. Science of the Total Environment., 407, 3372–3384. https://doi.org/10.1016/j.scitotenv.2009.01.009
Duan, Y., Schaefer, M. V., Wang, Y., Gan, Y., Yu, K., Deng, Y., & Fendorf, S. (2019). Experimental constraints on redox-induced arsenic release and retention from aquifer sediments in the central Yangtze River Basin. Science of the Total Environment., 649, 629–639. https://doi.org/10.1016/j.scitotenv.2018.08.205
Endo, K., Takezaki, S., Takai, A., Katsumi, T., (2015). Monitoring results of experimental banking using recovered soils from disaster waste; -First year results-. In Proceedings of the 11th Japanese geotechnical society symposium on environmental geotechnics. 105–112
Fendorf, S., Michael, H. A., & Geen, A. (2010). Spatial and temporal variations of groundwater arsenic in South and Southeast Asia. Science, 328, 1123–1127. https://doi.org/10.1126/science.1172974
Feng, Q., Zhang, Z., Chen, Y., Liu, L., Zhang, Z., & Chen, C. (2013). Adsorption and desorption characteristics of arsenic on soils: Kinetics, equilibrium, and effect of Fe(OH)3 colloid, H2SiO3 colloid and phosphate. Procedia Environmental Sciences, 18, 26–36. https://doi.org/10.1016/j.proenv.2013.04.005
Frohne, T., Rinklebe, J., Diaz-Bone, R. A., & Laing, G. D. (2011). Controlled variation of redox conditions in afloodplain soil: Impact on metal mobilization and biomethylation ofarsenic and antimony. Geoderma, 160, 414–424. https://doi.org/10.1016/j.geoderma.2010.10.012
Fukushi, K., Sakai, M., Munemoto, T., Yokoyama, Y., & Takahashi, Y. (2016). Arsenate sorption on monohydrocalcite by coprecipitation during transformation to aragonite. Journal of Hazardous Materials, 304, 110–117. https://doi.org/10.1016/j.jhazmat.2015.10.019
Goldberg, S., & Johnston, C. T. (2001). Mechanisms of adsorption on amorphous oxide evaluated using macroscopic measurements, vibrational spectroscopy, andsurface complexation modeling. Journal of Colloid and Interface Science., 234, 204–216. https://doi.org/10.1006/jcis.2000.7295
Gorny, J., Billon, G., Noiriel, C., Dumoulin, D., Lesven, L., & Madé, M. (2018). Redox behaviour of arsenic in the surface sediments of the Marque River (Northern France). Journal of Geochemical Exploration, 188, 111–122. https://doi.org/10.1016/j.gexplo.2018.01.021
Han, D. S., Song, J. K., Batchelor, B., & Abdel-Wahab, A. (2013). Removal of arsenite(As(III)) and arsenate(As(V)) by synthetic pyrite (FeS2): Synthesis, effect of contact time, and sorption/desorption envelopes. Journal of Colloid and Interface Science, 392, 311–318. https://doi.org/10.1016/j.jcis.2012.09.084
Huminicki, D. M. C., & Rimstidt, J. D. (2009). Iron oxyhydroxide coating of pyrite for acid mine drainage control. Applied Geochemistry, 24, 1626–1634. https://doi.org/10.1016/j.apgeochem.2009.04.032
Jerz, J. K., & Rimstidt, J. D. (2004). Pyrite oxidation in moist air. Geochimica Et Cosmochimica Acta, 68, 701–714. https://doi.org/10.1016/S0016-7037(03)00499-X
Kamata, A., & Katoh, M. (2019). Arsenic release from marine sedimentary rock after excavation from urbanized coastal areas: Oxidation of framboidal pyrite and subsequent natural suppression of arsenic release. Science of the Total Environment, 670, 752–759. https://doi.org/10.1016/j.scitotenv.2019.03.217
Katoh, M., Matsuoka, H., & Sato, T. (2015a). Stability of lead immobilized by apatite in lead-containing rhizosphere soil of buckwheat (Fagopyrum esculentum) and hairy vetch (Vicia villosa). International Journal of Phytoremediation, 17, 604–611. https://doi.org/10.1080/15226514.2014.950413
Katoh, M., Wang, Y., Kitahara, W., & Sato, T. (2015b). Impact of phosphorus and water-soluble organic carbon in cattle and swine manure composts on lead immobilization in soil. Environmental Technology, 36, 1943–1953. https://doi.org/10.1080/09593330.2015.1016461
Katoh, M., Moriguchi, S., Takagi, N., Akashi, Y., & Sato, T. (2018). Simultaneous control of cadmium release and acidic pH neutralization in excavated sedimentary rock with concurrent oxidation of pyrite using steel slag. Journal of Soils and Sediments, 18, 1194–1204. https://doi.org/10.1007/s11368-017-1892-0
Katsumi, T. (2015). Soil excavation and reclamation in civil engineering: Environmentalaspects. Soil Science Plant Nutrition, 61, 22–29. https://doi.org/10.1080/00380768.2015.1020506
Langner, H. W., & Inskeep, W. P. (2000). Microbial reduction of arsenate in the presence of ferrihydrite. Environmental Science & Technology, 34, 3131–3136. https://doi.org/10.1021/es991414z
LeMonte, J. J., Stuckey, J. W., Sanchez, J. Z., Tappero, R., Rinklebe, J., & Sparks, D. L. (2017). Sea level rise induced arsenic release from historically contaminated coastal soils. Environmental Science & Technology, 51, 5913–5922. https://doi.org/10.1021/acs.est.6b06152
Lengke, M. F., & Tempel, R. N. (2003). Natural realgar and AsS oxidation kinetics. Geochimica Et Cosmochimica Acta, 67, 859–871. https://doi.org/10.1016/S0016-7037(02)01227-9
Lin, H. T., Wang, M. C., & Li, G. C. (2004). Complexation of arsenate with humic substance in water extract of compost. Chemosphere, 56, 1105–1112. https://doi.org/10.1016/j.chemosphere.2004.05.018
Lin, Z., & Puls, R. W. (2000). Adsorptions, desorption and oxidation of arsenic affected by clay minerals and aging process. Environmental Geology, 39, 753–759. https://doi.org/10.1007/s002540050490
Mamindy-Pajany, Y., Hurel, C., Marmier, N., & Roméo, M. (2009). Arsenic adsorption ontohematite and goethite. Comptes Rendus Chimie, 12, 876–881. https://doi.org/10.1016/j.desal.2011.07.046
Masscheleyn, P. H., Delaune, R. D., & Patrick, W. H. (1991). Effect of redox potential and pH on arsenic speciation and solubility in a contaminated soil. Environmental Science & Technology, 25, 1414–1419. https://doi.org/10.1021/es00020a008
Matamoros-Veloza, A., Peacock, C. L., & Benning, L. G. (2014). Selenium speciation inframboidal and euhedral pyrites in shales. Environmental Science Technology, 48, 8972–8979. https://doi.org/10.1021/es405686q
Mitsunobu, S., Harada, T., & Takahasi, Y. (2006). Comparison of antimony behavior with that of arsenic under various soil redox conditions. Environmental Science and Technology, 40, 7270–7276. https://doi.org/10.1021/es060694x
Neumann, T., Scholz, F., Kramar, U., Ostermaier, M., Rausch, N., & Berner, Z. (2013). Arsenic in framboidal pyrite from recent sediments of a shallow water lagoon of the Baltic Sea. Sedimentology, 60, 1389–1904. https://doi.org/10.1111/sed.12031
Nesbitt, H. W., Muir, I. J., & Pratt, A. R. (1995). Oxidation of arsenopyrite by air and air-saturated, distilled water, and implications for mechanism of oxidation. Geochimica Et Cosmochimica Acta, 59, 1773–1786. https://doi.org/10.1016/0016-7037(95)00081-A
Nickson, R. T., Mcarthur, J. M., Ravenscroft, P., Burgess, W. G., & Ahmed, K. M. (2000). Mechanism of arsenic release to groundwater, Bangladesh and West Bengal. Applied Geochemistry, 15, 403–413. https://doi.org/10.1016/S0883-2927(99)00086-4
O’Shay, T., Hossner, L. R., & Dixon, J. B. (1990). A modified hydrogen peroxide oxidation method for determination of potential acidity in pyritic overburden. Journal of Environmental Quality, 19, 4. https://doi.org/10.2134/jeq1990.00472425001900040024x
Ogawa, S., Kaoth, M., Numako, C., Kitahara, K., Miyazaki, S., & Sato, T. (2016). Immobilization of antimony(III) in oxic soil using combined application of hydroxyapatite and ferrihydrite. Water, Air, & Soil Pollution, 227, 124. https://doi.org/10.1007/s11270-016-2826-y
Pi, K., Wang, Y., Xie, X., Huang, S., Yu, Q., & Yu, M. (2015). Geochemical effects of dissolved organic matter biodegradation on arsenic transport in groundwater systems. Journal of Geochemical Exploration, 149, 8–21. https://doi.org/10.1016/j.gexplo.2014.11.005
Pierce, M. L., & Moore, C. B. (1982). Adsorption of arsenite and arsenate on amorphous ironhydroxide. Water Research, 16, 1247–1253. https://doi.org/10.1016/0043-1354(82)90143-9
Qiu, G., Gao, T., Hong, J., Tan, W., Liu, F., & Zheng, L. (2017). Mechanisms of arsenic-containing pyrite oxidation by aqueous arsenate under anoxic conditions. Geochimica et Cosmochimica Acta, 217, 306–319. https://doi.org/10.1016/j.gca.2017.08.030
Schippers, A., & Jorgensen, B. B. (2001). Oxidation of pyrite and iron sulfide by manganese dioxide in marine sediment. Geochimica Et Cosmochimica Acta, 65, 915–922. https://doi.org/10.1016/S0016-7037(00)00589-5
Schippers, A., & Jorgensen, B. B. (2002). Biogeochemistry of pyrite and iron sulfide oxidation in marine sediments. Geochimica Et Cosmochimica Acta, 66, 85–92. https://doi.org/10.1016/S0016-7037(01)00745-1
Shuman, L. M. (1985). Fractionation method for soil microelements. Soil Science, 140, 11–22. https://doi.org/10.1097/00010694-198507000-00003
Smedley, P. L., & Kinniburgh, D. G. (2002). A review of the source, behavior and distribution of arsenic in natural waters. Applied Geochemistry, 17, 517–568. https://doi.org/10.1016/S0883-2927(02)00018-5
Smith, A. H., Lingas, E. O., & Rahman, M. (2000). Contamination of drinking–water by arsenic in Bangladesh: A public health emergency. Bulletin of the World Health Organization, 78, 1093–1103. https://doi.org/10.1590/S0042-96862000000900005
Sun, F., Dempsey, B. A., & Osseo-Asare, K. A. (2012). As(V) and As(III) reactions on pristine pyrite and on surface-oxidized pyrite. Journal of Colloid and Interface Science, 388, 170–175. https://doi.org/10.1016/j.jcis.2012.08.019
Tabelin, C. B., & Igarashi, T. (2009). Mechanisms of arsenic and lead release from hydrothermally altered rock. Journal of Hazardous Materials, 169, 980–990. https://doi.org/10.1016/j.jhazmat.2009.04.049
Tabelin, C. B., Igarashi, T., Villacorte-Tabelin, M., Park, I., Opiso, E. M., Ito, M., & Hiroyoshi, N. (2018). Arsenic, selenium, bolon, lead, cadmium, copper, and zinc in naturally contaminated rocks: A review of their sources, modes of enrichment, mechanisms of release, and mitigation strategies. Science of the Total Environment, 645, 1522–1553. https://doi.org/10.1016/j.scitotenv.2018.07.103
Tangviroon, P., Hayashi, R., & Igarashi, T. (2017). Effects of additional layer(s) on the mobility of arsenic from hydrothermally altered rock in laboratory column experiments. Water, Air, & Soil Pollution, 228, 191. https://doi.org/10.1007/s11270-017-3295-7
Tresintsi, S., Simeonidis, K., Katsikini, M., Paloura, E. C., Bantsis, G., & Mitrakas, M. (2014). A novel approach for arsenic adsorbents regeneration using MgO. Journal of Hazardous Materials, 265, 217–225. https://doi.org/10.1016/j.jhazmat.2013.12.003
Ueshima, M., Takemura, T., Saito, T., Ito, Y., Hamamoto, S., Saito, H., & Komatsu, T. (2017). Relationship between trace elements and depositional environments in shallowsediments: A case study from Southern Kanto Plain. Central Japan. Environmental Earth Science, 76, 633. https://doi.org/10.1007/s12665-017-6968-z
Vongphuthone, B., Kobayashi, M., & Igarashi, T. (2017). Factors affecting arsenic content of unconsolidated sediments and its mobilization in the Ishikari Plain, Hokkaido. Japan. Environmental Earth Science, 76, 645. https://doi.org/10.1007/s12665-017-6993-y
Walker, F. P., Schreiber, M. E., & Rimstidt, J. D. (2006). Kinetics of arsenopyrite oxidative dissolution by oxygen. Geochimica Et Cosmochimica Acta, 70, 1668–1676. https://doi.org/10.1016/j.gca.2005.12.010
Wenzel, W. W., Kirchbaumer, N., Prohaska, T., Stingeder, G., Lombi, E., & Adriano, D. C. (2001). Arsenic fractionation in soils using an improved sequential extraction procedure. Analytica Chimica Acta, 436, 309–323. https://doi.org/10.1016/S0003-2670(01)00924-2
Xie, X., Wang, Y., Su, C., Liu, H., Duan, M., & Xie, Z. (2008). Arsenic mobilization in shallow aquifers of Datong Basin: Hydrochemical and mineralogical evidences. Journal of Geochemical Exploration, 98, 107–115. https://doi.org/10.1016/j.gexplo.2008.01.002
Xu, W., Wang, H., Liu, R., Zhao, X., & Qu, J. (2011). Arsenic release from arsenic-bearing Fe-Mn binary oxide: Effects of Eh condition. Chemosphere, 83, 1020–1027. https://doi.org/10.1016/j.chemosphere.2011.01.066
Yamaguchi, N., Nakamura, T., Dong, D., Takahashi, Y., Amachi, S., & Makino, T. (2011). Arsenic release from flooded paddy soils is influenced by speciation, Eh, pH, and iron dissolution. Chemosphere, 83, 925–932. https://doi.org/10.1016/j.chemosphere.2011.02.044
Yun, S. W., Park, C. G., Jeon, J. H., Darnault, C. J. G., Baveye, P. C., & Yu, C. (2016). Dissolution behavior of As and Cd in submerged paddy soil after treatment with stabilizing agents. Geoderma, 270, 10–20. https://doi.org/10.1016/j.geoderma.2015.11.036
Zhang, M., Dai, M., Xia, L., & Song, S. (2017). Comparison of arsenic adsorption on goethite and amorphous ferric oxyhydroxide in water. Water, Air, & Soil Pollution. https://doi.org/10.1007/s11270-017-3602-3
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kamata, A., Ueshima, M., Sakanakura, H. et al. The effects of redox conditions on arsenic re-release from excavated marine sedimentary rock with naturally suppressed arsenic release. Environ Geochem Health 44, 4157–4171 (2022). https://doi.org/10.1007/s10653-021-01178-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-021-01178-5