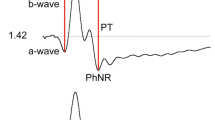
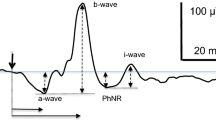
Abstract
Purpose
We studied the conditions under which c-waves of the electroretinogram (ERG), that represent retinal pigment epithelium (RPE) function, were detectable using an alternating current (AC) amplifier and whether the c-wave recorded using an AC amplifier was useful for evaluating RPE function.
Methods
We recorded ERG responses in rats to 5 s stimuli under the conditions in which the low-cut frequency and the stimulus luminance were varied. In addition, changes in ERGs were studied after intravenous injection of sodium iodate (SI) to induce RPE degeneration.
Results
The c-wave was detected clearly when the frequency of the low-cut filter was set at 0.01 Hz and light stimulus luminances were ≥ − 1.0 log cd/m2. The c-wave was attenuated earlier than other waves (e.g., a-wave and b-wave) after SI administration.
Conclusions
The c-wave was easily detectable using an AC amplifier with the low-cut filter set at 0.01 Hz. Using the AC amplifier may allow easier c-wave recording, compared with the conventional use of a direct current (DC) amplifier, and could be useful for evaluating RPE function.





Similar content being viewed by others
References
McCulloch DL, Marmor MF, Brigell MG, Hamilton R, Holder GE, Tzekov R, Bach M (2015) ISCEV Standard for full-field clinical electroretinography (2015 update). Doc Ophthalmol 130:1–12. https://doi.org/10.1007/s10633-014-9473-7
Nilsson SE, Wrigstad A (1997) Electrophysiology in some animal and human hereditary diseases involving the retinal pigment epithelium. Eye (Lond) 11:698–706. https://doi.org/10.1038/eye.1997.180
Granit R (1933) The components of the retinal action potential in mammals and their relation to the discharge in the optic nerve. J Physiol 77:207–239. https://doi.org/10.1113/jphysiol.1933.sp002964
Steinberg RH, Linsenmeier RA, Griff ER (1985) Chapter 2 Retinal pigment epithelial cell contributions to the electroretinogram and electrooculogram. Prog Retin Res 4:33–66. https://doi.org/10.1016/0278-4327(85)90004-5
Oakley B 2nd, Green DG (1976) Correlation of light-induced changes in retinal extracellular potassium concentration with c-wave of the electroretinogram. J Neurophysiol 39:1117–1133. https://doi.org/10.1152/jn.1976.39.5.1117
Kinoshita J, Peachey NS (2018) Noninvasive electroretinographic procedures for the study of the mouse retina. Curr Protoc Mouse Biol 8:1–16. https://doi.org/10.1002/cpmo.39
Wu J, Marmorstein AD, Kofuji P, Peachey NS (2004) Contribution of Kir4.1 to the mouse electroretinogram. Mol Vis 10:650–654
Dodt E, Echte K (1961) Dark and light adaptation in pigmented and white rat as measured by electroretinogram threshold. J Neurophysiol 24:427–445. https://doi.org/10.1152/jn.1961.24.4.427
Graves AL, Green DG, Fisher LJ (1985) Light exposure can reduce selectively or abolish the C-wave of the albino rat electroretinogram. Invest Ophthalmol Vis Sci 26:388–393
Shirao Y, Wajima R, Kaneko T, Nishimura A (1997) Neural retinal contribution to the slow negative potential of the canine electroretinogram. Doc Ophthalmol 94:293–306. https://doi.org/10.1007/BF02580855
Steinberg RH, Frishman LJ, Sieving PA (1991) Chapter 6 negative components of the electroretinogram from proximal retina and photoreceptor. Prog Retin Res 10:121–160. https://doi.org/10.1016/0278-4327(91)90011-P
Sieving PA, Frishman LJ, Steinberg RH (1986) Scotopic threshold response of proximal retina in cat. J Neurophysiol 56:1049–1061. https://doi.org/10.1152/jn.1986.56.4.1049
Katz BJ, Xu Z, Zheng J, Oakley B 2nd (1992) PII component of the toad electroretinogram. J Neurophysiol 68:333–341. https://doi.org/10.1152/jn.1992.68.1.333
Kikawada N (1968) Variations in the corneo-retinal standing potential of the vertebrate eye during light and dark adaptations. Jpn J Physiol 18:687–702. https://doi.org/10.2170/jjphysiol.18.687
Rohrer B, Long Q, Coughlin B, Wilson RB, Huang Y, Qiao F, Tang PH, Kunchithapautham K, Gilkeson GS, Tomlinson S (2009) A targeted inhibitor of the alternative complement pathway reduces angiogenesis in a mouse model of age-related macular degeneration. Invest Ophthalmol Vis Sci 50(7):3056–3064. https://doi.org/10.1167/iovs.08-2222
Redfern WS, Storey S, Tse K, Hussain Q, Maung KP, Valentin JP, Ahmed G, Bigley A, Heathcote D, McKay JS (2011) Evaluation of a convenient method of assessing rodent visual function in safety pharmacology studies: effects of sodium iodate on visual acuity and retinal morphology in albino and pigmented rats and mice. J Pharmacol Toxicol Methods 63:102–114. https://doi.org/10.1016/j.vascn.2010.06.008
Yang Y, Ng TK, Ye C, Yip YW, Law K, Chan SO, Pang CP (2014) Assessing sodium iodate induced outer retinal changes in rats using confocal scanning laser ophthalmoscopy and optical coherence tomography. Invest Ophthalmol Vis Sci 55:1696–1705. https://doi.org/10.1167/iovs.13-12477
Liu Y, Li Y, Wang C, Zhang Y, Su G (2019) Morphologic and histopathologic change of sodium iodate-induced retinal degeneration in adult rats. Int J Clin Exp Pathol 12(2):443–454
Lurie M, Marmor MF (1980) Similarities between the c-wave and slow PIII in the rabbit eye. Invest Ophthalmol Vis Sci 19:1113–1117
Hanitzsch R, Lichtenberger T (1997) Two neuronal retinal components of the electroretinogram c-wave. Doc Ophthalmol 94:275–285. https://doi.org/10.1007/BF02582985
Raitta C, Carlson S, Vannas-Sulonen K (1990) Gyrate atrophy of the choroid and retina: ERG of the neural retina and the pigment epithelium. Br J Ophthalmol 74:363–367. https://doi.org/10.1136/bjo.74.6.363
Wu J, Peachey NS, Marmorstein AD (2004) Light-evoked responses of the mouse retinal pigment epithelium. J Neurophysiol 91:1134–1142. https://doi.org/10.1152/jn.00958.2003
Samuels IS, Sturgill GM, Grossman GH, Rayborn ME, Hollyfield JG, Peachey NS (2010) Light-evoked responses of the retinal pigment epithelium: changes accompanying photoreceptor loss in the mouse. J Neurophysiol 104:391–402. https://doi.org/10.1152/jn.00088.2010
Kiuchi K, Yoshizawa K, Shikata N, Moriguchi K, Tsubura A (2002) Morphologic characteristics of retinal degeneration induced by sodium iodate in mice. Curr Eye Res 25:373–379. https://doi.org/10.1076/ceyr.25.6.373.14227
Textorius O, Welinder E (1981) Early effects of sodium iodate on the directly recorded standing potential of the eye and on the c-wave of the DC registered electroretinogram in albino rabbits. Acta Ophthalmol (Copenh) 59:359–368. https://doi.org/10.1111/j.1755-3768.1981.tb03001.x
Linsenmeier RA, Steinberg RH (1983) A light-evoked interaction of apical and basal membranes of retinal pigment epithelium: c-wave and light peak. J Neurophysiol 50:136–147. https://doi.org/10.1152/jn.1983.50.1.136
Acknowledgements
The authors thank Mr. Hidetaka Kudo of Mayo Corporation for technical assistance.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Naohisa Umeya designed the study, collected and analyzed data, and wrote the initial draft of the manuscript. Izuru Miyawaki and Hiroshi Inada contributed to the interpretation of data and assisted in the preparation of the manuscript. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethics approval (include appropriate approvals or waivers)
All procedures in this study were approved by the Institutional Animal Care and Use Committee of Drug Research Division, Sumitomo Pharma Co., Ltd.
Informed consent
This article does not contain any studies with humans performed by any of the authors.
Statement of human rights
This article does not contain any studies with humans performed by any of the authors.
Statement on the welfare of animals
All procedures in this study were approved by the Institutional Animal Care and Use Committee of Drug Research Division, Sumitomo Pharma Co., Ltd.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Umeya, N., Miyawaki, I. & Inada, H. Use of an alternating current amplifier when recording the ERG c-wave to evaluate the function of retinal pigment epithelial cells in rats. Doc Ophthalmol 145, 147–155 (2022). https://doi.org/10.1007/s10633-022-09890-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10633-022-09890-y