Abstract
Background
Patients with immune-mediated conditions such as IBD and RA are at risk for vaccine-preventable infections. Despite guideline recommendations, prior studies have shown suboptimal vaccination rates.
Aim
We conducted a systematic review and meta-analysis to compare the different interventions intended to increase vaccination rates.
Methods
A systematic search was conducted of MEDLINE/PubMed, Embase, CINAHL, and Cochrane Library up to 2020 for studies with interventions intended to increase vaccination rates. We performed a random-effects meta-analysis to generate pooled odds ratios (ORs) to assess all interventions against no interventions. Our primary outcome was pneumococcal vaccination (PCV) rate.
Results
Our review found 8580 articles, for which 15 IBD and 8 RA articles met the inclusion criteria; 21 articles were included in the analysis. PCV was the predominant vaccination (91%). In our analysis of patients with IBD, almost all interventions (patient-oriented, physician-oriented, or barrier-oriented) increased PCV uptake [OR, 4.74; 95% CI, 2.44–6.56, I2 = 90%] compared to no intervention. The greatest effect was seen in barrier-oriented studies [OR, 12.68; 95% CI, 2.21–72.62, I2 = 92%]. For RA data, all interventions had increased PCV uptake compared to no interventions (OR 2.74; 95% CI, 1.80–4.17, I2 = 95%).
Conclusion
Our data suggest that many different interventions can increase PCV rates. It appears that barrier-oriented interventions may have the greatest positive effect on increasing PCV uptake. However, clinicians should be encouraged to implement measures best suited to their practice. Future high-quality randomized controlled trials are needed to determine the best approach to optimize vaccination rates.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patients with immune-mediated conditions such as inflammatory bowel disease (IBD) and rheumatoid arthritis (RA) are at risk for vaccine-preventable infections [1], with rates of serious life-threatening infection ranging from 8.4 to 22.4 per 1000 person years [2]. In the USA, it is estimated that between 9 and 45 million illnesses can be prevented by vaccinations [3]. Additionally, vaccinations are extremely important cost-saving interventions, as the cost of vaccine-preventable illness in the USA is approximately $27 billion [3].
Unfortunately, vaccination rates in patients with IBD and RA are suboptimal for a multitude of reasons. Only about 50% of gastroenterologists routinely collected vaccination status information, despite most perceiving vaccinations as “extremely important” [4, 5]. Forty-five percent of gastroenterologists felt that primary care physicians should administer vaccinations; however, some primary care physicians felt that gastroenterologists should be responsible [6, 7]. When surveyed, 28% of patients received an annual flu shot and only 9% were up-to-date on pneumococcal vaccination (PCV) [8]. When asked for reasons for low vaccination rates, 49% stated they lacked awareness and 18% had concerns about side-effects [8, 9]. Other limitations were linked to concern for safety of vaccination in the immunosuppressed and the possibility of exacerbation of underlying IBD [10].
Over the years, there have been many studies that have assessed interventions attempting to increase vaccination rates in both IBD and RA patients. These interventions ranged from reminders and education for patients to education and electronic medical record interventions for physicians. Lastly, in the recent years, there has been implementation interventions to address barriers to patient adherence in the form of patient navigators. The sole responsibility of patient navigators is to provide patients with personalized guidance to navigate the health system. Therefore, we conducted a systematic review and meta-analysis of the different modalities currently studied to increase vaccination rates in these patients.
Methods
Literature Search
This study was reported according to the Preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA) guidelines [11]. We searched MEDLINE (PubMed-New), Embase (Elsevier), Cumulative Index to Nursing and Allied Health Literature (EBSCO), and Cochrane Library (Wiley). The search was created and conducted by a librarian (IKG) in consultation with the study authors. Strategies consisted of free text keywords and database-specific controlled vocabulary terms for IBD, RA, vaccines, and the concept of uptake. The search was built to be highly sensitive due to the heterogeneous nature of terms used to describe vaccines and vaccination uptake rates. The decision to use patients with either IBD or RA was because they are both immune-mediated conditions that are treated with the same or similar immunosuppressive medications, which in theory will increase the risk of vaccine-preventable diseases. All databases were searched from database inception PubMed 1966; Embase 1947; CINAHL 1937; and Cochrane Library 1993 through July 2020 (for IBD) or August 2020 (for RA) for studies in patients with these diseases. Separate searches were done for each patient population but the terms for vaccines and uptake remained the same in each. Full search strategies, including applied limits, for each database are available in supplementary data (see Supplementary data). Vaccinations that were included in the systematic review were based on and consistent with the recommendations from American College of Gastroenterology and European Crohn’s and Colitis Organization in collaboration of Canadian IBD society vaccination and preventive care guidelines [12, 13]. Manual searches of bibliographies and the journals Crohn’s and Colitis 360 were performed to identify any further studies that met inclusion criteria. Potentially relevant articles were obtained in full text and reviewed independently. Study review and selection were done by two independent reviewers [DF and JP], using Covidence systematic review software [14].
Selection Criteria
Our inclusion criteria consisted of patients ≥ 18 years old, patients with either IBD or RA, full-length peer-reviewed publications, and articles in English. We excluded conference proceedings, abstract-only articles, and review articles.
We classified interventions into four groups: (1) patient-oriented (i.e., reminders and education), (2) physician-oriented (i.e., education and electronic medical record), (3) combined patient- + physician-oriented and (4) barrier-oriented (i.e., navigator systems, which are defined as persons whose primary responsibility is to provide personalized guidance to patients as they navigate through the health system). Disagreements between the two independents reviewers were discussed with a third party reviewer [MP].
Data Extraction
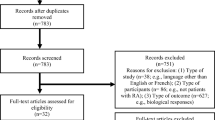
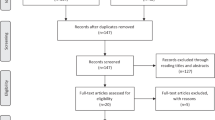
The PRIMSA flow diagrams for IBD (Fig. 1) and RA (Fig. 2) show the number of studies included at each stage of the screening. A data extraction sheet was designed in Microsoft Excel for the following items: year published, country, dates of study, type of study, setting (i.e., academic vs private practice), office type (general gastroenterology vs IBD clinic vs rheumatology clinic), number of gastroenterologists/rheumatologists, number of patients, IBD phenotype, classification of intervention, specific vaccination studies with pre- and post-intervention vaccination rates, and confirmation of vaccination (see supplemental documents).
Statistical Analysis
We performed the analysis in two different steps. First, we described pre- and post-intervention outcomes of the included studies. Second, based on the descriptive review of the studies, PCV rates were the most studied; therefore, PCV rate became our primary outcome for the meta-analysis.
The meta-analysis was performed by 2 of us [MN, AW]; we calculated pooled odds ratios (ORs) and 95% confidence intervals (CIs) using Mantel–Haenszel random-effects model to assess all interventions against no interventions. Statistical heterogeneity was assessed using the I-square statistic (< 25% was indicative of low heterogeneity and > 75% was indicative of high heterogeneity not due to chance). We performed direct comparisons using RevMan (Cochrane Collaboration, Copenhagen, Denmark, version 5.4). Due to the small number of included studies, we were not able to assess for publication bias [15].
Results
Literature Search/Demographics
IBD
As shown in Fig. 1, our IBD literature search identified 5625 citations. After removal of duplicate studies, screening, and full-text review, 15 studies met the inclusion criteria for the systematic review (Table 1) [16,17,18,19,20,21,22,23,24,25,26,27,28,29,30]. The studies were published between 2013 and 2019 in the USA (n = 10), Europe (n = 4), or Australia/United Kingdom (n = 1). A total of 6959 patients and at least 284 gastroenterologists were studied (33.0% of the studies did not include number of gastroenterologists). Practice settings varied, with the majority being academic centers (n = 10); the remaining centers were community-based (n = 2), Veteran Affairs system (n = 1), private practice (n = 1), and a combination of academic and private practice (n = 1). Most of the studies (13/15) were pre-post-test interventions and the remaining were randomized controlled trials. Many of the studies were physician-oriented (8/15), and the remaining were patient-oriented (3/15), combined patient and physician-oriented (2/15), and barrier-oriented (2/15). Barrier-oriented was implemented with either an infectious disease physician or an IBD nurse acting as a navigator. Vaccinations included in these studies were pneumococcal (15/15), influenza (14/15), hepatitis A/B (5/15), Human Papilloma (3/15), tetanus (2/15), and Herpes Zoster (1/15). For PCV rates, 9/15 study endpoints were based on chart audits and 6/15 were patient reported (see supplemental Documents). Regardless of the intervention, there was an increase in PCV rates in 14/15 interventions compared to no intervention (Fig. 3).
Pre- and Post-intervention pneumococcal vaccination rates in inflammatory bowel disease studies. There was an increase in vaccination rates noted with all interventions except one. *Feuerstein J et al. was divided into two as there were two different groups: one that was an academic practice and the other a private practice
RA
As shown in Fig. 2, our RA literature search identified 2955 citations. After removal of duplicate studies, screening, and full-text review, 8 studies met the inclusion criteria for the systematic review (Table 2) [31,32,33,34,35,36,37,38]. The studies were published between 2009 and 2020 in the USA (n = 8) or Europe (n = 2). A total of 12,950 patients and at least 52 rheumatologists were studied (63.0% of the studies did not document the number of rheumatologists). Practice settings varied among academic centers (n = 4), a combination of academic and community practices (n = 3), and a tertiary care center (n = 1). All studies were pre-post-test interventions (8/8). Half of the studies were combined patient- and physician-oriented (4/8) and the remaining were physician-oriented (3/8) and barrier-oriented with a nurse navigator (1/8). Vaccinations included in these studies were pneumococcal (6/8), influenza (5/8), and Herpes Zoster (2/8). For PCV rates, 4/6 study endpoints were based on chart audits, 1/6 was patient reported and 1/6 was both chart audit and patient reported (see supplemental Documents). There was an increase in PCV rates for all interventions compared to no intervention (Fig. 4).
Meta-Analysis
IBD Meta-Analysis
Of the 15 studies in the IBD literature search, 14 studies were included in the meta-analysis. One study was entered as two separate entries because study participants were divided between academic and private practice setting [25]. For the 14 studies, there were 508 patients in the pre-intervention arm (440 in the patient and/or physician-oriented and 68 in the navigator-oriented) and 1224 patients in the post-intervention arm (1111 in the patient- and/or physician-oriented and 113 in the navigator-oriented). As shown in Fig. 5, all interventions had increased PCV uptake compared with no intervention (OR, 4.74; 95% CI, 2.94–7.64, I2 = 90%). The greatest effect was seen in barrier-oriented studies (OR, 12.68; 95% CI, 2.21–72.62, I2 = 92%).
Pooled estimates of association between interventions and vaccination uptake in inflammatory bowel disease patients. Compared with no intervention, all interventions (patient-oriented, /physician-oriented, combined patient + physician-oriented, or barrier-oriented) increased vaccination uptake [Odds ratio 4.74; 95% confidence interval, 2.94–7.64, I2 = 90%]. The greatest effect was seen in barrier-oriented studies [Odds ratio, 12.68; 95% confidence interval, 2.21–72.62, I2 = 92%]
RA Meta-Analysis
Of the 8 studies in the RA literature search, 6 studies were included in the meta-analysis. One study was entered as two separate entries because there were two different sites at which patients were studied [34]. For the 6 studies, there were 3,215 patients in the pre-intervention arm and 4,399 patients in the post-intervention arm. As shown in Fig. 6, all interventions had increased PCV uptake compared to no intervention (OR, 2.74; 95% CI, 1.80–4.17, I2 = 95%).
Pooled estimates of association between interventions and vaccination uptake in rheumatoid arthritis patients. All interventions (patient-oriented, physician-oriented, combined patient + physician-oriented, or barrier-oriented) increased vaccination uptake [Odds ratio, 2.74; 95% confidence interval, 1.80–4.17, I2 = 95%] compared to no intervention
Influenza Vaccination
Influenza vaccination was the second most studied vaccination. In the IBD studies 14 out of 15 studies and in the RA studies 5 out of 7 studies included influenza vaccination [16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38]. Pre-intervention vaccination rates for the annual influenza vaccination was higher than the baseline pre-intervention rates for pneumococcal vaccination in 14 out of the 19 studies (11 out of the 14 IBD studies and 3 out of 5 RA studies) as depicted in Tables 3 and 4 [16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38]. In the IBD studies, all intervention types (Patient-Oriented, Physician-Oriented, Combined Patient- + Physician-Oriented, and Barrier-Oriented) increased in vaccination rates in IBD. Of the 3 RA studies, only one study showed an increased in vaccination rate post-intervention for influenza vaccination [34]. Of the remaining two studies, one study the pre- and post-intervention vaccination rates remained exactly the same [31] and the other actually had a drop in post-intervention vaccination rate by 4% [32].
Discussion
From our knowledge, this is the first systematic review and meta-analysis to assess interventions to increase PCV rates in both IBD and RA patients. After an extensive review of over 8,580 studies, a detailed analysis of 23 articles was completed. Our study suggests that any intervention intended to increase PCV rates may be beneficial. Additionally, our data may provide more emphasis on the need for barrier-oriented interventions, such as healthcare navigators.
Patients with IBD are noted to be at an increased risk of vaccine-preventable illnesses, such as influenza, pneumococcal pneumonia, and even Herpes Zoster infections [5, 28, 39, 40]. Studies have suggested that many factors may cause vaccination rates to be low, such as lack of both patient and physician awareness of guidelines and both institutional and societal barriers that may all lead to decrease vaccination rates [8, 9]. Furthermore, a primary hurdle in optimal vaccination rates is patient perceptions of vaccinations. Over the years many models, such as Health Beliefs Model and Theory of Planned Behavior, have been used to study psychosocial predictors of vaccine behaviors [41, 42]. The Health Belief Model looked at social and behavioral determinants for patients’ behaviors to see how they affected how they affected a patient’s willingness to adopt disease prevention strategies or take screening tests [41]. Overcoming these barriers for our IBD and RA populations is of upmost importance, as vaccine-preventable diseases accounted for $27 billion dollars in treatment [1]. For example, pneumococcal pneumonia results in an estimated 150,000 hospitalizations each year in the USA with approximately 3000 to 4000 deaths from invasive forms of pneumococcal meningitis and bacteremia [43]. In the era of COVID-19, it is especially important to understand the interventions that may best increase vaccination rates.
Similar to previous studies, our data suggest that vaccination rates remain suboptimal in these patient populations. By performing a meta-analysis, we were able to highlight the importance of using multiple varying interventions to increase PCV rates in our patients. Specifically, pooling data from both IBD and RA patients allowed us to identify a possible trend that increasing intervention intensity (i.e., progressing from patient-oriented interventions, to physician-oriented interventions, to combined patient–physician interventions, and finally barrier-oriented interventions [such as navigators]) resulted in increased PCV uptake. Additionally, a recently published systematic review was consistent with our conclusion that interventions can increase adherence to preventative care in patients with IBD [44].
Barrier-oriented interventions, including healthcare navigation (i.e., designated nursing/medical assistant), outreach programs, and patient and physician education, have been implemented in many diseases and preventative strategies [45, 46]. For example, navigation systems have been suggested to be one of the most impactful interventions in colorectal cancer screening [47] and is the main component of multitarget stool DNA testing [48]. Additionally, other systems have used navigation to improve varying outcomes, such as treatment adherence for cancer patients and controlling hemoglobin A1c [46]. While our study highlights that barrier-oriented strategies can greatly improve PCV rates, we do not want to underemphasize that other modalities such as physician and patient education and systematic interventions can also be very effective and resources should best be evaluated by each institution.
Our study has many strengths. Our reproducible and comprehensive search strategy across multiple biomedical databases identified over potential 8000 articles. This highly sensitivity/low specificity search in combination with hand searching relevant journals led to a small but robust evidentiary foundation for our study. Furthermore, this study incorporated two similar immune-mediated disease states, IBD and RA, which to our knowledge has not been previously done to answer these questions.
Despite, the similarity in IBD and RA as being immune-medicated disease states, both the populations are different. In general, patients with RA are older females and likely have other comorbidities that would recommend them to have a pneumococcal vaccination. Limitations of our study included incomplete reporting of vaccination status prior to and after intervention; in most of the studies the vaccination status was based on self-reporting or documentation in chart audits that were based on patient reporting. Not all the patients received vaccinations at the various practices. There is also publication bias that is present. Other aspects that added to the high heterogeneity included the following: variations in primary documentation and in intervention populations and a lack of randomized controlled trials as most of the studies were quasi-experimental. There were also a small number of studies included in each sub-classification (patient-oriented vs physician-oriented vs combined patient- and physician-oriented); however, we were able to increase the power in our analysis by grouping these sub-classifications together. Despite the high heterogeneity, this study is clinically relevant as it shows that any intervention resulted in an increased uptake in PCV rates. This high heterogeneity is reflective of the different types of interventions used in the underlying studies of the meta-analysis. The existence of heterogeneity suggests that there may not be a single intervention effect but a distribution of intervention effects. Lastly, as previously stated, there is a discrepancy between internal medicine physicians and gastroenterologists on who’s responsibility vaccination are [6, 7]. It is possible that gastroenterologist and the rheumatologist view on who is responsible for recommendations of vaccination can also affect the vaccination rates. A meta-analysis was not completed on influenza vaccination for both IBD and RA data due to the lack of studies. In the IBD studies only one study was barrier-oriented and in the RA studies of the 3 studies that included influenza vaccination only one studies showed increased in vaccination rates.
The results of this study demonstrate that all of the studied interventions demonstrate evidence for increasing vaccination rates in patients with IBD and RA. Each healthcare organization and provider should evaluate their resources and assess which intervention is best suited for their patients. Our study also suggests that using barrier-oriented interventions, such as navigators, may have a substantial effect in increasing vaccination rates; however, any substantial conclusion cannot be made as we are limited by only two studies that assessed navigators. Further randomized controlled trials, especially in the USA, need to be conducted to further evaluate navigation and its degree of efficacy in our healthcare system.
Data availability
The data underlying this article are available in the article and in its online supplementary material. Data were obtained from prior publications which are cited in the references.
References
Farshidpour M, Charabaty A, Mattar MC. Improving immunization strategies in patients with inflammatory bowel disease. Ann Gastroenterol 2019;32:247–256.
Kirchgesner J, Lemaitre M, Carrat F, Zureik M, Carbonnel F, Dray-Spira R. Risk of serious and opportunistic infections associated with treatment of inflammatory bowel diseases. Gastroenterology 2018;155(337–46):e10.
Assessing the Cost of Vaccine-Preventable Diseases. The American Journal of Managed Care, December 2019 https://www.ajmc.com/view/assessing-the-cost-of-vaccinepreventable-diseases Accessed November 3, 2021.
Macaluso FS, Mazzola G, Ventimiglia M et al. Physicians’ knowledge and application of immunization strategies in patients with inflammatory bowel disease: a survey of the italian group for the study of inflammatory bowel disease. Digestion 2020;101:433–440.
Wasan SK, Baker SE, Skolnik PR, Farraye FA. A practical guide to vaccinating the inflammatory bowel disease patient. Am J Gastroenterol 2010;105:1231–1238.
Anyane-Yeboa A, Khiani A, Khamo N, Abdou S. The Primary Care Physician (PCP) and the gastroenterologist: what is your view on the management of patients with Inflammatory Bowel Disease (IBD)? Am J Gastroenterol 2015;110:S842.
Long MD, Gulati A, Wohl D, Herfarth H. Immunizations in pediatric and adult patients with inflammatory bowel disease: a practical case-based approach. Inflamm Bowel Dis 2015;21:1993–2003.
Melmed GY, Ippoliti AF, Papadakis KA et al. Patients with inflammatory bowel disease are at risk for vaccine-preventable illnesses. Am J Gastroenterol 2006;101:1834–1840.
Wasan SK, Calderwood AH, Long MD, Kappelman MD, Sandler RS, Farraye FA. Immunization rates and vaccine beliefs among patients with inflammatory bowel disease: an opportunity for improvement. Inflamm Bowel Dis 2014;20:246–250.
Zullow S, Farraye FA. Updates on vaccinating the inflammatory bowel disease patient. Gastroenterol Hepatol 2019;13:229–239.
McLeroy KR, Northridge ME, Balcazar H, Greenberg MR, Landers SJ. Reporting guidelines and the american journal of public health’s adoption of prefered reporting items for systematic reviews and meta-analyses. Am J of Public Health 2012;102:780–784.
Farraye FA, Melmed GY, Lichtenstein GR, Kane SV. ACG clinical guideline: preventive care in inflammatory bowel disease. Am J Gastroenterol 2017;112:241–258.
Kucharzik T, Ellul P, Greuter T et al. ECCO Guidelines on the prevention, diagnosis, and management of infections in inflammatory bowel disease. J Crohns Colitis 2021;15(6):879–913.
Covidence systematic review software, Veritas Health Innovation, Melbourne, Australia. www.covidence.org
Ioannidis JP, Trikalinos TA. The appropriateness of asymmetry tests for publication bias in meta-analyses: a large survey. CMAJ 2007;176:1091–1096.
Sitte J, Frentiu E, Baumann C et al. Vaccination for influenza and pneumococcus in patients with gastrointestinal cancer or inflammatory bowel disease: a prospective cohort study of methods for improving coverage. Aliment Pharmacol Ther 2019;49:84–90.
Ewelukwa O, Perez R, Carter LE, Fernandez A, Glover S. Incorporation of scribes into the inflammatory bowel disease clinic improves quality of care and physician productivity. Inflamm Bowel Dis 2018;24:552–557.
Christensen KR, Steenholdt C, Buhl SS, Ainsworth MA, Thomsen OO, Brynskov J. Systematic information to health-care professionals about vaccination guidelines improves adherence in patients with inflammatory bowel disease in anti-TNFalpha therapy. Am J Gastroenterol 2015;110:1526–1532.
Parker S, Chambers White L, Spangler C et al. A quality improvement project significantly increased the vaccination rate for immunosuppressed patients with IBD. Inflamm Bowel Dis 2013;19:1809–1814.
Walsh AJ, Weltman M, Burger D et al. Implementing guidelines on the prevention of opportunistic infections in inflammatory bowel disease. J Crohns Colitis 2013;7:e449–e456.
Coenen S, Weyts E, Jorissen C et al. Effects of education and information on vaccination behavior in patients with inflammatory bowel disease. Inflamm Bowel Dis 2017;23:318–324.
Greene L, Moreo K. Quality improvement education to improve performance on ulcerative colitis quality measures and care processes aligned with National Quality Strategy priorities. BMJ Qual Improv Rep 2015;4:u208829.w3554.
Greene L, Sapir T, Moreo K, Carter JD, Patel B, Higgins PD. Impact of quality improvement educational interventions on documented adherence to quality measures for adults with Crohn’s disease. Inflamm Bowel Dis 2015;21:2165–2171.
Valluru N, Kang L, Gaidos JKJ. Health maintenance documentation improves for veterans with IBD using a template in the computerized patient record system. Dig Dis Sci 2018;63:1782–1786. https://doi.org/10.1007/s10620-018-5093-5.
Feuerstein JD, Papamichael K, Popejoy S et al. Targeted physician education and standardizing documentation improves documented reporting with inflammatory bowel disease quality measures in a large academic and private practice. Dig Dis Sci 2018;63:36–45. https://doi.org/10.1007/s10620-017-4845-y.
Sapir T, Moreo K, Carter JD, Greene L, Patel B, Higgins PD. Continuing medical education improves gastroenterologists’ compliance with inflammatory bowel disease quality measures. Dig Dis Sci 2016;61:1862–1869. https://doi.org/10.1007/s10620-016-4061-1.
Farraye FA, Wasan SK, Noronha A et al. The use of an EHR patient portal (Mychart-Epic) in patients with inflammatory bowel disease. Crohn’s & Colitis 360 2019;1:3.
Reich JS, Miller HL, Wasan SK et al. Influenza and pneumococcal vaccination rates in patients with inflammatory bowel disease. Gastroenterol Hepatol 2015;11:396–401.
Bensinger A, Wilson F, Green P, Bloomfeld R, Dharod A. Sustained improvement in inflammatory bowel disease quality measures using an electronic health record intervention. Appl Clin Inform 2019;10:918–926.
Lee AJ, Kraemer DF, Smotherman C, Eid E. Providing our fellows in training with education on inflammatory bowel disease health maintenance to improve the quality of care in our health care system. Inflamm Bowel Dis 2016;22:187–193.
Murray K, Low C, O’Rourke A et al. A quality improvement intervention failed to significantly increase pneumococcal and influenza vaccination rates in immunosuppressed inflammatory arthritis patients. Clin Rheumatol 2020;39:747–754.
Baker DW, Brown T, Lee JY et al. A multifaceted intervention to improve influenza, pneumococcal, and herpes zoster vaccination among patients with rheumatoid arthritis. J Rheumatol 2016;43:1030–1037.
Sheth H, Moreland L, Peterson H, Aggarwal R. Improvement in herpes zoster vaccination in patients with rheumatoid arthritis: a quality improvement project. J Rheumatol 2017;44:11–17.
Ledwich LJ, Harrington TM, Ayoub WT, Sartorius JA, Newman ED. Improved influenza and pneumococcal vaccination in rheumatology patients taking immunosuppressants using an electronic health record best practice alert. Arthritis Rheum 2009;61:1505–1510.
Desai SP, Lu B, Szent-Gyorgyi LE et al. Increasing pneumococcal vaccination for immunosuppressed patients: a cluster quality improvement trial. Arthritis Rheum 2013;65:39–47.
Broderick R, Ventura I, Soroosh S, Franco L, Giles JT. Reducing missed opportunities for influenza vaccination in patients with rheumatoid arthritis: evaluation of a multisystem intervention. J Rheumatol 2018;45:1220–1228.
Gossec L, Soubrier M, Foissac F et al. Screening for and management of comorbidities after a nurse-led program: results of a 3-year longitudinal study in 769 established rheumatoid arthritis patients. RMD Open 2019;5:e000914.
Aguirre A, Trupin L, Margaretten M, Goglin S, Noh JH, Yazdany J. Using process improvement and systems redesign to improve rheumatology care quality in a safety net clinic. J Rheumatol 2020;47:1712–1720.
Long MD, Martin C, Sandler RS, Kappelman MD. Increased risk of pneumonia among patients with inflammatory bowel disease. Am J Gastroenterol 2013;108:240–248.
Long MD, Martin C, Sandler RS, Kappelman MD. Increased risk of herpes zoster among 108,604 patients with inflammatory bowel disease. Aliment Pharmacol Ther 2013;37:420–429.
Jones CL, Jensen JD, Scherr CL, Brown NR, Christy K, Weaver J. The health belief model as an explanatory framework in communication research: exploring parallel, serial, and moderated mediation. Health Commun 2015;30:566–576.
Ajzen I. The theory of planned behavior. Organ Behav Hum Decis Process 1991;50:179–211.
Fast Facts You Need to Know about Pneumococcal Disease. Center for Disease Control and Prevention, 2021 https://www.cdc.gov/pneumococcal/about/facts.html Accessed November 2, 2021.
Yu N, Basnayake C, Connell W et al. Interventions to improve adherence to preventive care in inflammatory bowel disease: a systematic review. Inflamm Bowel Dis. 2022;28:1177–1188.
Mailloux C, Halesey E. Patient navigators as essential members of the healthcare team: a review of the literature. J. Nurs. Care. 2018. https://doi.org/10.4172/2573-4571.1000122.
McBrien KA, Ivers N, Barnieh L et al. Patient navigators for people with chronic disease: a systematic review. PLoS One 2018;13:e0191980.
Dougherty MK, Brenner AT, Crockett SD et al. Evaluation of interventions intended to increase colorectal cancer screening rates in the United States: a systematic review and meta-analysis. JAMA Intern Med 2018;178:1645–1658.
Kisiel JB, Limburg PJ. Colorectal cancer screening with the multitarget stool DNA test. Am J Gastroenterol 2020;115:1737–1740.
Acknowledgments
Jalpa Patel and Dina Fakhouri had full access to all data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Funding
This work was not funded.
Author information
Authors and Affiliations
Contributions
JP, BW, AKW, FAF, and MP contributed to study concept and design. JP, DF, MN, AKW, FAF, and MP contributed to acquisition, analysis, or interpretation of data. JP, MN, MP, and JF contributed to drafting of the manuscript. Critical revision of the manuscript for important intellectual content: all authors. MN and AKW contributed to statistical analysis. JP, DF, and IK: administrative, technical, material support, and acquisition of data. MP contributed to study supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
An editorial commenting on this article is available at https://doi.org/10.1007/s10620-023-07905-5.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Patel, J., Noureldin, M., Fakhouri, D. et al. Interventions Increase Vaccination Rates in Inflammatory Bowel Disease and Rheumatoid Arthritis: A Systematic Review and Meta-Analysis. Dig Dis Sci 68, 2921–2935 (2023). https://doi.org/10.1007/s10620-023-07903-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-023-07903-7