Abstract
Background
Excision repair cross-complementation group 6-like (ERCC6L) is overexpressed in some malignancies; however, its role in hepatocellular carcinoma (HCC) remains to be further investigated.
Aims
In the present study, we explored the expression and function of ERCC6L in HCC.
Methods and Results
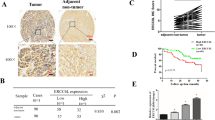
We investigated the expression of ERCC6L by microarray analysis, using the Cancer Genome Atlas database, and by HCC tissue microarray. The results showed that ERCC6L expression was upregulated in tumor specimens and HCC cell lines. High ERCC6L expression in tumor tissues was significantly correlated with poor prognosis and could serve as an independent prognostic indicator for HCC patients. Results of in vitro and in vivo assays revealed that ERCC6L substantially promoted cell proliferation, and our flow cytometry analysis revealed that this was accomplished by acceleration of the G1/S transition. Finally, gene set enrichment analysis and western blotting results indicated that ERCC6L might regulate HCC proliferation by activating p53 signaling.
Conclusions
Our study suggests that ERCC6L plays an important role in HCC proliferation and that it might serve as a promising therapeutic target in HCC.






Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. https://doi.org/10.3322/caac.21492.
Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet (London, England). 2018;391:1301–1314. https://doi.org/10.1016/s0140-6736(18)30010-2.
Yu XN, Chen H, Liu TT, Wu J, Zhu JM, Shen XZ. Targeting the mTOR regulatory network in hepatocellular carcinoma: Are we making headway? Biochim Biophys Acta. 2019;1871:379–391. https://doi.org/10.1016/j.bbcan.2019.03.001.
Xu Y, Chen X, Li Y. Ercc6l, a gene of SNF2 family, may play a role in the teratogenic action of alcohol. Toxicol Lett. 2005;157:233–239. https://doi.org/10.1016/j.toxlet.2005.02.013.
Albers E, Sbroggio M, Pladevall-Morera D, et al. Loss of PICH results in chromosomal instability, p53 activation, and embryonic lethality. Cell Rep. 2018;24:3274–3284. https://doi.org/10.1016/j.celrep.2018.08.071.
Baumann C, Korner R, Hofmann K, Nigg EA. PICH, a centromere-associated SNF2 family ATPase, is regulated by Plk1 and required for the spindle checkpoint. Cell. 2007;128:101–114. https://doi.org/10.1016/j.cell.2006.11.041.
Holtrich U, Wolf G, Brauninger A, et al. Induction and down-regulation of PLK, a human serine/threonine kinase expressed in proliferating cells and tumors. Proc Natl Acad Sci USA. 1994;91:1736–1740. https://doi.org/10.1073/pnas.91.5.1736.
Knecht R, Elez R, Oechler M, Solbach C, von Ilberg C, Strebhardt K. Prognostic significance of polo-like kinase (PLK) expression in squamous cell carcinomas of the head and neck. Can Res. 1999;59:2794–2797.
Takai N, Hamanaka R, Yoshimatsu J, Miyakawa I. Polo-like kinases (Plks) and cancer. Oncogene. 2005;24:287–291. https://doi.org/10.1038/sj.onc.1208272.
Wolf G, Elez R, Doermer A, et al. Prognostic significance of polo-like kinase (PLK) expression in non-small cell lung cancer. Oncogene. 1997;14:543–549. https://doi.org/10.1038/sj.onc.1200862.
Zhang G, Yu Z, Fu S, et al. ERCC6L that is up-regulated in high grade of renal cell carcinoma enhances cell viability in vitro and promotes tumor growth in vivo potentially through modulating MAPK signalling pathway. Cancer Gene Ther. 2019;26:323–333. https://doi.org/10.1038/s41417-018-0064-8.
Huang Y, Li W, Yan W, et al. Loss of PICH promotes chromosome instability and cell death in triple-negative breast cancer. Cell Death Dis. 2019;10:428. https://doi.org/10.1038/s41419-019-1662-6.
Liu J, Sun J, Zhang Q, Zeng Z. shRNA knockdown of DNA helicase ERCC6L expression inhibits human breast cancer growth. Mol Med Rep. 2018;18:3490–3496. https://doi.org/10.3892/mmr.2018.9317.
Pu SY, Yu Q, Wu H, et al. ERCC6L, a DNA helicase, is involved in cell proliferation and associated with survival and progress in breast and kidney cancers. Oncotarget. 2017;8:42116–42124. https://doi.org/10.18632/oncotarget.14998.
Ma XL, Jiang M, Zhao Y, et al. Application of serum annexin A3 in diagnosis, outcome prediction and therapeutic response evaluation for patients with hepatocellular carcinoma. Ann Surg Oncol. 2018;25:1686–1694. https://doi.org/10.1245/s10434-018-6402-0.
Yang XR, Xu Y, Yu B, et al. CD24 is a novel predictor for poor prognosis of hepatocellular carcinoma after surgery. Clin Cancer Res Off J Am Assoc Cancer Res. 2009;15:5518–5527. https://doi.org/10.1158/1078-0432.ccr-09-0151.
Jiang XM, Yu XN, Huang RZ, et al. Prognostic significance of eukaryotic initiation factor 4E in hepatocellular carcinoma. J Cancer Res Clin Oncol. 2016;142:2309–2317. https://doi.org/10.1007/s00432-016-2232-2.
Sun J, Zhou C, Ma Q, et al. High GCLC level in tumor tissues is associated with poor prognosis of hepatocellular carcinoma after curative resection. J Cancer. 2019;10:3333–3343. https://doi.org/10.7150/jca.29769.
Liu Z, Sun Q, Wang X. PLK1, a potential target for cancer therapy. Transl Oncol. 2017;10:22–32. https://doi.org/10.1016/j.tranon.2016.10.003.
Ran Z, Chen W, Shang J, et al. Clinicopathological and prognostic implications of polo-like kinase 1 expression in colorectal cancer: a systematic review and meta-analysis. Gene. 2019;721:144097. https://doi.org/10.1016/j.gene.2019.144097.
Affatato R, Carrassa L, Chila R, Lupi M, Restelli V, Damia G. Identification of PLK1 as a new therapeutic target in mucinous ovarian carcinoma. Cancers. 2020;. https://doi.org/10.3390/cancers12030672.
Bhola NE, Jansen VM, Bafna S, et al. Kinome-wide functional screen identifies role of PLK1 in hormone-independent. ER-positive breast cancer. Cancer Res. 2015;75:405–414. https://doi.org/10.1158/0008-5472.can-14-2475.
Helmke C, Becker S, Strebhardt K. The role of Plk3 in oncogenesis. Oncogene. 2016;35:135–147. https://doi.org/10.1038/onc.2015.105.
Massague J. G1 cell-cycle control and cancer. Nature. 2004;432:298–306. https://doi.org/10.1038/nature03094.
Mladenov E, Magin S, Soni A, Iliakis G. DNA double-strand-break repair in higher eukaryotes and its role in genomic instability and cancer: cell cycle and proliferation-dependent regulation. Semin Cancer Biol. 2016;37–38:51–64. https://doi.org/10.1016/j.semcancer.2016.03.003.
Tsai CH, Chen YJ, Yu CJ, et al. SMYD3-mediated H2A.Z.1 methylation promotes cell cycle and cancer proliferation. Can Res. 2016;76:6043–6053. https://doi.org/10.1158/0008-5472.can-16-0500.
Maddika S, Ande SR, Panigrahi S, et al. Cell survival, cell death and cell cycle pathways are interconnected: implications for cancer therapy. Drug Resist Updates Rev Comment Antimicrob Anticancer Chemother. 2007;10:13–29. https://doi.org/10.1016/j.drup.2007.01.003.
Murray JM, Carr AM. Integrating DNA damage repair with the cell cycle. Curr Opin Cell Biol. 2018;52:120–125. https://doi.org/10.1016/j.ceb.2018.03.006.
Ke Y, Huh JW, Warrington R, et al. PICH and BLM limit histone association with anaphase centromeric DNA threads and promote their resolution. EMBO J. 2011;30:3309–3321. https://doi.org/10.1038/emboj.2011.226.
Kmet LM, Cook LS, Magliocco AM. A review of p53 expression and mutation in human benign, low malignant potential, and invasive epithelial ovarian tumors. Cancer. 2003;97:389–404. https://doi.org/10.1002/cncr.11064.
Russo A, Bazan V, Iacopetta B, Kerr D, Soussi T, Gebbia N. The TP53 colorectal cancer international collaborative study on the prognostic and predictive significance of p53 mutation: influence of tumor site, type of mutation, and adjuvant treatment. J Clin Oncol Off J Am Soc Clin Oncol. 2005;23:7518–7528. https://doi.org/10.1200/jco.2005.00.471.
Gao Q, Zhu H, Dong L, et al. Integrated proteogenomic characterization of HBV-related hepatocellular carcinoma. Cell. 2019;179:561.e22–577.e22. https://doi.org/10.1016/j.cell.2019.08.052.
Abbas T, Dutta A. p21 in cancer: intricate networks and multiple activities. Nat Rev Cancer. 2009;9:400–414. https://doi.org/10.1038/nrc2657.
Georgakilas AG, Martin OA, Bonner WM. p21: a two-faced genome guardian. Trends Mol Med. 2017;23:310–319. https://doi.org/10.1016/j.molmed.2017.02.001.
Johnson LN, De Moliner E, Brown NR, et al. Structural studies with inhibitors of the cell cycle regulatory kinase cyclin-dependent protein kinase 2. Pharmacol Ther. 2002;93:113–124. https://doi.org/10.1016/s0163-7258(02)00181-x.
Wadler S. Perspectives for cancer therapies with cdk2 inhibitors. Drug Resist Updates Rev Comment Antimicrob Anticancer Chemother. 2001;4:347–367. https://doi.org/10.1054/drup.2001.0224.
Acknowledgments
This study was funded by the National Natural Science Foundation of China (Grant Numbers 81760497 and 81960446).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, G., Ma, J., Xiong, J. et al. Upregulation of Excision Repair Cross-Complementation Group 6-Like (ERCC6L) Promotes Tumor Growth in Hepatocellular Carcinoma. Dig Dis Sci 66, 1097–1109 (2021). https://doi.org/10.1007/s10620-020-06277-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-020-06277-4