Abstract
Background
HER2 protein expression indicates adverse prognosis in gastric adenocarcinoma (GCa). GCa HER2 positivity ranges from 10 to 22.8%. Similar data are scarce in South Asia and unavailable in Sri Lanka.
Aim
To evaluate HER2 protein expression, its clinicopathological relationship and survival in a Sri Lankan GCa cohort.
Methods
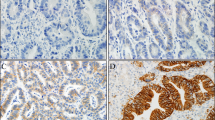
One hundred consecutive GCa patients were recruited prospectively for 2 years. Histological diagnosis was confirmed on endoscopic biopsies/gastrectomy specimens. Clinicopathological and overall survival data were collected. HER2 expression was assessed using immunohistochemistry. 2+ and 3+ scores were considered positive. HER2 expression and clinicopathological parameters were analyzed by Chi-squared test and multivariate analysis with logistic regression using SPSS-21. Kaplan–Meier method and log-rank test were used for survival analysis.
Results
Study includes 56 biopsies and 44 resections. Male/female ratio was 1.9:1. Mean age of diagnosis was 61.1 years (range 32–82). Majority tumors were proximally located (58%). HER2 positivity was 9%. Even though intestinal subtype predominated HER2 positivity was mostly among diffuse variant (14.8%). In multivariate analysis, mitotic count >5/hpf, high nuclear grade and tumor necrosis were significantly associated with HER2 positivity, while poor differentiation, signet cells, extracellular mucin, perineural invasion and pathological nodal metastasis (all p < 0.05) showed a correlation in univariate analysis. Mean follow-up duration was 37.4 weeks (range 0–104). HER2 positivity was associated with a significantly lower median overall survival (p = 0.046).
Conclusion
GCa HER2 positivity was 9%, associated with a lower median overall survival. Adverse histological features had a positive correlation with HER2 positivity. These histological features could direct patients for confirmatory HER2 testing in limited resource settings.






Similar content being viewed by others
Abbreviations
- HER-2:
-
Human epidermal growth factor receptor
- GCa:
-
Gastric adenocarcinoma
- CECT:
-
Contrast enhance tomography
- IHC:
-
Immunohistochemistry
- HPF:
-
High-power field
- CEP:
-
Centromeric probe
- FISH:
-
Fluorescence in situ hybridization
- SISH:
-
Silver in situ hybridization
- GOJ:
-
Gastroesophageal junctional
References
Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–E386. doi:10.1002/ijc.29210.
Kamangar F, Dores GM, Anderson WF. Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol. 2006;24:2137–2150.
Botterweck AA, Schouten LJ, Volovics A, Dorant E, van Den Brandt PA. Trends in incidence of adenocarcinoma of the oesophagus and gastric cardia in ten European countries. Int J Epidemiol. 2000;29:645–654.
Yang L. Incidence and mortality of gastric cancer in China. World J Gastroenterol. 2006;12:17–20.
Fock KM, Ang TL. Epidemiology of Helicobacter pylori infection and gastric cancer in Asia. J Gastroenterol Hepatol. 2010;25:479–486.
Guggenheim DE, Shah MA. Gastric cancer epidemiology and risk factors. J Surg Oncol. 2013;107:230–236.
American Cancer society. Stomach cancer detailed guide: stomach cancer survival rates. http://www.cancer.org/cancer/stomachcancer/. Accessed 11 Nov 2013.
Blakely AM, Miner TJ. Surgical considerations in the treatment of gastric cancer. Gastroenterol Clin North Am. 2013;42:337–357.
Kamangar F, Dores GM, Anderson WF. Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol. 2006;24:2137–2150.
Cunningham SC, Kamangar F, Kim MP, et al. Survival after gastric adenocarcinoma resection: eighteen-year experience at a single institution. J Gastrointest Surg. 2005;9:718–725.
Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Eng J Med. 2006;355:11–22.
Macdonald JS, Smalley SR, Benedetti J, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Eng J Med. 2001;345:725–730.
Seyedin S, Wang PC, Zhang Q, Lee P. Benefit of adjuvant chemoradiotherapy for gastric adenocarcinoma: a SEER population analysis. Gastrointest Cancer Res. 2014;7:82–90.
Wagner AD, Unverzagt S, Grothe W, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev. 2010;3:CD004064.
Rüschoff J, Dietel M, Baretton G, et al. HER2 diagnostics in gastric cancer—guideline validation and development of standardized immunohistochemical testing. Virchows Arch. 2010;457:299–307.
Park DI, Yun JW, Park JH, et al. HER-2/neu amplification is an independent prognostic factor in gastric cancer. Dig Dis Sci. 2006;51:1371–1379.
Kim KC, Koh YW, Chang HM, et al. Evaluation of HER2 protein expression in gastric carcinomas: comparative analysis of 1,414 cases of whole-tissue sections and 595 cases of tissue microarrays. Ann Surg Oncol. 2011;18:2833–2840.
Wang Q, Zhou J, Guo J, et al. Lin28 promotes Her2 expression and Lin28/Her2 predicts poorer survival in gastric cancer. Tumour Biol. 2014;35:11513–11521.
Jørgensen JT, Hersom M. HER2 as a prognostic marker in gastric cancer—a systematic analysis of data from the literature. J Cancer. 2012;3:137–144.
Nitta H, Kelly BD, Padilla M, et al. A gene-protein assay for human epidermal growth factor receptor 2 (HER2): brightfield tricolor visualization of HER2 protein, the HER2 gene, and chromosome 17 centromere (CEN17) in formalin-fixed, paraffin-embedded breast cancer tissue sections. Diagn Pathol. 2012;7:60.
Nitta H, Hauss-Wegrzyniak B, Lehrkamp M, et al. Development of automated brightfield double in situ hybridization (BDISH) application for HER2 gene and chromosome 17 centromere (CEN 17) for breast carcinomas and an assay performance comparison to manual dual color HER2 fluorescence in situ hybridization (FISH). Diagn Pathol. 2008;3:41.
Bang YJ, Van Cutsem E, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376(9742):687–697.
Hofmann M, Stoss O, Shi D, et al. Assessment of a HER2 scoring system for gastric cancer: results from a validation study. Histopathology. 2008;52:797–805.
Cancer incidence data. Sri Lanka 2010. Colombo: National cancer control programme; 2010.
Siriwardana HDRC, Pathirana A. Adenocarcinoma of the stomach in a tertiary care hospital in Sri Lanka. Ceylon Medical Journal. 2007;52:53–55.
Ba-Ssalamah A, Prokop M, Uffmann M, Pokieser P, Teleky B, Lechner G. Dedicated multidetector CT of the stomach: spectrum of diseases. Radiographics 2003;23:625–644.
Lauren P. The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma—an attempt at a histo-clinical classification. Acta Pathol Microbiol Scand. 1965;64:31–49.
Liang JW, Zhang JJ, Zhang T, Zheng ZC. Clinicopathological and prognostic significance of HER2 overexpression in gastric cancer: a meta-analysis of the literature. Tumour Biol. 2014;35:4849–4858.
Qiu MZ, Li Q, Wang ZQ, et al. Xu RH:HER2-positive patients receiving trastuzumab treatment have a comparable prognosis with HER2-negative advanced gastric cancer patients: a prospective cohort observation. Int J Cancer. 2014;134:2468–2477.
Akiyama T, Sudo C, Oqawara H, et al. The product of the human c-erbB-2 gene: a 185-kilodalton glycoprotein with tyrosine kinase activity. Science. 1986;232:1644–1646.
Kurokawa Y, Sugimoto N, Miwa H, et al. Phase II study of trastuzumab in combination with S-1 plus cisplatin in HER2-positive gastric cancer (HERBIS-1). Br J Cancer. 2014;110:1163–1168.
Allgayer H, Babic R, Gruetzner KU, et al. c-erbB-2 is of independent prognostic relevance in gastric cancer and is associated with the expression of tumor associated protease systems. J Clin Oncol. 2000;18:2201–2209.
De Carli DM, Da Rocha MP, Antunes LCM, Fagundes RB. Immunohistochemical expression of HER2 in adeno carcinoma of the stomach. Arq Gastroenterol. 2015;52:152–155.
Yano T, Doi T, Ohtsu A, et al. Comparison of HER2 gene amplification assessed by fluorescence in situ hybridization and HER2 protein expression assessed by immunohistochemistry in gastric cancer. Oncol Rep. 2006;15:65–71.
Sekaran A, Kandagaddala RS, Darisetty S, Lakhtakia S, Ayyagari S, et al. HER2 expression in gastric cancer in Indian population—an immunohistochemistry and fluorescent in situ hybridization study. Indian J Gastroenterol. 2012;31:106–110.
Grabsch H, Sivakumar S, Gray S, Gabbert HE, Müller W. HER2 expression in gastric cancer: rare, heterogeneous and of no prognostic value—conclusions from 924 cases of two independent series. Cell Oncol. 2010;32:57–65.
Leea HE, Parkb KU, Yooc SB, et al. Clinical significance of intratumoral HER2 heterogeneity in gastric cancer. Eur J Cancer. 2013;49:1448–1457.
Yang J, Luo H, Li Y, et al. Intratumoral heterogeneity determines discordant results of diagnostic tests for human epidermal growth factor receptor (HER) 2 in gastric cancer specimens. Cell Biochem Biophys. 2012;62:221–228.
Grillo F, Fassan M, Sarocchi F, Fiocca R, Mastracci L. HER2 heterogeneity in gastric/gastroesophageal cancers: from benchside to practice. World J Gastroenterol. 2016;22:5879–5887.
Shan L, Ying J, Ning L. HER2 expression and relevant clinicopathological features in gastric and gastroesophageal junction adenocarcinoma in a Chinese population. Diagn Pathol. 2013;8:76. doi:10.1186/1746-1596-8-76.
Boers JE, Meeuwissen H, Methorst N. HER2 status in gastro-oesophageal adenocarcinomas assessed by two rabbit monoclonal antibodies (SP3 and 4B5) and two in situ hybridization methods (FISH and SISH). Histopathology. 2011;58:383–394.
Nakajima M, Sawada H, Yamada Y, et al. The prognostic significance of amplification and overexpression of c-met and c-erb B-2 in human gastric carcinomas. Cancer. 1999;85:1894–1902.
Kumarasinghe MP, de Boer WB, Khor TS, et al. HER2 status in gastric/gastro-oesophageal junctional cancers: should determination of gene amplification by SISH use HER2 copy number or HER2:CEP17 ratio? Pathology. 2014;46:184–187.
Rüschoff J, Hanna W, Bilous M, et al. HER2 testing in gastric cancer: a practical approach. Mod Pathol. 2012;25:637–650.
Uprak TK, Attaallah W, Çelikel CA, Ayranc G, Yeğen C. HER-2 incidence in gastric cancer, its association with prognosis and clinicopathological parameters. Ulus Cerrahi Derg. 2015;31:207–213.
Janjigian YY, Werner D, Pauligk C, et al. Prognosis of metastatic gastric and gastroesophageal junction cancer by HER2 status: a European and USA International collaborative analysis. Ann Oncol. 2012. doi:10.1093/annonc/mds104.
Gravalos C, Jimeno A. HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol. 2008;19:1523–1529.
Tanner M, Hollmén M, Junttila TT, et al. Amplification of HER-2 in gastric carcinoma: association with Topoisomerase IIalpha gene amplification, intestinal type, poor prognosis and sensitivity to trastuzumab. Ann Oncol. 2005;16:273–278.
Qiu M, Zhou Y, Zhang X, et al. Lauren classification combined with HER2 status is a better prognostic factor in Chinese gastric cancer patients. BMC Cancer. 2014;14:823. doi:10.1186/1471-2407-14-823.
He C, Bian X-Y, Ni X-Z, et al. Correlation of human epidermal growth factor receptor 2 expression with clinicopathological characteristics and prognosis in gastric cancer. World J Gastroenterol. 2013;19:2171–2178.
Tafe LJ, Janjigian YY, Zaidinski M, et al. Human epidermal growth factor receptor 2 testing in gastroesophageal cancer: correlation between immunohistochemistry and fluorescence in situ hybridization. Arch Pathol Lab Med. 2011;135:1460–1465.
Acknowledgments
The authors wish to thank Dr. Gayani Ranaweera for contributing toward the literature survey and proposal writing for funding and Mrs. G. K. Wijesinghe (Staff technical officer) for technical assistance for the laboratory work. Authors wish to acknowledge The National Research Council, Sri Lanka, for funding this study. The Authors wish to thank Dr. Medhavini Dissanayake and Dr. Sameera Ravishan who helped in data collection and Dr. P. K. B. Mahesh for assistance rendered for statistical analysis.
Funding
The National Research Council of Sri Lanka.
Author information
Authors and Affiliations
Contributions
MDSL designed the study with contributions from MPK, SS, AS and DNS. DS was involved in laboratory work, data collection, analysis and writing the manuscript. MDSL critically evaluated and edited the manuscript with SS and MPK. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Availability of data and materials
The data will not be made available in order to protect the participant’s identity.
Conflict of interest
Authors declare that they have no conflict of interests.
Ethics approval and consent to participate
This study was conducted according to the principles expressed in the Declaration of Helsinki and approved by the Ethics review committee of Faculty of Medicine, University of Colombo and The National hospital of Sri Lanka.
Rights and permissions
About this article
Cite this article
Subasinghe, D., Sivaganesh, S., Samarsekera, A. et al. Human Epidermal Growth Factor Receptor-2 in Sri Lankan Gastric Carcinoma Patients with Clinicopathological Association and Survival. Dig Dis Sci 62, 2498–2510 (2017). https://doi.org/10.1007/s10620-017-4647-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-017-4647-2