Abstract
Background
There has been no study on the efficacy of lafutidine for patients with reflux esophagitis in Korea.
Aim
To evaluate the efficacy of a new-generation histamine-2 receptor antagonist, lafutidine, in comparison with famotidine in patients with reflux esophagitis.
Methods
This was a randomized, double-blind, non-inferiority trial enrolling patients with erosive esophagitis. The efficacy and safety of 20 mg lafutidine (treatment group) were compared with those of 40 mg famotidine (control group) and 20 mg omeprazole (reference group). The primary endpoint was the complete healing rates of reflux esophagitis on endoscopy after 8 weeks of treatment. The non-inferiority margin was assumed to be −15 %.
Results
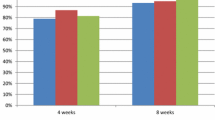
The healing rates of reflux esophagitis on endoscopy after 8 weeks of treatment were 70.14 % (101/144) in the lafutidine, 63.45 % (92/145) in the famotidine, and 85.71 % (126/147) in the omeprazole group. The difference in healing rates between the lafutidine and famotidine groups was 6.69 % (95 % confidence interval = [−4.14 to 17.52]). In addition, lafutidine was superior to famotidine in clinical improvement (53.73 % vs. 39.55 %, P = 0.0200).
Conclusions
Lafutidine was non-inferior to famotidine in healing of reflux esophagitis. Lafutidine, however, was superior to famotidine in terms of symptom relief of reflux esophagitis.


Similar content being viewed by others
References
Cho YS, Choi MG, Jeong JJ, et al. Prevalence and clinical spectrum of gastroesophageal reflux: a population-based study in Asan-si, Korea. Am J Gastroenterol. 2005;100:747–753.
Bai Y, Du Y, Zou D, et al. Gastroesophageal reflux disease questionnaire (GerdQ) in real-world practice: a national multicenter survey on 8065 patients. J Gastroenterol Hepatol. 2013;28:626–631.
Cho YK, Kim GH, Kim JH, et al. Diagnosis of gastroesophageal reflux disease: a systematic review. Korean J Gastroenterol. 2010;55:279–295.
Wong BC, Kinoshita Y. Systematic review on epidemiology of gastroesophageal reflux disease in Asia. Clin Gastroenterol Hepatol. 2006;4:398–407.
Khan M, Santana J, Donnellan C, et al. Medical treatments in the short term management of reflux oesophagitis. Cochrane Database Syst Rev. 2007;18:CD003244.
Moayyedi P, Talley NJ. Gastro-oesophageal reflux disease. Lancet. 2006;367:2086–2100.
Gray SL, LaCroix AZ, Larson J, et al. Proton pump inhibitor use, hip fracture, and change in bone mineral density in postmenopausal women: results from the Women’s Health Initiative. Arch Intern Med. 2010;170:765–771.
Herzig SJ, Howell MD, Ngo LH, et al. Acid-suppressive medication use and the risk for hospital-acquired pneumonia. JAMA. 2009;301:2120–2128.
Hess MW, Hoenderop JG, Bindels RJ, et al. Systematic review: hypomagnesaemia induced by proton pump inhibition. Aliment Pharmacol Ther. 2012;36:405–413.
Howell MD, Novack V, Grgurich P, et al. Iatrogenic gastric acid suppression and the risk of nosocomial Clostridium difficile infection. Arch Intern Med. 2010;170:784–790.
Sierra F, Suarez M, Rey M, et al. Systematic review: proton pump inhibitor-associated acute interstitial nephritis. Aliment Pharmacol Ther. 2007;26:545–553.
Inamori M, Togawa J, Iwasaki T, et al. Early effects of lafutidine or rabeprazole on intragastric acidity: which drug is more suitable for on-demand use? J Gastroenterol. 2005;40:453–458.
Nakano M, Ajioka H, Abe M, et al. Possible involvement of host defense mechanism in the suppression of rat acute reflux esophagitis by the particular histamine H2 receptor antagonist lafutidine. Pharmacology. 2012;90:205–211.
Saegusa Y, Ichikawa T, Iwai T, et al. Effects of acid antisecretory drugs on mucus barrier of the rat against 5-fluorouracil-induced gastrointestinal mucositis. Scand J Gastroenterol. 2008;43:531–537.
Edelsbrunner ME, Nakano M, Holzer P. Afferent signalling from the acid-challenged rat stomach is inhibited and gastric acid elimination is enhanced by lafutidine. BMC Gastroenterol. 2009;9:40.
Nakano M, Kitano S, Nanri M, et al. Lafutidine, a unique histamine H2-receptor antagonist, inhibits distention-induced gastric acid secretion through an H2 receptor-independent mechanism. Eur J Pharmacol. 2011;658:236–241.
Higuchi K, Watanabe T, Tominaga K, et al. Lafutidine can improve the quality of gastric ulcer healing in humans: a randomized, controlled, multicenter trial. Inflammopharmacology. 2006;14:226–230.
Ohara S, Haruma K, Kinoshita Y, et al. A double-blind, controlled study comparing lafutidine with placebo and famotidine in Japanese patients with mild reflux esophagitis. J Gastroenterol. 2010;45:1219–1227.
Armstrong D, Bennett JR, Blum AL, et al. The endoscopic assessment of esophagitis: a progress report on observer agreement. Gastroenterology. 1996;111:85–92.
Boeckxstaens GE, Rohof WO. Pathophysiology of gastroesophageal reflux disease. Gastroenterol Clin North Am. 2014;43:15–25.
DeVault KR, Castell DO. Updated guidelines for the diagnosis and treatment of gastroesophageal reflux disease. Am J Gastroenterol. 2005;100:190–200.
Holzer P. Neural emergency system in the stomach. Gastroenterology. 1998;114:823–839.
Akiba Y, Kaunitz JD. Lafutidine, a protective H(2) receptor antagonist, enhances mucosal defense in rat esophagus. Dig Dis Sci. 2010;55:3063–3069.
Fass R, Fennerty MB, Vakil N. Nonerosive reflux disease–current concepts and dilemmas. Am J Gastroenterol. 2001;96:303–314.
Habu Y, Maeda K, Kusuda T, et al. “Proton-pump inhibitor-first” strategy versus “step-up” strategy for the acute treatment of reflux esophagitis: a cost-effectiveness analysis in Japan. J Gastroenterol. 2005;40:1029–1035.
Reynolds JC. Individualized acute treatment strategies for gastroesophageal reflux disease. Scand J Gastroenterol Suppl. 1995;213:17–24.
Acknowledgments
This study was funded by Boryung Pharmaceutical Co., Ltd.
Conflict of interest
All authors reported no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kim, E.H., Lee, Y.C., Chang, Y.W. et al. Efficacy of Lafutidine Versus Famotidine in Patients with Reflux Esophagitis: A Multi-Center, Randomized, Double-Blind, Non-inferiority Phase III Trial. Dig Dis Sci 60, 1724–1732 (2015). https://doi.org/10.1007/s10620-014-3489-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-014-3489-4