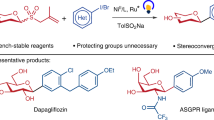
A series of glycosylated chrysin derivatives have been synthesized in good yields with simple procedures and mild reaction conditions. Six different kinds of sugar moieties were introduced through each ester linker.
Similar content being viewed by others
References
J. B. Xiao, T. T Chen, and H. Cao, Biotechnol. Adv., 33, 214 (2015).
M. Li and A. E. Hagerman, Curr. Drug Metab., 14, 432 (2013).
T. Lapidot, M. D. Walker, and J. Kanner, J. Agric. Food. Chem., 50, 7220 (2002).
N. Gresa-Arribas, J. Serratosa, J. Saura, and C. Sola, J. Neurochem., 115, 526 (2010).
J. Wang, J. Qiu, J. Dong, H. Li, M. Luo, X. Dai, Y. Zhang, B. Leng, X. Niu, S. Zhao, and X. Deng, J. Appl. Microbiol., 111, 1551 (2011).
P. G. Wadibhasme, M. M. Ghaisas, and P. A. Thakurdesai, Pharm. Biol., 49, 508 (2011).
M. Torres-Piedra, R. Ortiz-Andrade, R. Villalobos-Molina, N. Singh, J. L. Medina-Franco, S. P. Webster, M. Binnie, G. Navarrete-Vazquez, and S. Estrada-Soto, Eur. J. Med. Chem., 45, 2606 (2010).
E′. Brown, N. S. Hurd, S. McCall, and T. E. Ceremuga, AANA J., 75, 333 (2007).
G. A. Oliveira, E. R. Ferraz, A. O. Souza, R. A. Lourenco, D. P Oliveira, and D. J. Dorta, J. Toxicol. Environ. Health. A, 75, 1000 (2012).
A. Pick, H. Muller, R. Mayer, B. Haenisch, I. K. Pajeva, M. Weigt, H. Bonisch, C. E. Muller, and M. Wiese, Bioorg. Med. Chem., 19, 2090 (2011).
R. Anandhi, P. A. Thomas, and P. Geraldine, Mol. Cell. Biochem., 385, 103 (2014).
G. Pushpavalli, P. Kalaiarasi, C. Veeramani, and K. V. Pugalendi, Eur. J. Pharmacol., 631, 36 (2010).
A. D. Kandhare, V. Shivakumar, A. Rajmane, P. Ghosh, and S. L. Bodhankar, J. Nat. Med., 68, 586 (2014).
P. A. Tsuji, R. N. Winn, and T. Walle, Chem. Biol. Interact., 164, 85 (2006).
U. K. Walle, A. Galijatovic, and T. Walle, Biochem. Pharmacol., 58, 431 (1999).
S. P. Bondarenko and M. S. Frasinyuk, Chem. Nat. Compd., 49, 841 (2013).
X. Zheng, W. D. Meng, Y. Y. Xu, J. G. Cao, and F. L Qing, Bioorg. Med. Chem. Lett., 13, 881 (2003).
H. A. Mohammed, L. A. Ba, T. Burkholz, E. Schumann, B. Diesel, J. Zapp, A. K. Kiemer, C. Ries, R. W. Hartmann, M. Hosny, and C. Jacob, Nat. Prod. Commun., 6, 31 (2011).
H. Che, H. Lim, H. P. Kim, and H. Park, Eur. J. Med. Chem., 46, 4657 (2011).
M. V. Veselovskaya, M. M. Garazd, A. S. Ogorodniichuk, Ya. L. Garazd, and V. P. Khilya, Chem. Nat. Compd., 44, 704 (2008).
C. R. Bertozzi and L. L. Kiessling, Science, 291, 2357 (2001).
P. H. Seeberger and D. B. Werz, Nat. Rev. Drug Discov., 4, 751 (2005).
X. F. Fan, L. L. Jing, H. P. Ma, P. C. Fan, and Z. P. Jia, Chem. Reagents, 35, 975 (2013).
F. F. Zhang, L. L. Gan, and C. H. Zhou, Bioorg. Med. Chem. Lett., 20, 1881 (2010).
Y. Luo, Y. H. Lu, L. L. Gan, C. H. Zhou, J. Wu, R. X. Geng, and Y. Y. Zhang, Arch. Pharm. (Weinheim), 342, 386 (2009).
Acknowledgment
Financial support from the National Natural Science Foundation of China (81202458), the China Postdoctoral Science Foundation (2012M521926), and the Scientific Research Foundation of Gansu Province (1308RJYA061) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 4, July–August, 2016, pp. 522–528.
Rights and permissions
About this article
Cite this article
Fei, G., Fan, X., Ma, H. et al. Synthesis of Glycosylated Chrysin Derivatives Via Ester Linkers. Chem Nat Compd 52, 602–610 (2016). https://doi.org/10.1007/s10600-016-1721-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-016-1721-5