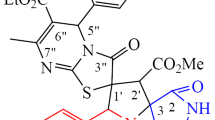
A novel series of dispiro(indolinonepyrrolidineiminothiazolidinones) were synthesized by 1,3-dipolar cycloaddition of 5-arylmethylidene-2-iminothiazolidin-4-ones and azomethine ylides generated from N-alkylamino acids and isatin derivatives. A number of iminothiazolidinones and dispiro compounds were tested for antiproliferative activity. Seven compounds showed moderate antiproliferative activity at micromolar concentrations against cancer cell lines.


Similar content being viewed by others
References
(a) Nirwan, S.; Chahal, V.; Kakkar, R. J. Heterocycl. Chem. 2019, 56, 1239. (b) Mech, D.; Kurowska, A.; Trotsko, N. Int. J. Mol. Sci. 2021, 22, 11533. (c) Kumar, H.; Aggarwal, N.; Marwaha, M. G.; Deep, A.; Chopra, H.; Matin, M. M.; Roy, A.; Emran, T. B.; Mohanta, Y. K.; Ahmed, R.; Mohanta, T. K.; Saravanan, M.; Marwaha, R. K.; Al-Harrasi, A. Molecules 2022, 27, 6763. (d) Izmest'ev, A. N.; Streltsov, A. A.; Kravchenko, A. N.; Gazieva, G. A. Chem. Heterocycl. Compd. 2022, 58, 483.
(a) Rabelo Pessoa de Siqueira, L.; Teixeira de Moraes Gomes, P. A.; de Lima Ferreira, L. P.; Barreto de Melo Rêgo, M. J.; Lima Leite, A. C. Eur. J. Med. Chem. 2019, 170, 237. (b) Szczepański, J.; Tuszewska, H.; Trotsko, N. Molecules 2022, 27, 3750. (c) Gazieva, G. A.; Izmest'ev, A. N. Chem. Heterocycl. Compd. 2015, 50, 1515. (d) Kovaleva, K.; Mamontova, E.; Yarovaya, O.; Zakharova, O.; Zakharenko, A.; Lavrik, O.; Salakhutdinov, N. Mol. Diversity 2021, 25, 2389.
(a)Tratrat, C.; Petrou, A.; Geronikaki, A.; Ivanov, M.; Kostić, M.; Soković, M.; Vizirianakis, I. S.; Theodoroula, N. F.; Haroun, M. Molecules 2022, 27, 1930. (b) Trotsko, N. Eur. J. Med. Chem. 2021, 215, 113266.
Levshin, I. B.; Simonov, A. Y.; Lavrenov, S. N.; Panov, A. A.; Grammatikova, N. E.; Alexandrov, A. A.; Ghazy, E. S. M. O.; Savin, N. A.; Gorelkin, P. V.; Erofeev, A. S.; Polshakov, V. I. Pharmaceuticals 2022, 15, 563.
Ma, L.; Xie, C.; Ma, Y.; Liu, J.; Xiang, M.; Ye, X.; Zheng, H.; Chen, Z.; Xu, Q.; Chen, T.; Chen, J.; Yang, J.; Qiu, N.; Wang, G.; Liang, X.; Peng, A.; Yang, S.; Wei, Y.; Chen L. J. Med. Chem. 2011, 54, 2060.
(a) Izmest'ev, A. N.; Gazieva, G. A.; Kolotyrkina, N. G.; Daeva, E. D.; Kravchenko, A. N. Chem. Heterocycl. Compd. 2020, 56, 1569. (b) Izmest'ev, A. N.; Streltsov, A. A.; Karnoukhova, V. A.; Kolotyrkina, N. G.; Strelenko, Y. A.; Kravchenko, A. N.; Gazieva, G. A. ChemistrySelect 2022, 7, e202104128. (c) Song, Y.-X.; Du, D.-M. Org. Biomol. Chem. 2020, 18, 6018.
(a) Murugan, R.; Anbazhagan, S.; Narayanan, S. S. Eur. J. Med. Chem. 2009, 44, 3272. (b) Toumi, A.; Boudriga, S.; Hamden, K.; Sobeh, M.; Cheurfa, M.; Askri, M.; Knorr, M.; Strohmann, C.; Brieger, L. Bioorg. Chem. 2021, 106, 104507.
Beloglazkina, A. A.; Mazhuga, A. G.; Beloglazkina, E. K.; Mefedova, S. R.; Karpov, N. A. RU Patent 2730286 C1.
(a) Izmest'ev, A. N.; Gazieva, G. A.; Karnoukhova, V. A.; Kravchenko, A. N. Org. Biomol. Chem. 2020, 18, 6905. (b) Izmest'ev, A. N.; Karnoukhova, V. A.; Larin, A. A.; Kravchenko, A. N.; Fershtat, L. L.; Gazieva, G. A. Int. J. Mol. Sci. 2022, 23, 13820.
(a) Izmest'ev, A. N.; Vasileva, D. A.; Melnikova, E. K.; Kolotyrkina, N. G.; Borisova, I. A.; Kravchenko, A. N.; Gazieva, G. A. New J. Chem. 2019, 43, 1038. (b) Izmest'ev, A. N.; Gazieva, G. A.; Anikina, L. V.; Pukhov, S. A.; Karnoukhova, V. A.; Kolotyrkina, N. G.; Kravchenko, A. N. New J. Chem. 2021, 45, 12271.
Lafayette, E. A.; Vitalino de Almeida, S. M.; Cavalcanti Santos, R. V.; Ferreira de Oliveira, J.; da Cruz Amorim, C. A.; Ferreira da Silva, R. M.; Galdino da Rocha Pitta, M.; da Rocha Pitta, I.; de Moura, R. O.; Bezerra de Carvalho, L., Jr.; Barreto de Melo Rêgo, M. J.; Alves de Lima, M. d. C. Eur. J. Med. Chem. 2017, 136, 511.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2023, 59(4/5), 309–316
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Izmest’ev, A.N., Streltsov, A.A., Kravchenko, A.N. et al. 5-Arylmethylidene-2-iminothiazolidin-4-ones in the synthesis of novel dispiro-fused oxindolepyrrolidineiminothiazolidinones. Chem Heterocycl Comp 59, 309–316 (2023). https://doi.org/10.1007/s10593-023-03198-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-023-03198-8