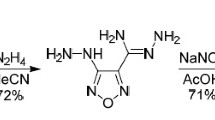
N,N-Dimethyl-3,5-dinitro-1H-pyrazol-4-amine was prepared by the reaction of 4-chloro-3,5-dinitropyrazole and DMF at 100°C for 4 h. Based on this compound, four new energetic derivatives, i.e., N,N,1-trimethyl-3,5-dinitro-1H-pyrazol-4-amine, 1,1'-methanediylbis- (N,N-dimethyl-3,5-dinitro-1H-pyrazol-4-amine), 1,1'-ethane-1,2-diylbis(N,N-dimethyl-3,5-dinitro-1H-pyrazol-4-amine), and 1,1'-propane- 1,3-diylbis(N,N-dimethyl-3,5-dinitro-1H-pyrazol-4-amine), were prepared through nucleophilic reactions. The structures of the compounds were confirmed by single crystal X-ray diffraction. One of the synthesized compounds has promising melting point and thermal stability and can be considered as potential melt-castable explosive.









Similar content being viewed by others
References
Chemical Rocket Propulsion. A Comprehensive Review of Energy Materials; Deluca, L. T.; Shimada, T.; Sinditsky, V. P.; Calabro, M., Eds.; Springer, 2017.
Lysien, K.; Stolarczyk, A.; Jarosz, T. Materials 2021, 14, 6657.
Wu, B.; Yang, L.; Zhai, D.; Ma, C.; Pei, C. FirePhysChem 2021, 1, 76.
Chen, F.; Song, S.; Wang, K.; Wang, Y.; Zhang, Q. Chem. Eng. J. 2022, 435, 135053.
Wingard, P. E.; Guzman, E. C.; Johnson, J. J.; Sabatini, G. W.; Drake, G. W.; Byrd, E. F. C. ChemPlusChem 2017, 82, 195.
Bennion, J. C.; Siddiqia, Z. R.; Matzger, A. J. ChemComm 2017, 53, 6065.
Wang, H.; Wang, X.; Luo, Y. Explos. Mater. 2021, 50, 1.
Cao, D.; Li. Y.; Du, Y.; Wang, J.; Li, Y. Chin. J. Energ. Mater. 2013, 21, 157.
Ravi, P.; Badgujar, D. M.; Gore, G. M.; Tewari, S. P.; Sikder, A. K. Propellants, Explos., Pyrotech. 2011, 36, 393.
Zheng, B.; Luo, G.; Shu, Y. Chem. Ind. Eng. Prog. 2013, 32, 1341.
Tan, Y.; Liu, Y.; Yang, Z. Shandong Chem. Ind. 2011, 40, 22.
Zhang, G.; Dong, H. Chin. J. Energ. Mater. 2010, 18, 604.
Dalinger, I. L.; Suponitsky, K. Yu.; Pivkina, A. N.; Sheremetev, A. B. Propellants, Explos., Pyrotech. 2016, 41, 789.
Bennion, J. C.; Jenkins, T. A. Propellants, Explos., Pyrotech. 2021, 46, 1421.
Johnson, E.C.; Sabatini, J. J.; Chavez, D. E.; Wells, L. A.; Banning, J. E.; Sausa, R. C.; Byrd, E. F. C.; Orlicki, J. A. ChemPlusChem 2020, 85, 237.
Klapötke, T. M.; Kofen, M.; Stierstorfer, J. Dalton Trans. 2021, 50, 13656.
Thaltiri, V.; Chavva, K.; Kumar, B. S.; Panda, P. K. New J. Chem. 2019, 43, 12318.
Cho, J. R.; Kim, K. J.; Cho, S. G.; Kim, J. K. J. Heterocycl. Chem. 2002, 39, 141.
Johnson, E. C.; Sabatini, J. J.; Chavez, D. E.; Sausa, R. C.; Byrd, E. F. C.; Wingard, L. A.; Guzman, P. E. Org. Process Res. Dev. 2018, 22, 736.
Wang, W.; Yang, W.; Ji, Y.; Ding, F. Chin. J. Explos. Propellants 2008, 6, 32.
Zhang, Y.; Zhou, C.; Wang, B.; Zhou, Y.; Xu, K.; Jia, S.; Zhao, F. Propellants, Explos., Pyrotech. 2014, 39, 809.
Jing, S.; Jiang, Z.; Jiao, Q.; Li, Z.; Liu, Y.; Yang, L. J. Energ. Mater. 2022, 40, 206.
Chen. F.; Liu, Y.; Wang, Y.; Zhang, Q. Chin. J. Energ. Mater. 2022, 28, 1109.
Chen, F.; Song, S.; Wang, Y.; Liu, Y.; Zhang, Q. Energ. Mater. Front. 2022, 1, 157.
Schmidt, R. D.; Lee, G. S.; Pagoria, P. F.; Mitchell, A. R.; Gilardi, R. J. Heterocycl. Chem. 2001, 38, 1227.
Pagoria, P. F.; Lee, G. S.; Mitchell, A. R.; Schmidt, R. D. Thermochim. Acta 2002, 384, 187.
Wang, Y.-L.; Zhao, F.-Q.; Ji, Y.-P.; Pan, Q.; Yi, J.-H.; An, T.; Wang, W.; Yu, T.; Lu, X.-M. J. Anal. Appl. Pyrolysis 2012, 98, 231.
Zaitsev, A. A.; Dalinger, I. L.; Shevelev, S. A. Russ. Chem. Rev. 2009, 78, 589.
Zhang, S.; Gao, Z.; Lan, D.; Jia, Q.; Liu, N.; Zhang, J.; Kou, K. Molecules 2020, 25, 3475.
Agarwal, A.; Chauhan, P. M. Synth. Commun. 2004, 34, 2925.
Maiti, D.; Fors, B. P.; Henderson, J. L.; Nakamura, Y.; Buchwald, S. L. Chem. Sci. 2011, 2, 57.
Dalinger, I. L.; Vatsadze, I. A.; Shkineva, T. K.; Popova, G. P.; Shevelev, S. A. Mendeleev Commun. 2010, 20, 355.
Dalinger, I. L.; Vatsadze, I. A.; Shkineva, T. K.; Popova, G. P.; Shevelev, S. A.; Nelyubina, Y. V. J. Heterocycl. Chem. 2013, 50, 911.
Yin, P.; Zhang, J.; Imler, G. H.; Parrish, D. A.; Shreeve, J. M. Angew. Chem. Int. Ed., 2017, 129, 8960.
Spackman, M. A.; Jayatilaka, D. CrystEngComm. 2009, 11, 19.
Xia, H.; Zhang, W.; Jin, Y.; Song, S.; Wang, K.; Zhang, Q. ACS Appl. Mater. Interfaces 2019, 11, 45914.
Agrawal, J. P. Propellants, Explos., Pyrotech. 2005, 30, 316.
Ma, Q.; Zhang, Z.; Yang, W.; Li, W.; Ju, J.; Fan, G. Energ. Mater. Front. 2021, 2, 69.
Yin, P.; He, C.; Shreeve, J. M. J. Mater. Chem. A 2016, 4, 1514.
Dolomanov, O. V.; Bourhis, L. J.; Gildea, R. J; Howard, J. A. K.; Puschmann, H. J. Appl. Crystallogr. 2009, 42, 339.
Sućeska, M. EXPLO5. Ver. 6.02; Zagreb, 2013.
Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. A.; Nakatsuji, H.; Caricato, M.; Li, X.; Hratchian, H. P.; Izmaylov, A. F.; Bloino, J.; Zheng, G.; Sonnenberg, J. L.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Montgomery, J. A., Jr.; Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd, J. J.; Brothers, E.; Kudin, K. N.; Staroverov, V. N.; Kobayashi, R.; Normand, J. ; Raghavachari, K.; Rendell, A.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Cossi, M.; Rega, N.; Millam, J. M.; Klene, M.; Knox, J. E.; Cross, J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Zakrzewski, V. G.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Dapprich, S.; Daniels, A. D.; Farkas, Ö.; Foresman, J. B.; Ortiz, J. V.; Cioslowski, J.; Fox, D. J. Gaussian 09. Rev. D.01; Gaussian, Inc.: Wallingford, 2009.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2022, 58(10), 493–499
Supplementary Information
ESM 1
(PDF 1782 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, J., Lv, R., Jiang, L. et al. Synthesis of N,N-dimethyl-3,5-dinitro-1H-pyrazol-4-amine and its energetic derivatives as promising melt-castable explosives. Chem Heterocycl Comp 58, 493–499 (2022). https://doi.org/10.1007/s10593-022-03118-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-022-03118-2