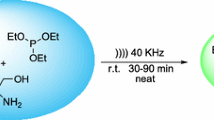
A novel and efficient procedure for the generation of quinazolinone phosphonate derivatives employing the reaction of euparin, isatin or its derivatives, primary amines, dialkyl acetylenedicarboxylates, trimethyl phosphite or triphenyl phosphite, and acidic solution of hydrogen peroxide in aqueous media at ambient temperature under ultrasonic irradiation was developed. Without ultrasonic irradiation, the reaction does not proceed and agitation of the reaction mixture is difficult. Some advantages of this procedure are: short time of reaction, high yields of products, easy isolation of products.


Similar content being viewed by others
References
(a) Kantam, M. L.; Rajasekhar, Ch. V.; Gopikrishna, G.; Reddy, K. R.; Choudary, B. M. Tetrahedron Lett. 2006, 47, 5965. (b) Liu, Y.-Q.; Li, L.-H.; Yang, L.; Li, H.-Y. Chem. Pap. 2010, 64, 533.
Kaur, N. Synth. Commun. 2018, 48, 1235.
Mohammadi Ziarani, Gh.; kheilkordi, Z.; Gholamzadeh Mol. Diversity 2020, 24, 771.
(a) Mečiarová, M.; Poláčková, V.; Toma, Š. Chem. Pap. 2002, 56, 208. (b) Meciarova, M.; Toma, S.; Babiak, P. Chem. Pap. 2004, 58, 104.
Tabatabaeian, K.; Mamaghani, M.; Mahmoodi, N. O.; Khorshidi, A. Catal. Commun. 2008, 9, 416.
Meciarova, M.; Toma, S.; Luche, J. L. Ultrason. Sonochem. 2001, 8, 119.
Vinatoru, M.; Bartha, E.; Badea, F.; Luche, J. L. Ultrason. Sonochem. 1998, 5, 27.
Ando, T.; Kimura, T.; Fujita, M.; Levêque, J.-M.; Luche, J.-L. Tetrahedron Lett. 2001, 42, 6865.
Cabello, N.; Cintas, P.; Luche, J.-L. Ultrason. Sonochem. 2003, 10, 25.
Kumar, V.; Sharma, A.; Sharma, M.; Sharma, U. K.; Sinha, A. K. Tetrahedron 2007, 63, 9718.
Sinha, A. K.; Sharma, A.; Joshi, B. P. Tetrahedron 2007, 63, 960.
Rostami-Charati, F.; Hossaini, Z. S. Synlett 2012, 2397.
(a) Hailes, H. C. Org. Process Res. Dev. 2007, 11, 114. (b) Jiang, B.; Cao, L.-J.; Tu, S.-J.; Zheng, W.-R.; Yu, H.-Z. J. Comb. Chem. 2009, 11, 612. (c) Tu, S.-J.; Zhang, X.-H.; Han, Z.-G.; Cao, X.-D.; Wu, S.-S.; Yan, S.; Hao, W.-J.; Zhang, G.; Ma, N. J. Comb. Chem. 2009, 11, 428. (d) Wu, H.; Lin, W.; Wan, Y.; Xin, H.-q.; Shi, D.-q.; Shi, Y.-h.; Yuan, R.; Bo, R.-c.; Yin, W. J. Comb. Chem. 2010, 12, 31. (e) Erdmenger, T.; Guerrero-Sanchez, C.; Vitz, J.; Hoogenboom, R.; Schubert, U. S. Chem. Soc. Rev 2010, 39, 3317. (b) Jiang, B.; Cao, L.-J.; Tu, S.-J.; Zheng, W.-R.; Yu, H.-Z. J. Comb. Chem. 2009, 11, 612. (c) Tu, S.-J.; Zhang, X.-H.; Han, Z.-G.; Cao, X.-D.; Wu, S.-S.; Yan, S.; Hao, W.-J.; Zhang, G.; Ma, N. J. Comb. Chem. 2009, 11, 428. (d) Wu, H.; Lin, W.; Wan, Y.; Xin, H.-q.; Shi, D.-q.; Shi, Y.-h.; Yuan, R.; Bo, R.-c.; Yin, W. J. Comb. Chem. 2010, 12, 31. (e) Erdmenger, T.; Guerrero-Sanchez, C.; Vitz, J.; Hoogenboom, R.; Schubert, U. S. Chem. Soc. Rev 2010, 39, 3317.
(a) Imamoto, T. Handbook of Organophosphorus Chemistry; Engel, R., Ed.; Marcel Dekker: New York, 1992, Chap. 1, p. 1. (b) Quin, L. D. A Guide to Organophosphorus Chemistry; Wiley-Interscience: New York, 2000. (c) Sasaki, M. Chirality in Agrochemicals; Kurihara, N., Miyamoto, J., Eds.; Wiley & Sons: Chichester, 1998, p. 85.
(a) Baumgartner, T.; Réeau, R. Chem. Rev. 2006, 106, 4681. (b) Engel, R. Chem. Rev. 1977, 77, 349. (c) Witt, M.; Roesky, H. W. Chem. Rev. 1994, 94, 1163. (d) Arduengo, A. J., III; Stewart, C. A. Chem. Rev. 1994, 94, 1215. (e) Cristau, H.-J. Chem. Rev. 1994, 94, 1299. (f) Gorenstein, D. G. Chem. Rev. 1994, 94, 1315. (g) Pietrusiewicz, K. M.; Zablocka, M. Chem. Rev. 1994, 94, 1375.
(a) Kim, Y.-C.; Brown, S. G.; Harden, T. K.; Boyer, J. L.; Dubyak, G.; King, B. F.; Burnstock, G.; Jacobson, K. A. J. Med. Chem. 2001, 44, 340. (b) Kumar, T. S.; Zhou, S.-Y.; Joshi, B. V.; Balasubramanian, R.; Yang, T.; Liang, B. T.; Jacobson, K. A. J. Med. Chem. 2010, 53, 2562. (c) Shie, J.-J.; Fang, J.-M.; Wang, S.-Y.; Tsai, K.-C.; Cheng, Y.-S. E.; Yang, A.-S.; Hsiao, S.-C.; Su, C.-Y.;Wong, C.-H. J. Am. Chem. Soc. 2007, 129, 11892.
(a) Kukhar, V. P.; Hudson, H. R. Aminophosphonic and Aminophosphinic Acids: Chemistry and Biological Activity; Wiley & Sons: Chichester, 2000. (b) Sawa, M.; Tsukamoto, T.; Kiyoi, T.; Kurokawa, K.; Nakajima, F.; Nakada, Y.; Yokota, K.; Inoue, Y.; Kondo, H.; Yoshino, K. J. Med. Chem. 2002, 45, 930. (c) Camp, N. P.; Perry, D. A.; Kinchington, D.; Hawkins, P. C. D.; Hitchcock, P. B.; Gani, D. Bioorg. Med. Chem. 1995, 3, 297. (d) Zhou, Y.; Yin, S.; Gao, Y.; Zhao, Y.; Goto, M.; Han, L.- B. Angew. Chem., Int. Ed. 2010, 49, 6852.
Allenberger, F.; Klare, L. J. Antimicrob. Chemother. 1999, 43, 211.
Allen, J. G.; Atherton, F. R.; Hall, M. J.; Hassall, C. H.; Holmes, S. W.; Lambert, R. W.; Nisbet, L. J.; Ringrose, P. S. Nature 1978, 272, 56.
Patel, D. V.; Rielly-Gauvin, K.; Ryono, D. E. Tetrahedron Lett. 1990, 31, 5587.
Bader, A. Aldrichimica Acta 1988, 21, 15.
Horsman, G. P., Zechel, D. L. Chem. Rev. 2017, 117, 5704.
Mikołajczyk, M. Pure Appl. Chem. 2019, 91, 811.
Kosolapoff, G. M.; Maier, L. Organic Phosphorus Compounds; Willey-Interscience, 1972.
Hudson, H. R. In The Chemistry of Organophosphorus Compounds: Primary Secondary and Tertiary Phosphines and Heterocyclic Organophosphorus(III) Compounds; Hantely, F. R.; Patai, S., Eds.; Wiley: New York, 1990, p. 386.
Engel R., Cohen, J. I. Synthesis of Carbon-Phosphorus Bonds; CRC Press: Boca Raton, 1998.
(a) Yavari, I.; Hossaini, Z.; Alizadeh, A. Monatsh. Chem. 2006, 137, 1083. (b) Alizadeh, A.; Yavari, I. Mendeleev Commun. 2005, 14, 154. (c) Yavari, I.; Alizadeh, A. Synthesis 2004, 237. (d) Yavari, I.; Alizadeh, A.; Anary-Abbasinejad, M. Tetrahedron. Lett. 2003, 44, 2877. (e) Yavari, I.; Anary- Abbasinejad, M.; Hossaini, Z. Org. Biomol. Chem. 2003, 1, 560.
Yavari, I.; Hossaini, Z.; Karimi, E. Monatsh. Chem. 2007, 138, 1267.
Anary-Abbasinejad, M.; Hassanabadi, A.; Mazraeh-Seffid, M. J. Chem. Res. 2007, 708.
Hallajian, S.; Alipour, S.; Foroughifar, N.; Khalilzadeh, M. A. Orient. J. Chem. 2014, 30, 1311.
Labaudiniere, L.; Burgada, R. Tetrahedron 1986, 42, 3521.
Bartroli, J.; Turmo, E.; Algueró, M.; Boncompte, E.; Vericat, M. L.; Conte, L.; Ramis, J.; Merlos, M.; Gracía-Rafanell, J.; Forn, J. J. Med. Chem. 1998, 41, 1869.
Zhu, S.; Wang, J.; Chandrashekar, G.; Smith, E.; Liu, X.; Zhang, Y. Eur. J. Med. Chem. 2010, 45, 3864.
Al-Rashood, S. T.; Hassan, G. S.; El-Messery, S. M.; Nagi, M. N.; Habib, E.-S. E.; Al-Omary, F. A. M.; El-Subbagh, H. I. Bioorg. Med. Chem. Lett. 2014, 24, 4557.
Hour, M.-J.; Huang, L.-J.; Kuo, S.-C.; Xia, Y.; Bastow, K.; Nakanishi, Y.; Hamel, E.; Lee, K.-H. J. Med. Chem. 2000, 43, 4479.
(a) Su, W.; Yang, B. Aust. J. Chem. 2002, 55, 695. (b) Shi, D.; Rong, L.; Wang, J.; Zhuang, Q.; Wang, X.; Hu, H. Tetrahedron Lett. 2003, 44, 3199.
Khurana, J. M.; Kukreja, G. J. Heterocycl. Chem. 2003, 40, 677.
Yale, H. L. J. Heterocycl. Chem. 1977, 14, 1357.
Moore, J. A.; Sutherland, G. J.; Sowerby, R.; Kelly, E. G.; Palermo, S.; Webster, W. J. Org. Chem. 1969, 34, 887.
Salehi, P.; Dabiri, M.; Zolfigol, M. A.; Baghbanzadeh, M. Synlett 2005, 1155.
Dabiri, M.; Salehi, P.; Otokesh, S.; Baghbanzadeh, M.; Kozehgary, G.; Mohammadi, A. A. Tetrahedron Lett. 2005, 46, 6123.
Khaleghi, F.; Bin Din, L.; Rostami Charati, F.; Yaacob, W. A.; Khalilzadeh, M. A.; Skelton, B.; Makha, M. Phytochem. Lett. 2011, 4, 254.
Rostami-Charati, F.; Hossaini, Z. Synlett 2012, 2397.
Dastoorani, P.; Maghsoodlou, M. T.; Khalilzadeh, M. A.; García-Granda, S.; Torre-Fernández, L.; Sarina, E. Heteroat. Chem. 2016, 27,102.
(a) Engle, R. Synthesis of Carbon-Phosphorus Bond; CRC Press: Boca Raton, 1988. (b) Organophosphorus Reagents in Organic Synthesis; Cadogan, J. I. G., Ed.; Academic Press: New York, 1977.
Organophosphorus Reagents in Organic Synthesis; Cadogan, J. I. G., Ed.; Academic Press: New York, 1979.
We gratefully acknowledge for spiritual support from Islamic Azad University of Qaemshahr Branch.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2020, 56(10), 1283–1291
Supplementary Information
ESM 1
(PDF 3707 kb)
Rights and permissions
About this article
Cite this article
Sharafian, S., Hossaini, Z., Rostami-Charati, F. et al. Green synthesis of novel phosphonate derivatives using ultrasonic irradiation. Chem Heterocycl Comp 56, 1283–1291 (2020). https://doi.org/10.1007/s10593-020-02812-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-020-02812-3