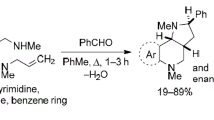
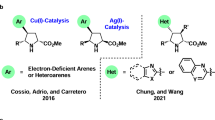
In this review, we consider 1,3-dipolar cycloaddition reactions between azomethine ylides and alkynes, resulting in the formation of pyrroles, as well as their hydrogenated and fused analogs. The references have been arranged according to the types of azomethine ylides and the methods used for their generation. The review is mainly focused on literature sources published since year 2000 and contains 133 references.

Similar content being viewed by others
Notes
Active lead was obtained by reducing lead acetate with sodium borohydride in aqueous acetic acid.
References
Synthetic Applications of 1,3-Dipolar Cycloaddition Chemistry Toward Heterocycles and Natural Products; Padwa, A.; Pearson, W. H., Eds.; Wiley: New York, 2002.
Kanemasa, S. Synlett 2002, 1371.
Nájera, C.; Sansano, J. M. Curr. Org. Chem. 2003, 7, 1105.
Gothelf, K. V.; Jørgensen, K. A. Chem. Rev. 1998, 98, 863.
Pellissier, H. Tetrahedron 2007, 63, 3235.
Stanley, L. M.; Sibi, M. P. Chem Rev. 2008, 108, 2887.
Bǎdoiu, A.; Brinkmann, Y.; Viton, F.; Kündig, E. P. Pure Appl. Chem. 2008, 80, 1013.
Gołȩbiewski, W. M.; Gucma, M. J. Heterocycl. Chem. 2008, 45, 1687.
Kanemasa, S. Heterocycles 2010, 82, 87.
Kissane, M.; Maguire, A. R. Chem. Soc. Rev. 2010, 39, 845.
Nájera, C.; Sansano, J. M.; Yus, M. J. Braz. Chem. Soc. 2010, 21, 377.
Xing, Y.; Wang, N.-X. Coord. Chem. Rev. 2012, 256, 938.
Hashimoto, T.; Maruoka, K. Chem. Rev. 2015, 115, 5366.
Nájera, C.; Sansano, J. M.; Yus, M. Org. Biomol. Chem. 2015, 13, 8596.
Bel'skaya, N. P.; Bakulev, V. A.; Fan, Z. Chem. Heterocycl. Compd. 2016, 52, 627. [Khim. Geterotsikl. Soedin. 2016, 52, 627.]
Döndas, H. A.; De Gracia Retamosa, M.; Sansano, J. M. Synthesis 2017, 2819.
Bdiri, B.; Zhao, B.-J.; Zhou, Z.-M. Tetrahedron: Asymmetry 2017, 28, 876.
Coldham, I.; Hufton, R. Chem. Rev. 2005, 105, 2765.
Pandey, G.; Dey, D.; Kumar Tiwari, S. Tetrahedron Lett. 2017, 58, 699.
Hladíková, V.; Váňa, J.; Hanusek, J. Beilstein J. Org. Chem. 2018, 14, 1317.
Wang, X.; Wang, X.; Wang, X.; Zhang, J.; Liu, C.; Hu, Y. Chem. Rec. 2017, 17, 1231.
Padwa, A. ARKIVOC 2018, (iv), 23.
Ess, D. H.; Houk, K. N. J. Am. Chem. Soc. 2008, 130, 10187.
Matsumoto, K.; Uchida, T.; Konishi, H.; Watanabe, Y.; Aoyama, K.; Asahi, M. Chem. Lett. 1987, 16, 807.
Díaz-Ortiz, A.; Díez-Barra, E.; de la Hoz, A.; Loupy, A.; Petit, A.; Sánchez, L. Heterocycles 1994, 38, 785.
Matsumoto, K.; Uchida, T.; Sugi, T.; Yagi, Y. Chem. Lett. 1982, 11, 869.
Shang, Y.; Zhang, M.; Yu, S.; Ju, K.; Wang, C.; He, X. Tetrahedron Lett. 2009, 50, 6981.
Belguedj, R.; Bouraiou, A.; Bouacida, S.; Merazig, H.; Chibani, A. Z. Naturforsch., B: J. Chem. Sci. 2015, 70, 885.
Belguedj, R.; Bouacida, S.; Merazig, H.; Belfaitah, A.; Bouraiou, A. Z. Naturforsch., B: J. Chem. Sci. 2015, 70, 555.
Liu, H.; He, D.; Sun, Z.; He, W.; Han, J.; Chen, J.; Deng, H.; Shao, M.; Zhang, H.; Cao, W. Tetrahedron 2018, 74, 135.
Nelina-Nemtseva, J. I.; Gulevskaya, A. V.; Pozharskii, A. F.; Nguyen, H. T. L.; Filatova, E. A. Tetrahedron 2016, 72, 2327.
Rotaru, A. V.; Druta, I. D.; Oeser, T.; Müller, T. J. J. Helv. Chim. Acta 2005, 88, 1798.
Georgescu, E.; Georgescu, F.; Danila, M.; Barbu, L.; Dumitrascu, F. Rev. Chim. (Bucharest) 2009, 60, 883.
Georgescu, E.; Georgescu, F.; Filip, P.; Dumitrescu, D. G.; Dumitrascu, F. Rev. Chim. (Bucharest) 2008, 59, 883.
Georgescu, E.; Georgescu, F.; Filip, P.; Popa, M. M.; Dumitrascu, F. Rev. Chim. (Bucharest) 2008, 59, 1224.
Georgescu, E.; Georgescu, F.; Filip, P.; Miu, B.; Dumitrascu, F. Rev. Chim. (Bucharest) 2009, 60, 377.
Sashida, H.; Kato, M.; Tsuchiya, T. Chem. Pharm. Bull. 1988, 36, 3826.
Tsuchiya, T.; Kato, M.; Sashida, H. Chem. Pharm. Bull. 1984, 32, 4666.
Mochulskaya, N. N.; Andreiko, A. A.; Charushin, V. N.; Shulgin, B. V.; Raikov, D. V.; Solomonov, V. I. Mendeleev Commun. 2001, 11, 19.
Georgescu, E.; Caira, M. R.; Georgescu, F.; Drăghici, B.; Popa, M. M.; Dumitrascu, F. Synlett 2009, 1795.
Belguedj, R.; Bouacida, S.; Merazig, H.; Belfaitah, A.; Chibani, A.; Bouraiou, A. Z. Naturforsch., B: J. Chem. Sci. 2016, 71, 231.
Dumitrascu, F.; Caira, M. R.; Georgescu, E.; Georgescu, F.; Draghici, C.; Popa, M. M. Heteroat. Chem. 2011, 22, 723.
Caira, M. R.; Popa, M. M.; Draghici, C.; Barbu, L.; Dumitrescu, D.; Dumitrascu, F. Tetrahedron Lett. 2014, 55, 5635.
Albota, F.; Drăghici, K.; Caira, M. R.; Dumitrascu, F. Tetrahedron 2015, 71, 9095.
Caira, M. R.; Georgescu, E.; Georgescu, F.; Albota, F.; Dumitrascu, F. Monatsh. Chem. 2011, 142, 743.
Dumitrascu, F.; Caproiu, M. T.; Georgescu, F.; Draghici, B.; Popa, M. M.; Georgescu, E. Synlett 2010, 2407.
Butler, R. N.; Coyne, A. G.; McArdle, P.; Cunningham, D.; Burke, L. A. J. Chem. Soc., Perkin Trans. 1 2001, 1391.
Dumitraşcu, F.; Mitan, C. I.; Drăghici, C.; Căproiu, M. T.; Răileanu, D. Tetrahedron Lett. 2001, 42, 8379.
Ito, S.; Tokimaru, Y.; Nozaki, K. Chem. Commun. 2015, 51, 221.
Dumitrascu, F.; Caira, M. R.; Draghici, C.; Caproiu, M. T.; Badoiu, A. Molecules 2005, 10, 321.
Dumitrascu, F.; Draghici, C.; Caira, M. R.; Badoiu, A.; Barbu, L.; Cristea, M. ARKIVOC 2005, (x), 165.
Dumitrascu, F.; Caira, M. R.; Draghici, C.; Caproiu, M. T.; Barbu, L. Rev. Chim. (Bucharest) 2009, 60, 851.
Dumitrascu, F.; Georgescu, E.; Caira, M. R.; Georgescu, F.; Popa, M.; Draghici, B.; Dumitrescu, D. G. Synlett 2009, 3336.
Yavari, I.; Naeimabadi, M. Synth. Commun. 2018, 48, 632
Padwa, A.; Austin, D. J.; Precedo, L.; Zhi, L. J. Org. Chem. 1993, 58, 1144.
Nicolescu, A.; Deleanu, C.; Georgescu, E.; Georgescu, F.; Iurascu, A.-M.; Shova, S.; Filip, P. Tetrahedron Lett. 2013, 54, 1486.
Georgescu, E.; Nicolescu, A.; Georgescu, F.; Teodorescu, F.; Marinescu, D.; Macsim, A.-M.; Deleanu, C. Beilstein J. Org. Chem. 2014, 10, 2377.
Dumitrescu, D.; Georgescu, E.; Caira, M. R.; Draghici, C.; Dumitrascu, F. Synlett 2017, 2241.
Moldoveanu, C.; Zbancioc, G.; Mantu, D.; Maftei, D.; Mangalagiu, I. PLoS One 2016, 11, e0156129. DOI: https://doi.org/10.1371/journal.pone.0156129
Georgescu, E.; Nicolescu, A.; Georgescu, F.; Shova, S.; Teodorescu, F.; Macsim, A.-M.; Deleanu, C. Synthesis 2015, 643.
Georgescu, E.; Nicolescu, A.; Georgescu, F.; Teodorescu, F.; Shova, S.; Marinoiu, A. T.; Dumitrascu, F.; Deleanu, C. Tetrahedron 2016, 72, 2507.
Feng, J.-J.; Zhang, J. ACS Catal. 2016, 6, 6651.
Gomes, P. J. S.; Nunes, C. M.; Pais, A. A. C. C.; Pinho e Melo, T. M. V. D.; Arnaut, L. G. Tetrahedron Lett. 2006, 47, 5475.
Dolbier, W. R.; Zheng, Z. J. Org. Chem. 2009, 74, 5626.
Novikov, M. S.; Khlebnikov, A. F.; Sidorina, E. S.; Kostikov, R. R. J. Chem. Soc., Perkin Trans.1 2000, 231.
Li, G.-Y.; Chen, J.; Yu, W.-Y.; Hong, W.; Che, C.-M. Org. Lett. 2003, 5, 2153.
Galliford, C. V.; Scheidt, K. A. J. Org. Chem. 2007, 72, 1811.
Rajasekaran, T.; Karthik, G.; Sridhar, B.; Subba Reddy, B. V. Org. Lett. 2013, 15, 1512.
Padwa, A.; Chen, Y.-Y.; Dent, W.; Nimmesgern, H. J. Org. Chem. 1985, 50, 4006.
Tran, G.; Meier, R.; Harris, L.; Browne, D. L.; Ley, S. V. J. Org. Chem. 2012, 77, 11071.
Ischay, M. A.; Takase, M. K.; Bergman, R. G.; Ellman, J. A. J. Am. Chem. Soc. 2013, 135, 2478.
Chen, S.; Bacauanu, V.; Knecht, T.; Mercado, B. Q.; Bergman, R. G.; Ellman, J. A. J. Am. Chem. Soc. 2016, 138, 12664.
Tsuge, O.; Hatta, T.; Tashiro, H.; Kakura, Y.; Maeda, H.; Kakehi, A. Tetrahedron 2000, 56, 7723.
Komatsu, M.; Ohno, M.; Tsuno, S.; Ohshiro, Y. Chem. Lett. 1990, 19, 575.
Iyoda, M.; Sultana, F.; Komatsu, M. Chem. Lett. 1995, 24, 1133.
Washizuka, K.-I.; Minakata, S.; Ryu, I.; Komatsu, M. Tetrahedron 1999, 55, 12969.
Komatsu, M.; Okada, H.; Yokoi, S.; Minakata, S. Tetrahedron Lett. 2003, 44, 1603.
Komatsu, M.; Okada, H.; Akaki, T.; Oderaotoshi, Y.; Minakata, S. Org. Lett. 2002, 4, 3505.
Okada, H.; Akaki, T.; Oderaotoshi, Y,; Minakata, S.; Komatsu, M. Tetrahedron 2003, 59, 197.
Oderaotoshi, Y.; Cheng, W.; Fujitomi, S.; Kasano, Y.; Minakata, S.; Komatsu, M. Org. Lett. 2003, 5, 5043.
Komatsu, M.; Kasano, Y.; Yonemori, J.-i.; Oderaotoshi, Y.; Minakata, S. Chem. Commun. 2006, 526.
Mancebo-Aracil, J.; Nájera, C.; Sansano, J. M. Org. Biomol. Chem. 2013, 11, 662.
Shi, F.; Luo, S.-W.; Tao, Z.-L.; He, L.; Yu, J.; Tu, S.-J.; Gong, L.-Z. Org. Lett. 2011, 13, 4680.
Shi, F.; Tao, Z.-L.; Yu, J.; Tu, S.-J. Tetrahedron: Asymmetry 2011, 22, 2056.
Shi, F.; Xing, G.-J.; Tan, W.; Zhu, R.-Y.; Tu, S. Org. Biomol. Chem. 2013, 11, 1482.
Tan, W.; Du, B.-X.; Guo, L.; Li, M.; Xing, G.-J.; Shi, F. J. Heterocycl. Chem. 2015, 52, 1055.
Shi, F.; Zhu, R.-Y.; Liang, X.; Tu, S.-J. Adv. Synth. Catal. 2013, 355, 2447.
Wu, P.; Gao, H.; Sun, J.; Yan, C.-G. Chin. Chem. Lett. 2017, 28, 329.
Tan, W.; Zhu, X.-T.; Zang, S.; Xing, G.-J.; Zhu, R.-Y.; Shi, F. RSC Adv. 2013, 3, 10875.
Bashiardes, G.; Safir, I.; Mohamed, A. S.; Barbot, F.; Laduranty, J. Org. Lett. 2003, 5, 4915.
Vidadala, S. R.; Waldmann, H. Tetrahedron Lett. 2015, 56, 3358.
Cardoso, A. L.; Kaczor, A.; Silva, A. M. S.; Fausto, R.; Pinho e Melo, T. M. V. D.; Rocha Gonsalves, A. M. d’A. Tetrahedron 2006, 62, 9861.
Gao, D.; Zhai, H.; Parvez, M.; Back, T. G. J. Org. Chem. 2008, 73, 8057.
Pardasani, R. T.; Pardasani, P.; Chaturvedi, V.; Yadav, S. K.; Saxena, A.; Sharma, I. Heteroat. Chem. 2003, 14, 36.
Singh, S. N.; Regati, S.; Paul, A. K.; Layek, M.; Jayaprakash, S.; Reddy, K. V.; Deora, G. S.; Mukherjee, S.; Pal, M. Tetrahedron Lett. 2013, 54, 5448.
Yang, F.; Sun, J.; Gao, H.; Yan, C.-G. RSC Adv. 2015, 5, 32786.
Shirsat, P. K.; Khomane, N. B.; Mali, P. R.; Maddi, R. R.; Nanubolu, J. B.; Meshram, H. M. ChemistrySelect 2017, 2, 11218.
Yavari, I.; Baoosi, L.; Halvagar, M. R. Mol. Diversity 2017, 21, 257.
Galbraith, A.; Small, T.; Boekelheide, V. J. Org. Chem. 1959, 24, 582.
Tsuge, O.; Kanemasa, S.; Hamamoto, T. Chem. Lett. 1983, 12, 763.
Sutcliffe, O. B.; Storr, R. C.; Gilchrist, T. L.; Rafferty, P. Tetrahedron 2000, 56, 10011.
Huang, H.-M.; Li, Y.-J.; Ye, Q.; Yu, W.-B.; Han, L.; Jia, J.-H.; Gao, J.-R. J. Org. Chem. 2014, 79, 1084.
Sugimoto, K.; Yamomoto, N.; Tominaga, D.; Matsuya, Y. Org. Lett. 2015, 17, 1320.
Sugimoto, K.; Hoshiba, Y.; Tsuge, K.; Matsuya, Y. Synthesis 2016, 1855.
Bobeck, D. R.; Warner, D. L.; Vedejs, E. J. Org. Chem. 2007, 72, 8506.
Vedejs, E.; Naidu, B. N.; Klapars, A.; Warner, D. L.; Li, V.-s.; Na, Y.; Kohn, H. J. Am. Chem. Soc. 2003, 125, 15796.
Gribble, G. W. In The Chemistry of Heterocyclic Compounds: Synthetic Applications of 1,3-Dipolar Cycloaddition Chemistry Toward Heterocycles and Natural Products; Padwa, A.; Pearson, W. H., Eds.; John Wiley & Sons: Hoboken, 2002, Vol. 59, p. 681.
Gribble, G. W. In The Chemistry of Heterocyclic Compounds, Oxazoles: Synthesis, Reactions, and Spectroscopy, Part A; Taylor, E. C.; Wipf, P., Eds.; John Wiley & Sons: Hoboken, 2003, Vol. 60, p. 473.
Miao, Q.; Sun, H. Chin. J. Org. Chem. 2016, 36, 913.
Reissig, H.-U.; Zimmer, R. Angew. Chem., Int. Ed. 2014, 53, 9708.
Padwa, A.; Burgess, E. M.; Gingrich, H. L.; Roush, D. M. J. Org. Chem. 1982, 47, 786.
Coppola, B. P.; Noe, M. C.; Schwartz, D. J.; Abdon, R. L., II; Trost, B. M. Tetrahedron 1994, 50, 93.
Roesler, P.; Fleury, J.-P. Bull. Soc. Chim. Fr. 1968, 631.
Clerin, D.; Meyer, B.; Fleury, J. P.; Fritz, H. Tetrahedron 1976, 32, 1055.
St. Cyr, D. J.; Arndtsen, B. A. J. Am. Chem. Soc. 2007, 129, 12366.
Krenske, E. H.; Houk, K. N.; Arndtsen, B. A; St. Cyr, D. J. J. Am. Chem. Soc. 2008, 130, 10052.
St-Cyr, D. J.; Morin, M. S. T.; Bélanger-Gariépy, F.; Arndtsen, B. A.; Krenske, E. H.; Houk, K. N. J. Org. Chem. 2010, 75, 4261.
Morin, M. S. T.; St-Cyr, D. J.; Arndtsen, B. A. Org. Lett. 2010, 12, 4916.
Dhawan, R.; Arndtsen, B. A. J. Am. Chem. Soc. 2004, 126, 468.
St. Cyr, D. J.; Martin, N.; Arndtsen, B. A. Org. Lett. 2007, 9, 449.
Morin, M. S. T.; St-Cyr, D. J.; Arndtsen, B. A.; Krenske, E. H.; Houk, K. N. J. Am. Chem. Soc. 2013, 135, 17349.
Shahvelayati, A. S.; Oladkazemi, M. Iran J. Org. Chem. 2013, 5, 1041.
Kayser, L. V.; Vollmer, M.; Welnhofer, M.; Krikcziokat, H.; Meerholz, K.; Arndtsen, B. A. J. Am. Chem. Soc. 2016, 138, 10516.
Tokuyama, H. J. Synth. Org. Chem., Jpn. 2015, 73, 1120.
Lopchuk, J. M.; Gribble, G. W. Tetrahedron Lett. 2015, 56, 3208.
Pandey, P. S.; Rao, T. S. Bioorg. Med. Chem. Lett. 2004, 14, 129.
Park, W. K. C.; Kennedy, R. M.; Larsen, S. D.; Miller, S.; Roth, B. D.; Song, Y.; Steinbaugh, B. A.; Sun, K.; Tait, B. D.; Kowala, M. C.; Trivedi, B. K.; Auerbach, B.; Askew, V.; Dillon, L.; Hanselman, J. C.; Lin, Z.; Lu, G. H.; Robertson, A.; Sekerke, C. Bioorg. Med. Chem. Lett. 2008, 18, 1151.
Sugimoto, K.; Miyakawa, Y.; Tokuyama, H. Tetrahedron 2015, 71, 3619.
Angle, S. R.; Qian, X. L.; Pletnev, A. A.; Chinn, J. J. Org. Chem. 2007, 72, 2015.
Erguven, H.; Leitch, D. C.; Keyzer, E. N.; Arndtsen, B. A. Angew. Chem., Int. Ed. 2017, 56, 6078.
Song, G.; Chen, D.; Su, Y.; Han, K.; Pan, C.-L.; Jia, A.; Li, X. Angew. Chem., Int. Ed. 2011, 50, 7791.
Vivanco, S.; Lecea, B.; Arrieta, A.; Prieto, P.; Morao, I.; Linden, A.; Cossío, F. P. J. Am. Chem. Soc. 2000, 122, 6078.
Freindorf, M.; Sexton, T.; Kraka, E.; Cremer, D. Theor. Chem. Acc. 2014, 133, 1423.
Acknowledgment
This work was performed with financial support from the Southern Federal University (internal grant BnGr-
07/2017-18).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor A. F. Pozharskii on his 80th birthday
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2018, 54(12), 1084–1107
Rights and permissions
About this article
Cite this article
Gulevskaya, A.V., Nelina-Nemtseva, J.I. 1,3-Dipolar cycloaddition reactions of azomethine ylides and alkynes. Chem Heterocycl Comp 54, 1084–1107 (2018). https://doi.org/10.1007/s10593-019-02398-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-019-02398-5