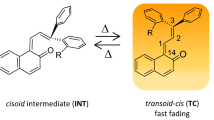
Two novel isomeric naphthopyrans substituted with phenyl (13-butyl-6,7-dimethoxy-3,3-bis(6-methoxybiphenyl-3-yl)-3,13-dihydrobenzo[h]indeno[2,1-f]chromen-13-ol) and 4-(naphthalen-1-yl)phenyl (13-butyl-6,7-dimethoxy-3,3-bis[6-methoxy-4'-(naphthalenyl)-biphenyl-3-yl]-3,13-dihydrobenzo[h]indeno[2,1-f]chromen-13-ol) moieties were synthesized. Their photochromism, electrochemical and fluorescent properties were investigated. They displayed faster color fading rate and larger fluorescence quantum yield than 13-butyl-6,7-dimethoxy-3,3-bis(4-methoxyphenyl)-3,13-dihydrobenzo[h]indeno[2,1-f]chromen-13-ol. Moreover, they showed excellent photochromic and fluorescent properties both in solution and in polymethylmethacrylate film. In addition, cyclic voltammetry tests showed that the aromatic substituents had a significant effect on the electrochemical behavior of the naphthopyran derivatives.





Similar content being viewed by others
References
(a) Photochromic Materials: Preparation, Properties and Applications; Tian, H.; Zhang, J., Eds.; Wiley-VCH: Weinheim, 2016. (b) New Frontiers in Photochromism; Irie, M.; Yokoyama, Y.; Seki, T., Eds.; Springer: Tokyo, 2013.
(a) Molecular Switches; Feringa, B. L.; Browne, W. R.; Eds.; Wiley-VCH: Weinheim, 2011. (b) Photochromism: Molecules and Systems; Dürr, H.; Bouas-Laurent, H., Eds.; Elsevier: Amsterdam, 2003.
(a) Sousa, C. M.; Berthet, J.; Delbaere, S.; Polónia, A.; Coelho, J. P. J. Org. Chem. 2017, 82, 12028. (b) Inagaki, Y.; Kobayashi, Y.; Mutoh, K.; Abe, J. J. Am. Chem. Soc. 2017, 139, 13429.
Ercole, F.; Malic, N.; Harrisson, S.; Davis, T. P.; Evans, R. A. Macromolecules 2010, 43, 249.
Berthet, J.; Coelho, P. J.; Carvalho, L. M.; Vermeersch, G.; Delbaere, S. J. Photochem. Photobiol., A 2009, 208, 180.
Moorthy, J. N.; Koner, A. L.; Samanta, S.; Roy, A.; Nau, W. M. Chem.–Eur. J. 2009, 15, 4289.
Ercole, F.; Davis, T. P.; Evans, R. A. Macromolecules 2009, 42, 1500.
Oliveira, M. M.; Salvador, M. A.; Delbaere, S.; Berthet, J.; Vermeersch, G.; Micheau, J. C.; Coelho, P. J.; Carvalho, L. M. J. Photochem. Photobiol., A 2008, 198, 242.
Sallenave, X.; Delbaere, S.; Vermeersch, G.; Saleh, A.; Pozzo, J.-L. Tetrahedron. Lett. 2005, 46, 3257.
Nabais, C. R. J. O. D.; Heron, B. M.; de Sousa, H. C.; Gil, M. H.; Sobral, A. J. F. N. J. Biomater. Sci., Polym. Ed. 2011, 22, 139.
Corns, S. N.; Partington, S. M.; Towns, A. D. Color. Technol. 2009, 125, 249.
Sousa, C. M.; Pina, J.; de Melo, J. S.; Berthet, J.; Delbaere, S.; Coelho, P. J. Org. Lett. 2011, 13, 4040.
Song, L.; Yang, Y.; Zhang, Q.; Tian, H.; Zhu, W. J. Phys. Chem. B 2011, 115, 14648.
Nelson, C. M.; Chopra, A.; Petrovskaia, O. G.; Knowles, D. B.; Van Gemert, B.; Kumar, A. US Patent US6296785 B1.
Liu, G.; Pu, S.; Wang, X. J. Photochem. Photobiol., A 2010, 214, 230.
Pu, S.; Li, H.; Liu, G.; Liu, W.; Cui, S.; Fan, C. Tetrahedron 2011, 67, 1438.
Juárez, R.; Gómez, R.; Segura, J. L.; Seoane, C. Tetrahedron Lett. 2005, 46, 8861.
Crosby, G. A.; Demas, J. N. J. Phys. Chem. A 1971, 75, 991.
Malic, N.; Campbell, J. A.; Evans, R. A. Macromolecules 2008, 41, 1206.
Bohne, C.; Fan, M. G.; Li, Z. J.; Liang, Y. C.; Lusztyk, J.; Scaiano, J. C. J. Photochem. Photobiol., A 1992, 66, 79.
Evans, R. A.; Hanley, T. L.; Skidmore, M. A. Nature Materials 2005, 4, 249.
Han, S.; Chen, Y. J. Mater. Chem. 2011, 21, 4961.
Ercole, F.; Malic, N.; Davis, T. P.; Evans, R. A. J. Mater. Chem. 2009, 19, 5612.
Yitzchaik, S.; Ratner, J.; Buchholtz, F.; Krongauz, V. Liq. Cryst. 1990, 8, 677.
Pardo, R.; Zayat, M.; Levy, D. Chem. Soc. Rev. 2011, 40, 672.
Van Gemert, B.; Bergomi, M.; Knowles, D. Mol. Cryst. Liq. Cryst. Sci. Technol., Sect. A 1994, 246, 67.
Nienhaus, G. U. Angew. Chem., Int. Ed. 2012, 51, 1312.
Bonifazi, D.; Scholl, M.; Song, F.; Echegoyen, L.; Accorsi, G.; Armaroli, N.; Diederich, F. Angew. Chem., Int. Ed. 2003, 42, 4966.
Browne, W. R.; de Jong, J. J. D.; Kudernac, T.; Walko, M.; Lucas, L. N.; Uchida, K.; van Esch, J. H.; Feringa, B. L. Chem.–Eur. J. 2005, 11, 6414.
Moriyama, Y.; Matsuda, K.; Tanifuji, N.; Irie, S.; Irie, M. Org. Lett. 2005, 7, 3315.
Zheng, C.; Pu, S.; Xu, J.; Luo, M.; Huang, D.; Shen, L. Tetrahedron 2007, 63, 5437.
Pu, S.; Yang, T.; Xu, J.; Shen, L.; Li, G.; Xiao, Q.; Chen, B. Tetrahedron 2005, 61, 6623.
Tsai, F.-C.; Chang, C.-C.; Liu, C.-L.; Chen, W.-C.; Jenekhe, S. A. Macromolecules 2005, 38, 1958.
Zhan, X.; Liu, Y.; Wu, X.; Wang, S.; Zhu, D. Macromolecules 2002, 35, 2529.
Perrier, A.; Maurel, F.; Aubard, J. J. Photochem. Photobiol., A 2007, 189, 167.
Kim, E.; Kim, M.; Kim, K. Tetrahedron 2006, 62, 6814.
The authors are grateful to the financial support from industry (1006-KFA17783). A project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information file containing IR, 1H and 13C NMR spectral data of all synthesized compounds, synthetic procedure of compound NOP1, and optical density change curves of compounds NOP1–3 in CH2Cl2 and PMMA is available on the journal website at http:// link.springer.com/journal/10593.
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2018, 54(9), 840–847
Electronic supplementary material
ESM 1
(PDF 2798 kb)
Rights and permissions
About this article
Cite this article
Zhao, Q., Yang, Y., Duan, Y. et al. Synthesis and photochromic properties of new naphthopyrans. Chem Heterocycl Comp 54, 840–847 (2018). https://doi.org/10.1007/s10593-018-2366-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-018-2366-z