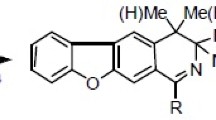
1-Ethynyl-6,18-endo-ethenodihydrothebainehydroquinone reacted with azides in the presence of CuSO4·5H2O and sodium ascorbate in DMF, forming the respective 1-(N-alkyl(arylalkyl)triazol-4-yl)-6,18-endo-ethenodihydrothebainehydroquinones, which underwent retrodiene cleavage upon heating in DMF, forming functionalized tetrahydrofuro[4,3,2-fg][3]benzazocines containing naphthohydroquinone and 1,2,3-triazolyl substituents.
Similar content being viewed by others
References
(a) Lewis, J. W.; Husbands, S. M. Curr. Pharm. Des. 2004, 10, 717. (b) Husbands, S. M. In Research and Development of Opioid-Related Ligands; Ko, M.-C.; Husbands, S. M., Eds.; American Chemical Society: New York, 2013, p. 127. (c) Hosztafi, S. Adv. Biosci. Biotechnol. 2014, 5, 704.
(a) Takemori, A. E.; Portoghese, P. S. Ann. Rev. Pharmacol. Toxicol. 1992, 32, 239. (b) Sharma, S. K.; Jones, R.; Metzger, T. G.; Ferguson, D. M.; Portoghese, P. S. J. Med. Chem. 2001, 44, 2073. (c) Schütz, J.; Dersch, C. M.; Horel, R.; Spetea, M.; Koch, M.; Meditz, R.; Greiner, E.; Rothman, R. B.; Schmidhammer, H. J. Med. Chem. 2002, 45, 5378.
(а) Tolstikova, T. G.; Davydova, V. A.; Lazareva, D. N.; Sarudiy, F. C.; Shults, E. E.; Tolstikov, G. A. Eur. J. Pharmacol. 1990, 183, 2336. (b) Tolstikov, G. A.; Shults, E. E.; Mukhametyanova, T. Sh.; Sultanova, V. S.; Spirikhin, L. V. Zh. Org. Khim. 1991, 27, 273.
Chen, W.; Parrish, D. A.; Deschamps, J. R.; Coop, A. Helv. Chim. Acta. 2005, 88, 822.
(a) Zhang, A.; Xiong, W.; Hilbert, J. E.; De Vita, E. K.; Bidlack, J. M.; Neumeyer, J. L. J. Med. Chem. 2004, 47, 1886. (b) Peng, X.; Knapp, B. I.; Bidlack, J. M.; Neumeyer, J. L. Bioorg. Med. Chem. 2007, 15, 4106.
Girán, L.; Gyulai, Z.; Antus, S.; Berényi, S.; Sipos, A. Monatsh. Chem. 2010, 141, 1135.
(a) Bauman, В. Т.; Shults, E. E.; Shakirov, М. М.; Tolstikov, G. А. Russ. J. Org. Chem. 2012, 48, 1473. [Zh. Org. Khim. 2012, 48, 1489.] (b) Bauman, V. T.; Shul'ts, E. E.; Kononchuk, V. V.; Bagryanskaya, I. Yu.; Shakirov, M. М.; Tolstikov, G. А. Russ. J. Org. Chem. 2013, 49, 1502. [Zh. Org. Khim. 2013, 49, 1522.]
Tolstikov, G. А.; Shultz, E. E.; Spirikhin, L. V. Tetrahedron. 1986, 42, 591.
(a) Raic-Malic, S.; Mescic, A. Curr. Med. Chem. 2015, 22, 1462. (b) Mukusheva, G. K.; Lipeeva, A. V.; Zanimkhanova, P. Z.; Shults, E. E.; Gatilov, Yu. V.; Shakirov, M. M.; Adekenov, S. M. Chem. Heterocycl. Compd. 2015, 51, 146. [Khim. Geterotsikl. Soedin. 2015, 51, 146.] (c) Lipeeva, A. V.; Pokrovsky, M. A.; Baev, D. S.; Shakirov, M. M.; Bagryanskaya, I. Y.; Tolstikova, T. G.; Pokrovsky, A. G.; Shults, E. E. Eur. J. Med. Chem. 2015, 100, 119. (d) Nguyen, B. C. Q.; Takahashi, H.; Uto, Y.; Shahinozzaman, M. d.; Tawata, S.; Maruta, H. Eur. J. Med. Chem. 2017, 126, 270. (e) Kasprzak, K.; Skiera, I.; Piasecka, M.; Paryzek, Z. Chem. Rev. 2016, 116, 5689.
(а) Bennett, I. S.; Brooks, G.; Broom, N. J. P.; Calvert, S. H.; Coleman, K.; François, I. J. Antibiot. 1991, 44, 969. (b) Soltis, M. J.; Yeh, H. J.; Cole, K. A.; Whittaker, N.; Wersto, R. P.; Kohn, E. C. Drug Metab. Dispos. 1996, 24, 799.
(a) Shafi, S.; Alam, M. M.; Mulakayala, N.; Mulakayala, C.; Vanaja, G.; Kalle, A. M.; Pallu, R.; Alam, M. S. Eur. J. Med. Chem. 2012, 49, 324. (b) Haider, S.; Alam, M. S.; Hamid, H.; Shafi, S.; Nargotra, A.; Mahajan, P.; Nazreen, S.; Kalle, A. M.; Kharbanda, C.; Ali, Y.; Alam, A.; Panda, A. K. Eur. J. Med. Chem. 2013, 70, 579.
(a) Moorhouse, A. D.; Santos, A. M.; Gunaratnam, M.; Moore, M.; Neidle, S.; Moses, J. E. J. Am. Chem. Soc. 2006, 128, 15972. (b) Thirumurugan, P.; Matosiuk, D.; Jozwiak, K. Chem. Rev. 2013, 113, 4905.
(a) Rosario-Amorin, D.; Gaboyard, M.; Clerac, R.; Vellutini, L.; Nlate, S.; Heuze, K. Chem.–Eur. J. 2012, 18, 3305. (b) Gimeno, N.; Martin-Rapun, R.; Rodriguez-Conde, S.; Serrano, J. L.; Folcia, C. L.; Pericas, M. A.; Ros, M. B. J. Mater. Chem. 2012, 22, 16791.
(a) Burley, G. A.; Gierlich, J.; Mofid, M. R.; Nir, H.; Tal, S.; Eichen, Y.; Carell, T. J. Am. Chem. Soc. 2006, 128, 1398. (b) Haridas, V.; Sahu, S.; Venugopalan, P. Tetrahedron 2011, 67, 727.
Creary, X., Anderson, A.; Brophy, C.; Crowell, F.; Funk, Z. J. Org. Chem. 2012, 77, 8756.
Yavari, I.; Seyfi, S. Synlett 2012, 23, 1209.
(a) Voskressensky, L. G.; Borisova, T. N.; Chervyakova, T. M.; Matveeva, M. D.; Galaktionova, D. V.; Tolkunov, S. V.; Tolkunova, V. S.; Eresko, A. B.; Varlamov, A. V. Chem. Heterocycl. Compd. 2014, 50, 1338. [Khim. Geterotsikl. Soedin. 2014, 1338.] (b) Penning, M.; Aeissen, E.; Christoffers, J. Synthesis 2015, 47, 1007.
Li, J.; Li, Ji.; Xu, Y.; Wang, Y.; Zhang, L.; Ding, L.; Xuan, Y.; Pang, T.; Lin, H. Nat. Prod. Res. 2016, 30, 800.
(a) Tolstikov, G. A.; Shults, E. E.; Mukhametyanova, T. Sh; Tolstikova, T. G.; Popov, V. G.; Ismagilova, A. F.; Lazareva, D. N.; Zarudii, F. S. Khim.-Farm. Zh. 1993 27, 20. (b) Tolstikov, G. A.; Shults, E. E.; Mukhametyanova, T. Sh.; Tolstikova, T. G.; Popov, V. G.; Zarudij, F. A.; Davydova, V. A.; Lazareva, D. N. RU Patent 2024526.
(а) Sandulenko, I. V.; Kovaleva, E. S.; Peregudov, A. S.; Kalinin V. N.; Moiseev, S. K. ChemistrySelect 2016, 1, 1004. (b) Sandulenko, I. V.; Semenova, D. V.; Zelentsova, M. V.; Moiseev, S. K.; Koldobskii, A. B.; Peregudov, A. S.; Bushmarinov, I. S.; Kalinin, V. N. J. Fluorine Chem. 2016, 189, 7.
Krisin, A. P. Zh. Org. Khim. 1986, 22, 1200.
Adams, R. E.; Press, J. B.; Deegan, E. G. Synth. Commun. 1991, 21, 675.
de Lourdes Ferreira, M. G.; Pinheiro, L. C. S.; Santos-Filho, O. A.; Peçanha, M. D. S.; Sacramento, C. Q.; Machado, V.; Ferreira, V. F.; Souza, T. M. L.; Boechat, N. Med. Chem. Res. 2014, 23, 1501.
Lipeeva, A. V.; Shul'ts, E. E. Russ. J. Org. Chem. 2015, 51, 957. [Zh. Org. Khim. 2015, 51, 977.]
Acknowledgement
This work was performed with financial support from the Russian Foundation for Basic Research (project No. 16-53-44027) and the Russian Science Foundation (project No. 14-13-00822).
The authors are grateful to the Chemical Service Center for Collective Use of the Siberian Branch of the Russian Academy of Sciences for spectral studies and analytical support.
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2017, 53(8), 913–919
Rights and permissions
About this article
Cite this article
Bauman, V.T., Ganbaatar, J. & Shults, E.E. Synthetic transformations of isoquinoline alkaloids: 1-(N-alkyl-1,2,3-triazol-4-yl)-6,18-endo-ethenodihydrothebainehydroquinones and triazolylnaphthohydroquinone-containing benzofuroazocines from thebaine. Chem Heterocycl Comp 53, 913–919 (2017). https://doi.org/10.1007/s10593-017-2145-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-017-2145-2