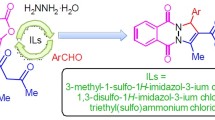
This microreview is devoted to the specific recent synthetic approaches toward 1,4-dihydropyridines which represent significant group of N-containing heterocycles due to the wide applications of their pharmacological activities. Mainly 1,4-dihydropyridines are synthesized in Hantzsch reaction and its modifications. Nowadays a number of strategies and various catalysts or catalytic systems have been developed to improve the yields of 1,4-dihydropyridines and to reduce chemical impact on environment. Here the contemporary synthetic aspects from last five years such as use of ionic liquids, metal oxide nanoparticles and some reports about enzymatic approaches are discussed.

Similar content being viewed by others
References
Edraki, N.; Mehdipour, A. R.; Khoshneviszadeh, M.; Miri, R. Drug Discovery Today 2009, 14, 1058.
Evans, B. E.; Rittle, K. E.; Bock, M. G.; Dipardo, R. M.; Freidinger, R. M.; Whitter, W. L.; Lundell, G. F.; Verber, D. F.; Anderson, P. S.; Chang, R. S. L.; Lotti, V. J.; Cerino, D. H.; Chen, T. B.; Kling, P. J.; Kunkel, K. A.; Springer, J. P.; Hirshfield, J. J. Med. Chem. 1998, 31, 2235.
Muller, G. Drug Discovery Today 2003, 8, 681.
Hantzsch, A. Chem. Ber. 1881, 14, 1637.
Wan, J.-P.; Liu, Y. RSC Adv. 2012, 2, 9763.
Carosati, E.; Ioan, P.; Micucci, M.; Broccatelli, F.; Cruciani, G.; Zhorov, B. S.; Chiarini, A.; Budriesi, R. Curr. Med. Chem. 2012, 19, 4306.
Broccatelli, F.; Zhorov, B. S.; Chiarini, A.; Budriesi, R. Curr. Med. Chem. 2011, 18, 4901.
Khedkar, S. A.; Auti P. B. Mini-Rev. Med. Chem. 2014, 14, 282.
Hyvönen, Z.; Plotniece, A.; Reine, I.; Chekavichus, B.; Duburs, G.; Urtti, A. Biochim. Biophys. Acta 2000, 1509, 451.
Pajuste, K.; Hyvönen, Z.; Petrichenko, O.; Kaldre, D.; Rucins, M.; Cekavicus, B.; Ose, V.; Skrivele, B.; Gosteva, M.; Morin-Picardat, E.; Plotniece, M.; Sobolev, A.; Duburs, G.; Ruponen, M.; Plotniece A. New J. Chem. 2013, 37, 3062.
Radadiya, A.; Khedkar, V.; Bavishi, A.; Vala, H.; Thakrar, S.; Bhavsar, D.; Shah, A.; Coutinho, E. Eur. J. Med Chem. 2014, 74, 375.
Cindric, M.; Cipak, A.; Serly, J.; Plotniece, A.; Jaganjac, M.; Mrakovcic, L.; Lovakovic, T.; Dedic, A.; Soldo, I.; Duburs, G.; Zarkovic, N.; Molnar, J. Anticancer Res. 2010, 30, 4063.
Jansone, B.; Kadish, I.; van Groen, T.; Beitnere, U.; Moore, D. R.; Plotniece, A.; Pajuste, K.; Klusa, V. PLoS ONE 2015, 10, e0127686.
Klusa, V. Pharmacol. Res. DOI: 10.1016/j.phrs.2016.05.017
Zicmanis, A.; Hinica, A.; Pavlovica S.; Klavins, M.; Latv. Kim. Z. 2009, 3, 235.
Priede, E.; Zicmanis, A. Helv. Chim. Acta 2015, 98, 1095.
Sharma, P.; Gupta, M. Green Chem. 2015, 17, 1100.
He, J.-Y.; Jia, H.-Z.; Yao, Q.-G.; Liu S.-J.; Yue H.-K.; Yu H.-W.; Hu R.-S. Ultrason. Sonochem. 2015, 22, 144.
Rad-Moghadam, K.; Youseftabar-Miri, L. J. Fluor. Chem. 2012, 135, 213.
Pajuste, K.; Plotniece, A.; Kore, K.; Intenberga, L.; Cekavicus, B.; Kaldre, D.; Duburs, G.; Sobolev, A. Cent. Eur. J. Chem. 2011, 9, 143.
Kumar, R.; Andhare, N. H.; Shard, A.; Richaa, Sinha, A. K. RSC Adv. 2014, 4, 19111.
Min, Y.; Akbulut, M.; Kristiansen, K.; Golan, Y.; Israelachvili, J. Nat. Mater. 2008, 7, 527.
Nasr-Esfahani, M.; Hoseini, S. J.; Montazerozohori, M.; Mehrabi, R.; Nasrabadi, H. J. Mol. Catal. A: Chem. 2014, 382, 99.
Amirheidari, B.; Seifi, M.; Abaszadeh, M. Res. Chem. Intermed. 2016, 42, 3413.
Dam, B.; Nandi, S.; Pal, A. K. Tetrahedron Lett. 2014, 55, 5236.
Murugan, R.; Ramamoorthy, K.; Sundarrajan, S.; Ramakrishna, S. Tetrahedron 2012, 68, 7196.
Zarnegar, Z.; Safari, J.; Kafroudi, Z. M. New J. Chem. 2015, 39, 1445.
Wang, J.-L.; Liu, B.-K.; Yin, C.; Wu, Q.; Lin, X.-F. Tetrahedron 2011, 67, 2689.
Tamaddon F.; Ghazi, S. Catal. Commun. 2015, 72, 63.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2016, 52(8), 538–540
Rights and permissions
About this article
Cite this article
Pajuste, K., Plotniece, A. Ionic liquids, metal oxide nanoparticles, and enzymes in synthesis of 1,4-dihydropyridines (microreview). Chem Heterocycl Comp 52, 538–540 (2016). https://doi.org/10.1007/s10593-016-1926-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-016-1926-3