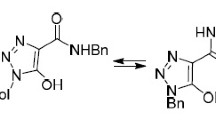
Reaction of the hydrazides of aromatic and pyridine-3(4)-carboxylic acids with 4-ethoxy-1,1,1-tri-fluorobut-3-en-2-one leads to the formation of derivatives having a 5-hydroxy-2-pyrazoline structure. In solution in DMSO-d6 they are capable of exhibiting ring-chain equilibrium involving the 5-hydroxy-2-pyrazoline and enehydrazine tautomers. The latter form is represented by a mixture of Z- and E-isomers with a significant preponderance of the latter. The product from the reaction of 4-aminobenzohydrazide with 4-ethoxy-1,1,1-trifluorobut-3-en-2-one in a ratio of 1:2 has a structure containing 5-hydroxy-2-pyrazoline and enamine fragments.
Similar content being viewed by others
References
S. I. Yakimovich, I. V. Zerova, and K. N. Zelenin, Zh. Org. Khim., 33, 418 (1997).
V. V. Pakal'nis, I. V. Zerova, A. I. Plyasunova, and S. I. Yakimovich, Vestn. SPbGU, Ser. 4, No. 4, 79 (2009).
S. I. Yakimovich, I. V. Zerova, and V. V. Pakal'nis, in Modern Problems of Organic Chemistry, Issue 15, St. Petersburg University Press, St. Petersburg (2011), р. 205.
I. I. Gerus, M. G. Gorbunova, S. I. Vdovenko, Yu. L. Yagupol'skii, and V. P. Kukhar', Zh. Org. Khim., 26, 1877 (1990).
S. I. Yakimovich and K. N. Zelenin, Zh. Obshch. Khim., 65, 705 (1995).
M. Hojo, R. Masuda, Y. Kokuryo, H. Shioda, and S. Matsuo, Chem. Lett., 499 (1976).
Author information
Authors and Affiliations
Corresponding author
Additional information
*Deceased.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1201–1206, August, 2014.
Rights and permissions
About this article
Cite this article
Pakal’nis, V.V., Zerov, A.V., Yakimovich, S.I. et al. Ring-Chain Tautomerism of 3-Unsubstituted Trifluoromethyl-Containing n-Acyl-5-Hydroxy-2-Pyrazolines. Chem Heterocycl Comp 50, 1107–1112 (2014). https://doi.org/10.1007/s10593-014-1570-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-014-1570-8