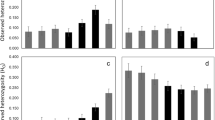
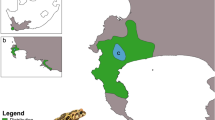
Abstract
Active management is essential to the survival of many threatened species globally. Captive breeding programmes can play an important role in facilitating the supplementation, translocation and reintroduction of wild populations. However, understanding the genetic dynamics within and among wild and captive populations is crucial to the planning and implementation of ex situ management, as adaptive potential is, in part, driven by genetic diversity. Here, we use 14 microsatellite loci and mitochondrial Control Region sequence to examine the population genetics of both wild populations and captive colonies of the endangered warru (the MacDonnell Ranges race of the black-footed rock-wallaby Petrogale lateralis) in central Australia, to understand how historical evolutionary processes have shaped current diversity and ensure effective ex situ management. Whilst microsatellite data reveal significant contemporary differentiation amongst remnant warru populations, evidence of contemporary dispersal and relatively weak isolation by distance, as well as a lack of phylogeographic structure suggests historical connectivity. Genetic diversity within current captive populations is lower than in the wild source populations. Based on our genetic data and ecological observations, we predict outbreeding depression is unlikely and hence make the recommendation that captive populations be managed as one genetic group. This will increase genetic diversity within the captive population and as a result increase the adaptive potential of reintroduced populations. We also identify a new site in the Musgrave Ranges which contains unique alleles but also connectivity with a population 6 km away. This novel genetic diversity could be used as a future source for supplementation.


Similar content being viewed by others
References
Armstrong DP, Wittmer HU (2011) Incorporating Allee effects into reintroduction strategies. Ecol Res 26(4):687–695
Arrendal J, Walker C, Sundqvist A, Hellborg L, Vilà C (2004) Genetic evaluation of an otter translocation program. Conserv Genet 5(1):79–88
Biebach I, Keller LF (2010) Inbreeding in reintroduced populations: the effects of early reintroduction history and contemporary processes. Conserv Genet 11(2):527–538
Browning TL, Taggart DA, Rummery C, Close RL, Eldridge MDB (2001) Multifaceted genetic analysis of the “Critically Endangered” brush-tailed rock-wallaby Petrogale penicillata in Victoria, Australia: implications for management. Conserv Genet 2(2):145–156
Byrne M, Yeates D, Joseph L, Kearney M, Bowler J, Williams M, Cooper S, Donnellan S, Keogh J, Leys R (2008) Birth of a biome: insights into the assembly and maintenance of the Australian arid zone biota. Mol Ecol 17(20):4398–4417
Cheyne S (2006) Wildlife reintroduction: considerations of habitat quality at the release site. BMC Ecol 6(5)
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9(10):1657–1659
Earl DA (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conser Genet Resour 4(2):359–361
Eldridge M, Pearson D (2008) Black-footed rock-wallaby Petrogale lateralis Gould 1842. In: van Dyck S., Strahan R (ed) The mammals of Australia, 3rd edn. pp 376–380
Eldridge MDB, Kinnear JE, Onus ML (2001) Source population of dispersing rock-wallabies (Petrogale lateralis) identified by assignment tests on multilocus genotypic data. Mol Ecol 10(12):2867–2876
Eldridge MDB, Piggott MP, Hazlitt SL (2010) Population genetic studies of the Macropodoidea: a review. In: Coulson G, Eldridge M.D.B. (eds) Macropods: the biology of kangaroos, wallabies and rat kangaroos. CSIRO Publishing, Collingwood
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14(8):2611–2620
Excoffier L, Lischer HE (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10(3):564–567
Frankham R (2008) Genetic adaptation to captivity in species conservation programs. Mol Ecol 17(1):325–333
Frankham R, Ballou JD, Briscoe DA (2002) Introduction to conservation genetics. Cambridge University Press, Cambridge
Frankham R, Ballou JD, Eldridge MDB, Lacy RC, Ralls K, Dudash MR, Fenster CB (2011) Predicting the probability of outbreeding depression. Conserv Biol 25(3):465–475
Frankham R, Ballou JD, Ralls K, Eldridge M, Dubash MR, Fenster CB, Lacy RC, Sunnucks P (2017) Genetic management of fragmented animal and plant populations. Oxford University Press, Oxford
Fumagalli L, Pope LC, Taberlet P, Moritz C (1997) Versatile primers for the amplification of the mitochondrial DNA control region in marsupials. Mol Ecol 6:1199–1201
Gaggiotti OE (2003) Genetic threats to population persistence. Ann Zool Fenn 40:155–168
Goudet J (1995) FSTAT (version 1.2): a computer program to calculate F-statistics. J Hered 86(6):485–486
Hazlitt S, Eldridge MDB, Goldizen A (2004) Fine-scale spatial genetic correlation analyses reveal strong female phiolpatry within a brush-tailed rock wallaby colony in southeast Queensland. Mol Ecol 13:3621–3632
Hazlitt S, Sigg D, Eldridge MDB, Goldizen A (2006a) Restricted mating dispersal and strong breeding group structure in a mid-sized marsupial mammal (Petrogale penicillata). Mol Ecol 15:2997–3007
Hazlitt SL, Goldizen AW, Eldridge MDB (2006b) Significant patterns of population genetic structure and limited gene flow in a threatened macropodid marsupial despite continuous habitat in southeast Queensland, Australia. Conserv Genet 7(5):675–689
Hazlitt SL, Goldizen AW, Nicholls JA, Eldridge MD (2014) Three divergent lineages within an Australian marsupial (Petrogale penicillata) suggest multiple major refugia for mesic taxa in southeast Australia. Ecol Evol 4(7):1102–1116
IBM (2013). IBM SPSS statistics for windows, version 22.0, IBM Corp., Armonk
IUCN/SSC (2013) Guidelines for reintroductions and other conservation translocations. Version 1.0. IUCN Species Survival Commission, Gland
Jamieson IG (2011) Founder effects, inbreeding, and loss of genetic diversity in four avian reintroduction programs. Conserv Biol 25(1):115–123
Jamieson IG, Lacy RC (2012) Managing genetic issues in reintroduction biology. In: Ewen JG, Armstrong DP, Parker KA, Seddon PJ (eds) Reintroduction biology: integrating science and management: Blackwell Publishing Ltd, Hoboken
Keller LF, Biebach I, Ewing SR, Hoeck PEA (2012) The genetics of reintroductions: inbreeding and genetic drift. In: Ewen JG, Armstrong DP, Parker KA, Seddon PJ (eds) Reintroduction Biology: integrating science and management, Blackwell Publishing Ltd, Hoboken
Kopelman NM, Mayzel J, Jakobsson M, Rosenberg NA, Mayrose I (2015) Clumpak: a program for identifying clustering modes and packaging population structure inferences across K. Mol Ecol Resour 15(5):1179–1191
Larson S, Jameson R, Bodkin J, Staedler M, Bentzen P (2002) Microsatellite DNA and mitochondrial DNA variation in remnant and translocated sea otter (Enhydra lutris) populations. J Mammal 83(3):893–906
Lennon MJ, Taggart DA, Temple-Smith PD, Eldridge MDB (2011) The impact of isolation and bottlenecks on genetic diversity in the Pearson Island population of the black-footed rock-wallaby (Petrogale lateralis pearsoni; Marsupialia: Macropodidae). Aust Mammal 33(2):152–161
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25(11):1451–1452
Maran T, Põdra M, Põlma M, Macdonald DW (2009) The survival of captive-born animals in restoration programmes—case study of the endangered European mink Mustela lutreola. Biol Conserv 142(8):1685–1692
Miller EJ, Eldridge MD, Morris KD, Zenger KR, Herbert CA (2011) Genetic consequences of isolation: island tammar wallaby (Macropus eugenii) populations and the conservation of threatened species. Conserv Genet 12(6):1619–1631
Moseby KE, Read JL, Paton DC, Copley P, Hill BM, Crisp HA (2011) Predation determines the outcome of 10 reintroduction attempts in arid South Australia. Biol Conserv 144(12):2863–2872
Muhic J, Abbott E, Ward MJ (2012) The warru (Petrogale lateralis MacDonnell Ranges Race) reintroduction project on the Anangu Pitjantjatjara Yankunytjatjara Lands, South Australia. Ecol Manag Restor 13(1):89–92
Nei M, Maruyama T, Chakraborty R (1975) The bottleneck effect and genetic variability in populations. Evolution 29(1):1–10
Paetkau D, Slade R, Burden M, Estoup A (2004) Genetic assignment methods for the direct, real-time estimation of migration rate: a simulation-based exploration of accuracy and power. Mol Ecol 13(1):55–65
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6(1):288–295
Piggott MP, Banks SC, Taylor AC (2006) Population structure of brush-tailed rock-wallaby (Petrogale penicillata) colonies inferred from analysis of faecal DNA. Mol Ecol 15:93–105
Piry S, Alapetite A, Cornuet J-M, Paetkau D, Baudouin L, Estoup A (2004) GENECLASS2: a software for genetic assignment and first-generation migrant detection. J Hered 95(6):536–539
Pope LC, Sharp A, Moritz C (1996) Population structure of the yellow-footed rock-wallaby Petrogale xanthopus (Gray, 1854) inferred from mtDNA sequences and microsatellite loci. Mol Ecol 5(5):629–640
Pearson DJ (2012) Recovery plan for five species of rock wallabies: Black-footed rock wallaby (Petrogale lateralis), Rothschild rock wallaby (Petrogale rothschildi), Short-eared rock wallaby (Petrogale brachyotis), Monjon (Petrogale burbidgei) and Nabarlek (Petrogale concinna) 2012–2022. Department of Environment and Conservation, Perth
Potter S, Eldridge MDB, Taggart DA, Cooper SJB (2012a) Multiple biogeographical barriers identified across the monsoon tropics of northern Australia: phylogeographic analysis of the brachyotis group of rock-wallabies. Mol Ecol 21(9):2254–2269
Potter S, Eldridge MDB, Cooper SJB, Paplinska JZ, Taggart DA (2012b) Habitat connectivity, more than species’ biology, influences genetic differentiation in a habitat specialist, the short-eared rock-wallaby (Petrogale brachyotis). Conserv Genet 13(4):937–952
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155(2):945–959
Rannala B, Mountain JL (1997) Detecting immigration by using multilocus genotypes. Proc Natl Acad Sci USA 94(17):9197–9201
Raymond M, Rousset F (1995) GENEPOP (version 1.2): population genetics software for exact tests and ecumenicism. J Hered 86:248–249
Read J, Ward MJ (2011a) Warru recovery plan: Recovery of Petrogale lateralis MacDonnell Ranges race in South Australia, 2010–2020. Department of Environment and Natural Resources, Adelaide
Read JL, Ward MJ (2011b) Bringing back warru: initiation and implementation of the South Australian Warru Recovery Plan. Aust Mammal 33(2):214–220
Reed DH, Frankham R (2003) Correlation between fitness and genetic diversity. Conserv Biol 17(1):230–237
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43(1):223–225
Robert A (2009) Captive breeding genetics and reintroduction success. Biol Conserv 142(12):2915–2922
Rozas J, Sánchez-DelBarrio JC, Messeguer X, Rozas R (2003) DnaSP, DNA polymorphism analyses by the coalescent and other methods. Bioinformatics 19(18):2496–2497
Ruykys L (2011) Ecology of warru (Petrogale lateralis MacDonnell Ranges race) in the Anangu Pitjantjatjara Yankunytjatjara Lands, South Australia. PhD University of Adelaide
Ruykys L, Lancaster ML (2015) Population structure and genetic diversity of the black-footed rock-wallaby (Petrogale lateralis MacDonnell Ranges race). Aust J Zool 63(2):91–100
Sharp A (1997) Insights into dispersal patterns of yellow-footed rock-wallabies, Petrogale xanthopus. Aust Mammal 19:229–238
Sigg DP (2006) Reduced genetic diversity and significant genetic differentiation after translocation: comparison of the remnant and translocated populations of bridled nailtail wallabies (Onychogalea fraenata). Conserv Genet 7(4):577–589
Stockwell CA, Mulvey M, Vinyard GL (1996) Translocations and the preservation of allelic diversity. Conserv Biol 10(4):1133–1141
Sunnucks P, Hales DF (1996) Numerous transposed sequences of mitochondrial cytochrome oxidase I-II in aphids of the genus Sitobion (Hemiptera: Aphididae). Mol Biol Evol 13(3):510–524
Taggart DA, Schultz D, White C, Whitehead P, Underwood G, Phillips K (2005) Cross-fostering, growth and reproductive studies in the brush-tailed rock-wallaby, Petrogale penicillata, (Marsupalia: Macropodidae): efforts to accelerate breeding in a threatened marsupial species. Aust J Zool 53:313–323
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Taylor A, Cooper D (1998) A set of tammar wallaby (Macropus eugenii) microsatellites tested for genetic linkage. Mol Ecol 7:925–926
Telfer WR, Eldridge MDB (2010) High levels of mitochondrial DNA divergence within short-eared rock-wallaby (Petrogale brachyotis) populations in northern Australia. Aust J Zool 58(2):104–112
Ward MJ, Urban R, Read JL, Dent A, Partridge T, Clarke A, van Weenen J (2011) Status of warru (Petrogale lateralis MacDonnell Ranges race) in the Anangu Pitjantjatjara Yankunytjatjara Lands of South Australia. 1. Distribution and decline. Aust Mammal 33(2):135–141
Weeks AR, Sgro CM, Young AG, Frankham R, Mitchell NJ, Miller KA, Byrne M, Coates DJ, Eldridge MDB, Sunnucks P, Breed MF, James EA, Hoffmann AA (2011) Assessing the benefits and risks of translocations in changing environments: a genetic perspective. Evol Appl 4(6):709–725
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 1358–1370
West R, Read JL, Ward MJ, Foster WK, Taggart DA (2016) Monitoring for adaptive management in a trial reintroduction of the black-footed rock-wallaby Petrogale lateralis. Oryx 51(3):1–10
Wright S (1951) The genetical structure of populations. Ann Eugen 15:323–354
Zenger KR, Cooper DW (2001) A set of highly polymorphic microsatellite markers developed for the eastern grey kangaroo (Macropus giganteus). Mol Ecol Notes 1(1–2):98–100
Acknowledgements
Approvals to conduct the project were granted by the Anangu Pitjantjatjara Yankunytjatjara Executive Board, South Australia Department of Environment and Natural Resources Wildlife Ethics Committee (15/2007) and University of Adelaide Animal Ethics Committee (S-2011-9). Funding for the study was provided by the Warru Recovery Team, Norman Wettenhall Foundation, Margaret Middleton Fund, Sir Mark Mitchell Research Foundation, Nature Foundation SA (Edwards Majory Scholarship) and Adelaide Off Road. Fieldwork was conducted by various members of the Warru Recovery Team and a fantastic team of volunteers. We thank Anangu tjuta for supporting the project.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
West, R., Potter, S., Taggart, D. et al. Looking back to go forward: genetics informs future management of captive and reintroduced populations of the black-footed rock-wallaby Petrogale lateralis . Conserv Genet 19, 235–247 (2018). https://doi.org/10.1007/s10592-017-1030-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-017-1030-y