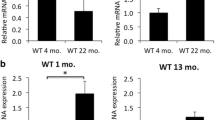
Abstract
Aging is a risk factor for multiple retinal degeneration diseases. Entraining brain gamma oscillations with gamma-flicker light (γFL) has been confirmed to coordinate pathological changes in several Alzheimer’s disease mouse models and aged mice. However, the direct effect of γFL on retinal aging remains unknown. We assessed retinal senescence-associated beta-galactosidase (β-gal) and autofluorescence in 20-month-old mice and found reduced β-gal-positive cells in the inner retina and diminished lipofuscin accumulation around retinal vessels after 6 days of γFL. In immunofluorescence, γFL was further demonstrated to ameliorate aging-related retinal changes, including a decline in microtubule-associated protein 1 light chain 3 beta expression, an increase in complement C3 activity, and an imbalance between the anti-oxidant factor catalase and pro-oxidant factor carboxymethyl lysine. Moreover, we found that γFL can increase the expression of activating transcription factor 4 (ATF4) in the inner retina, while revealing a decrease of ATF4 expression in the inner retina and positive expression in the outer segment of photoreceptor and RPE layer for aged mice. Western blotting was then used to confirm the immunofluorescence results. After mRNA sequencing (NCBI Sequence Read Archive database: PRJNA748184), we found several main mechanistic clues, including mitochondrial function and chaperone-mediated protein folding. Furthermore, we extended γFL to aged Apoe−/− mice and showed that 1-m γFL treatment even improved the structures of retinal-pigment-epithelium basal infolding and Bruch’s membrane. Overall, γFL can orchestrate various pathological characteristics of retinal aging in mice and might be a noninvasive, convenient, and tissue-specific therapeutic strategy for retinal aging.








Similar content being viewed by others
Data Availability
Raw data of RNA sequencing were deposited in NCBI Sequence Read Archive database (PRJNA748184). All other data generated or used during the study appear in the submitted article.
References
Allingham MJ, Nie Q, Lad EM, Izatt DJ, Mettu PS, Cousins SW, Farsiu S (2016) Semiautomatic segmentation of rim area focal hyperautofluorescence predicts progression of geographic atrophy due to dry age-related macular degeneration. Invest Ophthalmol vis Sci 57:2283–2289. https://doi.org/10.1167/iovs.15-19008
Anderson DH, Radeke MJ, Gallo NB et al (2010) The pivotal role of the complement system in aging and age-related macular degeneration: hypothesis re-visited. Prog Retin Eye Res 29:95–112. https://doi.org/10.1016/j.preteyeres.2009.11.003
Beswick EJ, Reyes VE (2009) CD74 in antigen presentation, inflammation, and cancers of the gastrointestinal tract. World J Gastroenterol 15:2855–2861. https://doi.org/10.3748/wjg.15.2855
Boya P, Esteban-Martinez L, Serrano-Puebla A, Gomez-Sintes R, Villarejo-Zori B (2016) Autophagy in the eye: development, degeneration, and aging. Prog Retin Eye Res 55:206–245. https://doi.org/10.1016/j.preteyeres.2016.08.001
Campisi J, Kapahi P, Lithgow GJ, Melov S, Newman JC, Verdin E (2019) From discoveries in ageing research to therapeutics for healthy ageing. Nature 571:183–192. https://doi.org/10.1038/s41586-019-1365-2
Chen M, Muckersie E, Forrester JV, Xu H (2010) Immune activation in retinal aging: a gene expression study. Invest Ophthalmol vis Sci 51:5888–5896. https://doi.org/10.1167/iovs.09-5103
Chen M, Luo C, Zhao J, Devarajan G, Xu H (2019) Immune regulation in the aging retina. Prog Retin Eye Res 69:159–172. https://doi.org/10.1016/j.preteyeres.2018.10.003
Copenhagen DR (1991) Synaptic transmission in the retina. Curr Opin Neurobiol 1:258–262. https://doi.org/10.1016/0959-4388(91)90087-n
Debacq-Chainiaux F, Erusalimsky JD, Campisi J, Toussaint O (2009) Protocols to detect senescence-associated beta-galactosidase (SA-betagal) activity, a biomarker of senescent cells in culture and in vivo. Nat Protoc 4:1798–1806. https://doi.org/10.1038/nprot.2009.191
Delaunay K, Sellam A, Dinet V, Moulin A, Zhao M, Gelize E, Canonica J, Naud MC, Crisanti-Lassiaz P, Behar-Cohen F (2021) Meteorin is a novel therapeutic target for wet age-related macular degeneration. J Clin Med. https://doi.org/10.3390/jcm10132973
Dieguez HH, Romeo HE, Alaimo A, Gonzalez Fleitas MF, Aranda ML, Rosenstein RE, Dorfman D (2019) Oxidative stress damage circumscribed to the central temporal retinal pigment epithelium in early experimental non-exudative age-related macular degeneration. Free Radic Biol Med 131:72–80. https://doi.org/10.1016/j.freeradbiomed.2018.11.035
Dithmar S, Curcio CA, Le NA, Brown S, Grossniklaus HE (2000) Ultrastructural changes in Bruch’s membrane of apolipoprotein E-deficient mice. Invest Ophthalmol vis Sci 41:2035–2042
Dong XX, Wang Y, Qin ZH (2009) Molecular mechanisms of excitotoxicity and their relevance to pathogenesis of neurodegenerative diseases. Acta Pharmacol Sin 30:379–387. https://doi.org/10.1038/aps.2009.24
Fernández-Robredo P, Sádaba LM, Salinas-Alamán A, Recalde S, Rodríguez JA, García-Layana A (2013) Effect of lutein and antioxidant supplementation on VEGF expression, MMP-2 activity, and ultrastructural alterations in apolipoprotein E-deficient mouse. Oxid Med Cell Longev 2013:213505. https://doi.org/10.1155/2013/213505
Garaschuk O, Semchyshyn HM, Lushchak VI (2018) Healthy brain aging: interplay between reactive species, inflammation and energy supply. Ageing Res Rev 43:26–45. https://doi.org/10.1016/j.arr.2018.02.003
Goroshinskaya IA, Kit OI, Zuderman N, Ushakova ND, Lysenko IB, Nemashkalova LA, Nikolaeva NV, Kapuza EA, Shatokhina ON (2018) Oxidative processes in the blood during chemotherapy and lethality of patients with multiple myeloma. J Clin Oncol 36:e20027-e. https://doi.org/10.1200/JCO.2018.36.15_suppl.e20027
Gupta V, Gupta VB, Chitranshi N et al (2016) One protein, multiple pathologies: multifaceted involvement of amyloid beta in neurodegenerative disorders of the brain and retina. Cell Mol Life Sci 73:4279–4297. https://doi.org/10.1007/s00018-016-2295-x
Hamilton JD, Suarez-Farinas M, Dhingra N et al (2014) Dupilumab improves the molecular signature in skin of patients with moderate-to-severe atopic dermatitis. J Allergy Clin Immunol 134:1293–1300. https://doi.org/10.1016/j.jaci.2014.10.013
Hartl FU, Bracher A, Hayer-Hartl M (2011) Molecular chaperones in protein folding and proteostasis. Nature 475:324–332. https://doi.org/10.1038/nature10317
Hayes MJ, Burgoyne T, Wavre-Shapton ST, Tolmachova T, Seabra MC, Futter CE (2019) Remodeling of the basal labyrinth of retinal pigment epithelial cells with osmotic challenge, age, and disease. Invest Ophthalmol vis Sci 60:2515–2524. https://doi.org/10.1167/iovs.19-26784
Hetz C, Zhang K, Kaufman RJ (2020) Mechanisms, regulation and functions of the unfolded protein response. Nat Rev Mol Cell Biol 21:421–438. https://doi.org/10.1038/s41580-020-0250-z
Iaccarino HF, Singer AC, Martorell AJ et al (2016) Gamma frequency entrainment attenuates amyloid load and modifies microglia. Nature 540:230–235. https://doi.org/10.1038/nature20587
Kamiya T, Takeuchi K, Fukudome S, Hara H, Adachi T (2018) Copper chaperone antioxidant-1, Atox-1, is involved in the induction of SOD3 in THP-1 cells. Biometals 31:61–68. https://doi.org/10.1007/s10534-017-0067-1
Kaushik S, Cuervo AM (2018) The coming of age of chaperone-mediated autophagy. Nat Rev Mol Cell Biol 19:365–381. https://doi.org/10.1038/s41580-018-0001-6
Khan SS, Singer BD, Vaughan DE (2017) Molecular and physiological manifestations and measurement of aging in humans. Aging Cell 16:624–633. https://doi.org/10.1111/acel.12601
Kokkinopoulos I, Colman A, Hogg C, Heckenlively J, Jeffery G (2013) Age-related retinal inflammation is reduced by 670 nm light via increased mitochondrial membrane potential. Neurobiol Aging 34:602–609. https://doi.org/10.1016/j.neurobiolaging.2012.04.014
Lee KS, Lin S, Copland DA, Dick AD, Liu J (2021) Cellular senescence in the aging retina and developments of senotherapies for age-related macular degeneration. J Neuroinflamm 18:32. https://doi.org/10.1186/s12974-021-02088-0
Liberatore F, Bucci D, Mascio G et al (2017) Permissive role for mGlu1 metabotropic glutamate receptors in excitotoxic retinal degeneration. Neuroscience 363:142–149. https://doi.org/10.1016/j.neuroscience.2017.09.005
Lin MT, Beal MF (2006) Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443:787–795. https://doi.org/10.1038/nature05292
Lyamzaev KG, Tokarchuk AV, Panteleeva AA, Mulkidjanian AY, Skulachev VP, Chernyak BV (2018) Induction of autophagy by depolarization of mitochondria. Autophagy 14:921–924. https://doi.org/10.1080/15548627.2018.1436937
Mansour H, Chamberlain CG, Weible MW 2nd, Hughes S, Chu Y, Chan-Ling T (2008) Aging-related changes in astrocytes in the rat retina: imbalance between cell proliferation and cell death reduces astrocyte availability. Aging Cell 7:526–540. https://doi.org/10.1111/j.1474-9726.2008.00402.x
Martorell AJ, Paulson AL, Suk HJ et al (2019) Multi-sensory gamma stimulation ameliorates Alzheimer’s-associated pathology and improves cognition. Cell 177(256–71):e22. https://doi.org/10.1016/j.cell.2019.02.014
McKernan DP, Caplis C, Donovan M, O’Brien CJ, Cotter TG (2006) Age-dependent susceptibility of the retinal ganglion cell layer to cell death. Invest Ophthalmol vis Sci 47:807–814. https://doi.org/10.1167/iovs.05-0520
Michowski W, Ferretti R, Wisniewska MB et al (2010) Morgana/CHP-1 is a novel chaperone able to protect cells from stress. Biochim Biophys Acta 1803:1043–1049. https://doi.org/10.1016/j.bbamcr.2010.05.005
Mishra A (2017) Binaural blood flow control by astrocytes: listening to synapses and the vasculature. J Physiol 595:1885–1902. https://doi.org/10.1113/JP270979
Morgan JI, Hunter JJ, Merigan WH, Williams DR (2009) The reduction of retinal autofluorescence caused by light exposure. Invest Ophthalmol vis Sci 50:6015–6022. https://doi.org/10.1167/iovs.09-3643
Nakamura M, Yako T, Kuse Y, Inoue Y, Nishinaka A, Nakamura S, Shimazawa M, Hara H (2018) Exposure to excessive blue LED light damages retinal pigment epithelium and photoreceptors of pigmented mice. Exp Eye Res 177:1–11. https://doi.org/10.1016/j.exer.2018.07.022
Pfanner N, Warscheid B, Wiedemann N (2019) Mitochondrial proteins: from biogenesis to functional networks. Nat Rev Mol Cell Biol 20:267–284. https://doi.org/10.1038/s41580-018-0092-0
Ramos D, Carretero A, Navarro M, Mendes-Jorge L, Nacher V, Rodriguez-Baeza A, Ruberte J (2013) Mimicking microvascular alterations of human diabetic retinopathy: a challenge for the mouse models. Curr Med Chem 20:3200–3217. https://doi.org/10.2174/09298673113209990028
Rodriguez-Muela N, Koga H, Garcia-Ledo L, de la Villa P, de la Rosa EJ, Cuervo AM, Boya P (2013) Balance between autophagic pathways preserves retinal homeostasis. Aging Cell 12:478–488. https://doi.org/10.1111/acel.12072
Schaaf MB, Keulers TG, Vooijs MA, Rouschop KM (2016) LC3/GABARAP family proteins: autophagy-(un)related functions. Faseb J 30:3961–3978. https://doi.org/10.1096/fj.201600698R
Shao W, Zhang Y, Wang J, Lv C, Chen C (2016) BcMtg2 is required for multiple stress tolerance, vegetative development and virulence in Botrytis cinerea. Sci Rep 6:28673. https://doi.org/10.1038/srep28673
Shen J, Tower J (2019) Effects of light on aging and longevity. Ageing Res Rev 53:100913. https://doi.org/10.1016/j.arr.2019.100913
Subramanian A, Tamayo P, Mootha VK et al (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 102:15545–15550. https://doi.org/10.1073/pnas.0506580102
Taubitz T, Fang Y, Biesemeier A, Julien-Schraermeyer S, Schraermeyer U (2019) Age, lipofuscin and melanin oxidation affect fundus near-infrared autofluorescence. EBioMedicine 48:592–604. https://doi.org/10.1016/j.ebiom.2019.09.048
Terra LF, Wailemann RAM, Dos Santos AF et al (2019) Heat shock protein B1 is a key mediator of prolactin-induced beta-cell cytoprotection against oxidative stress. Free Radic Biol Med 134:394–405. https://doi.org/10.1016/j.freeradbiomed.2019.01.023
Upadhyay M, Milliner C, Bell BA, Bonilha VL (2020) Oxidative stress in the retina and retinal pigment epithelium (RPE): role of aging, and DJ-1. Redox Biol 37:101623. https://doi.org/10.1016/j.redox.2020.101623
Valenza M, Facchinetti R, Steardo L, Scuderi C (2019) Altered waste disposal system in aging and Alzheimer’s disease: focus on astrocytic aquaporin-4. Front Pharmacol 10:1656. https://doi.org/10.3389/fphar.2019.01656
Wei H, Xun Z, Granado H, Wu A, Handa JT (2016) An easy, rapid method to isolate RPE cell protein from the mouse eye. Exp Eye Res 145:450–455. https://doi.org/10.1016/j.exer.2015.09.015
Wu J, Wu D, Zhang L et al (2020) NK cells induce hepatic ER stress to promote insulin resistance in obesity through osteopontin production. J Leukoc Biol 107:589–596. https://doi.org/10.1002/JLB.3MA1119-173R
Xia W, Xie C, Jiang M, Hou M (2015) Improved survival of mesenchymal stem cells by macrophage migration inhibitory factor. Mol Cell Biochem 404:11–24. https://doi.org/10.1007/s11010-015-2361-y
Xu H, Chen M, Manivannan A, Lois N, Forrester JV (2008) Age-dependent accumulation of lipofuscin in perivascular and subretinal microglia in experimental mice. Aging Cell 7:58–68. https://doi.org/10.1111/j.1474-9726.2007.00351.x
Xu H, Chen M, Forrester JV (2009) Para-inflammation in the aging retina. Prog Retin Eye Res 28:348–368. https://doi.org/10.1016/j.preteyeres.2009.06.001
Acknowledgements
We thank Jimmy for optimizing the electric circuit.
Funding
This work was supported by (1) National Natural Science Foundation of China (No. 32061160469); (2) the Fundamental Research Funds for the Central Universities of Central South University (No. 2019zzts368); (3) Research Grant of Key Laboratory of Regenerative Medicine, Ministry of Education, Jinan University (No. ZSYX-M-2019-00010); and (4) The Hong Kong Polytechnic University Joint Supervision Scheme with the Chinese Mainland, Taiwan and Macao Universities (No. G-SB1A).
Author information
Authors and Affiliations
Contributions
JSC, SBT, and BL designed the study. WS carried out the major experiment work. DL helped manage aged WT and Apoe−/− mice. ZKC helped analyze RNA sequencing data. YNW helped perform RT-qPCR. WS wrote the manuscript; JSC and SBT revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Ethical Approval
Animal procedures were approved by the Animal Welfare and Ethics Committee of Central South University and were conducted in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sheng, W., Lv, D., Cui, ZK. et al. Tissue-Specific Gamma-Flicker Light Noninvasively Ameliorates Retinal Aging. Cell Mol Neurobiol 42, 2893–2907 (2022). https://doi.org/10.1007/s10571-021-01160-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-021-01160-w