Abstract
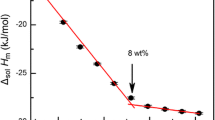
The effects of chloride salts on the dissolution of cellobiose in aqueous solution were investigated using calorimetry and 1H NMR. The dissolution of cellobiose in salt solutions is a typical entropy-driven process. The activity of ZnCl2 and LiCl hydrated ions is enhanced as the hydration number decreases with increasing temperature. Zn2+ and Li+ hydrates can interact with the oxygen atoms at the O5 and O6 positions of cellobiose and associate with the Cl− anions, leading to the breakage of cellobiose hydrogen bonds. We found that the solubility of cellobiose in aqueous solutions is on the order of ZnCl2 > LiCl > NaCl > H2O > KCl > NH4Cl, which is consistent with the Hofmeister series. For the first time, we recognized the specific ionic effects of the Hofmeister series on the dissolution of cellobiose in salt aqueous solutions. This finding is helpful for understanding the dissolving mechanism of cellulose in aqueous solvents with salts and providing fundamental knowledge for finding and designing new cellulose solvents.






Similar content being viewed by others
References
Allolio C, Salas-Illanes N, Desmukh YS, Hansen MR, Sebastiani D (2013) H-bonding competition and clustering in aqueous LiI. J Phys Chem B 117:9939–9946
Altaner CM, Thomas LH, Fernandes AN, Jarvis MC (2014) How cellulose stretches: synergism between covalent and hydrogen bonding. Biomacromolecules 15:791–798
Arakawa T, Timasheff SN (1984) Mechanism of protein salting in and salting out by divalent-cation salts—balance between hydration and salt binding. Biochemistry 23:5912–5923
Collins KD (2004) Ions from the Hofmeister series and osmolytes: effects on proteins in solution and in the crystallization process. Methods 34:300–311
Collins KD, Washabaugh MW (1985) The hofmeister effect and the behavior of water at interfaces. Q Rev Biophys 18:323–422
Collins KD, Neilson GW, Enderby JE (2007) Ions in water: characterizing the forces that control chemical processes and biological structure. Biophys Chem 128:95–104
Cooper A (2011) Microcalorimetry of heat capacity and volumetric changes in biomolecular interactions-the link to solvation? J Therm Anal Calorim 104:69–73
Cox JD, Riedel O (1974) Recommended reference materials for the realization of physicochemical properties: enthalpy. Pure Appl Chem 40:432–433
Fischer S, Voigt W, Fischer K (1999) The behaviour of cellulose in hydrated melts of the composition LiX·nH2O (X = I−, NO3 −, CH3COO−, ClO4 −). Cellulose 6:213–219
Fischer S, Leipner H, Thummler K, Brendler E, Peters J (2003) Inorganic molten salts as solvents for cellulose. Cellulose 10:227–236
Florin E, Kjellander R, Eriksson JC (1984) Salt effects on the cloud point of the poly(ethylene oxide) + water-system. J Chem Soc -Faraday Trans I 80:2889–2910
Garbacz P, Price WS (2014) 1H NMR diffusion studies of water self-diffusion in supercooled aqueous sodium chloride solutions. J Phys Chem A 118:3307–3312
Hattori M, Koga T, Shimaya Y, Saito M (1998a) Aqueous calcium thiocyanate solution as a cellulose solvent. Structure and interactions with cellulose. Polym J 30:43–48
Hattori M, Shimaya Y, Saito M (1998b) Solubility and dissolved cellulose in aqueous calcium- and sodium-thiocyanate solution. Polym J 30:49–55
Hattori M, Shimaya Y, Saito M (1998c) Structural changes in wood pulp treated by 55 wt% aqueous calcium thiocyanate solution. Polym J 30:37–42
Heinze T (1998) New ionic polymers by cellulose functionalization. Macromol Chem Phys 199:2341–2364
Heinze T, Liebert T (2001) Unconventional methods in cellulose functionalization. Prog Polym Sci 26:1689–1762
Hindman JC (1962) Nuclear magnetic resonance effects in aqueous solutions of 1–1 electrolytes. J Chem Phys 36:1000–1015
Isobe N, Chen XX, Kim UJ, Kimura S, Wada M, Saito T, Isogai A (2013) TEMPO-oxidized cellulose hydrogel as a high-capacity and reusable heavy metal ion adsorbent. J Hazard Mater 260:195–201
Jiang ZW et al (2014) Intermolecular interactions and 3D structure in cellulose-NaOH-urea aqueous system. J Phys Chem B 118:10250–10257
Kalcher I, Horinek D, Netz RR, Dzubiella J (2009) Ion specific correlations in bulk and at biointerfaces. J Phys-Condes Matter 21:424108
Klemm D, Heublein B, Fink HP, Bohn A (2005) Cellulose: Fascinating biopolymer and sustainable raw material. Angew Chem Int Ed 44:3358–3393
Kuga S (1980) The porous structure of cellulose gel regenerated from calcium thiocyanate solution. J Colloid Interface Sci 77:413–417
Kunz W, Henle J, Ninham BW (2004) ‘Zur Lehre von der Wirkung der Salze’ (about the science of the effect of salts): Franz Hofmeister’s historical papers. Curr Opin Colloid Interface Sci 9:19–37
Leipner H, Fischer S, Brendler E, Voigt W (2000) Structural changes of cellulose dissolved in molten salt hydrates. Macromol Chem Phys 201:2041–2049
Letters K (1932) Viscosimetric analysis on the reaction of cellulose with concentrated zinc chloride solutions. Kolloid-Zeitschrift 58:229–239
Liebert T (2010) Cellulose solvents—remarkable history, bright future. In: Liebert T, Heinze T, Edgar KJ (eds) Cellulose solvents: for analysis, shaping and chemical modification, vol 1033. American Chemical Society, Washington, DC, pp 3–54. doi:10.1021/bk-2010-1033
Lindman B, Karlstrom G, Stigsson L (2010) On the mechanism of dissolution of cellulose. J Mol Liq 156:76–81
Liu ZJ et al (2015) Effects of additives on dissolution of cellobiose in aqueous solvents. Cellulose 22:1641–1652
Lukanoff B, Stern W, Loth F, Dautzenber H (1984) Spherical or flat cellulose article preparation by adding formalin to cellulose suspended in calcium thiocyanate melt, heating, cooling, shaping the solution obtained and coagulating. Patent number: DD206675-A
Malinows ER, Knapp PS, Feuer B (1966) NMR studies of aqueous electrolyte solutions. I. Hydration number of NaCl determined from temperature effects on proton shift. J Chem Phys 45:4274–4279
Medronho B, Lindman B (2014) Competing forces during cellulose dissolution: from solvents to mechanisms. Curr Opin Colloid Interface Sci 19:32–40
Missoum K, Bras J, Belgacem MN (2012) Water redispersible dried nanofibrillated cellulose by adding sodium chloride. Biomacromolecules 13:4118–4125
Muller N (1965) Concerning structural models for water and chemical-shift data. J Chem Phys 43:2555–2556
Nishio Y, Chiba R, Miyashita Y, Oshima K, Miyajima T, Kimura N, Suzuki H (2002) Salt addition effects on mesophase structure and optical properties of aqueous hydroxypropyl cellulose solutions. Polym J 34:149–157
Nishiyama Y, Langan P, Chanzy H (2002) Crystal structure and hydrogen-bonding system in cellulose Iβ from synchrotron X-ray and neutron fiber diffraction. J Am Chem Soc 124:9074–9082
Okur HI, Kherb J, Cremer PS (2013) Cations bind only weakly to amides in aqueous solutions. J Am Chem Soc 135:5062–5067
Piekarski H, Nowicka B (2010) Calorimetric studies of interactions of some peptides with electrolytes, urea and ethanol in water at 298.15 K. J Therm Anal Calorim 102:31–36
Ragauskas AJ et al (2006) The path forward for biofuels and biomaterials. Science 311:484–489
Roshind MU, Tahtinen P, Niemitz M, Sjhohn R (2008) Complete assignments of the 1H and 13C chemical shifts and J(H, H) coupling constants in NMR spectra of D-glucopyranose and all D-glucopyranosyl-D-glucopyranosides. Carbohydr Res 343:101–112
Schaschel E, Day MC (1968) Ion–solvent interaction. Solvation of the sodium ion. J Am Chem Soc 90:503–504
Schneider WG, Bernstein HJ, Pople JA (1958) Proton magnetic resonance chemical shift of free (gaseous) and associated (liquid) hydride molecules. J Chem Phys 28:601–607
Shoolery JN, Alder BJ (1955) Nuclear magnetic resonance in concentrated aqueous electrolytes. J Chem Phys 23:805–811
Solomon BD, Barnes JR, Halvorsen KE (2007) Grain and cellulosic ethanol: history, economics, and energy policy. Biomass Bioenerg 31:416–425
Thormann E (2012) On understanding of the Hofmeister effect: how addition of salt alters the stability of temperature responsive polymers in aqueous solutions. RSC Adv 2:8297–8305
Traube J (1910) The attraction pressure. J Phys Chem 14:452–470
Walrafen GE (1966) Raman spectral studies of effects of temperature on water and electrolyte solutions. J Chem Phys 44:1546–1558
Warwicke JO (1967) Effect of chemical reagents on fine structure of cellulose. 4. Action of caustic soda on fine structure of cotton and ramie. J Polym Sci: Polym Chem 5:2579–2593
Yang YJ, Shin JM, Kang TH, Kimura S, Wada M, Kim UJ (2014) Cellulose dissolution in aqueous lithium bromide solutions. Cellulose 21:1175–1181
Zhang YJ, Cremer PS (2006) Interactions between macromolecules and ions: the Hofmeister series. Curr Opin Chem Biol 10:658–663
Zhang YJ, Cremer PS (2010) Chemistry of Hofmeister anions and osmolytes. Annu Rev Phys Chem 61:63–83
Zhang Y, Furyk S, Bergbreiter DE, Cremer PS (2005) Specific ion effects on the water solubility of macromolecules: PNIPAM and the Hofmeister series. J Am Chem Soc 127:14505–14510
Zhang C, Liu RG, Xiang JF, Kang HL, Liu ZJ, Huang Y (2014) Dissolution mechanism of cellulose in N, N-dimethylacetamide/lithium chloride: revisiting through molecular interactions. J Phys Chem B 118:9507–9514
Acknowledgments
Thanks to Prof. H. K. Yan from the Institute of Chemistry, Chinese Academy of Sciences for the valuable discussions. Financial support from the National Natural Science Foundation of China (grant nos. 21274154, 51473174 and 51373191) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Z., Zhang, C., Liu, R. et al. Dissolution of cellobiose in the aqueous solutions of chloride salts: Hofmeister series consideration. Cellulose 23, 295–305 (2016). https://doi.org/10.1007/s10570-015-0827-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-015-0827-4