Abstract
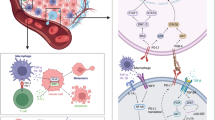
Loss of expression or protein kinase B (Akt1)-mediated post-translational modification of the RNA binding protein Poly r(C) binding protein 1 (PCBP1) is closely related to metastatic advancement of breast cancer. However, the role of PCBP1 in tumorigenesis is not completely defined. Using a xenograft orthotopic model of breast tumorigenesis (4T1-Pcbp1−/−), we show here that PCBP1 knockdown-induced tumorigenesis is inhibited by activation of the WNT signaling via treating with the glycogen synthase kinase 3 beta inhibitor TWS119, but not the Akt2/Akt3 inhibitor GSK690693. Mass cytometry-based evaluation of the tumor microenvironment (TME) revealed significantly more regulatory T cells (Tregs) and significantly less cytotoxic T cells in 4T1-Pcbp1−/−mice treated with saline control in comparison to mice treated with TWS119. Infiltrating cytotoxic T cells were phenotypically and functionally exhausted. Treatment with TWS119 resulted in rescue of cytotoxic T cell function and inhibition of suppressor activity of Tregs. Using cytotoxic T cells isolated from healthy donors, we show that TWS119-induced WNT signaling-mediated inhibition of cytotoxic T cell expansion is reliant on expression of PCBP1. In conclusion, decreased PCBP1 expression favors breast tumorigenesis by potentiating skewing of tumor infiltrating T cells towards Tregs, thereby effectively suppressing anti-tumor immunity.
Graphical abstract








Similar content being viewed by others
Data availability
Data related to results presented in this manuscript are either incorporated in the manuscript or will be accessible upon request to the corresponding author.
Code availability
No custom algorithm, software, or code was used in the present work.
References
Ansa-Addo, E. A., Huang, H. C., Riesenberg, B., Iamsawat, S., Borucki, D., Nelson, M. H., . . . Li, Z. (2020). RNA binding protein PCBP1 is an intracellular immune checkpoint for shaping T cell responses in cancer immunity. Sci Adv, 6(22), eaaz3865. doi:https://doi.org/10.1126/sciadv.aaz3865
C., G. E. B. C. T. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;365(9472):1687–717. https://doi.org/10.1016/s0140-6736(05)66544-0.
Chaudhury A, Hussey GS, Ray PS, Jin G, Fox PL, Howe PH. TGF-beta-mediated phosphorylation of hnRNP E1 induces EMT via transcript-selective translational induction of Dab2 and ILEI. Nat Cell Biol. 2010;12(3):286–93. https://doi.org/10.1038/ncb2029.
Cho SJ, Jung YS, Chen X. Poly (C)-binding protein 1 regulates p63 expression through mRNA stability. PLoS ONE. 2013;8(8): e71724. https://doi.org/10.1371/journal.pone.0071724.
de Bono JS, Tolcher AW, Rowinsky EK. The future of cytotoxic therapy: selective cytotoxicity based on biology is the key. Breast Cancer Res. 2003;5(3):154–9. https://doi.org/10.1186/bcr597.
Driessens G, Zheng Y, Locke F, Cannon JL, Gounari F, Gajewski TF. Beta-catenin inhibits T cell activation by selective interference with linker for activation of T cells-phospholipase C-gamma1 phosphorylation. J Immunol. 2011;186(2):784–90. https://doi.org/10.4049/jimmunol.1001562.
Finck R, Simonds EF, Jager A, Krishnaswamy S, Sachs K, Fantl W, Bendall SC. Normalization of mass cytometry data with bead standards. Cytometry A. 2013;83(5):483–94. https://doi.org/10.1002/cyto.a.22271.
Gattinoni L, Zhong XS, Palmer DC, Ji Y, Hinrichs CS, Yu Z, Restifo NP. Wnt signaling arrests effector T cell differentiation and generates CD8+ memory stem cells. Nat Med. 2009;15(7):808–13. https://doi.org/10.1038/nm.1982.
Ghanem LR, Chatterji P, Liebhaber SA. Specific enrichment of the RNA-binding proteins PCBP1 and PCBP2 in chief cells of the murine gastric mucosa. Gene Expr Patterns. 2014;14(2):78–87. https://doi.org/10.1016/j.gep.2014.01.004.
Greaves E, Temp J, Esnal-Zufiurre A, Mechsner S, Horne AW, Saunders PT. Estradiol is a critical mediator of macrophage-nerve cross talk in peritoneal endometriosis. Am J Pathol. 2015;185(8):2286–97. https://doi.org/10.1016/j.ajpath.2015.04.012.
Hovanes K, Li TW, Munguia JE, Truong T, Milovanovic T, Lawrence Marsh J, Waterman ML. Beta-catenin-sensitive isoforms of lymphoid enhancer factor-1 are selectively expressed in colon cancer. Nat Genet. 2001;28(1):53–7. https://doi.org/10.1038/ng0501-53.
Hussey GS, Chaudhury A, Dawson AE, Lindner DJ, Knudsen CR, Wilce MC, Howe PH. Identification of an mRNP complex regulating tumorigenesis at the translational elongation step. Mol Cell. 2011;41(4):419–31. https://doi.org/10.1016/j.molcel.2011.02.003.
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69–90. https://doi.org/10.3322/caac.20107.
Li J, Zhang BN, Fan JH, Pang Y, Zhang P, Wang SL, Qiao YL. A nation-wide multicenter 10-year (1999–2008) retrospective clinical epidemiological study of female breast cancer in China. BMC Cancer. 2011;11:364. https://doi.org/10.1186/1471-2407-11-364.
Lian WX, Yin RH, Kong XZ, Zhang T, Huang XH, Zheng WW, Yang XM. THAP11, a novel binding protein of PCBP1, negatively regulates CD44 alternative splicing and cell invasion in a human hepatoma cell line. FEBS Lett. 2012;586(10):1431–8. https://doi.org/10.1016/j.febslet.2012.04.016.
Mann B, Gelos M, Siedow A, Hanski ML, Gratchev A, Ilyas M, Hanski C. Target genes of beta-catenin-T cell-factor/lymphoid-enhancer-factor signaling in human colorectal carcinomas. Proc Natl Acad Sci U S A. 1999;96(4):1603–8. https://doi.org/10.1073/pnas.96.4.1603.
Pastushenko I, Blanpain C. EMT transition states during tumor progression and metastasis. Trends Cell Biol. 2019;29(3):212–26. https://doi.org/10.1016/j.tcb.2018.12.001.
Reiser J, Banerjee A. Effector, memory, and dysfunctional CD8(+) T cell fates in the antitumor immune response. J Immunol Res. 2016;2016:8941260. https://doi.org/10.1155/2016/8941260.
Roose J, Huls G, van Beest M, Moerer P, van der Horn K, Goldschmeding R, Clevers H. Synergy between tumor suppressor APC and the beta-catenin-Tcf4 target Tcf1. Science. 1999;285(5435):1923–6. https://doi.org/10.1126/science.285.5435.1923.
Sayer HG, Kath R, Kliche KO, Hoffken K. Premenopausal breast cancer: chemotherapy and endocrine therapy. Drugs. 2002;62(14):2025–38. https://doi.org/10.2165/00003495-200262140-00003.
Shi Z, Zhang T, Long W, Wang X, Zhang X, Ling X, Ding H. Down-regulation of poly(rC)-binding protein 1 correlates with the malignant transformation of hydatidiform moles. Int J Gynecol Cancer. 2012;22(7):1125–9. https://doi.org/10.1097/IGC.0b013e3182606ac3.
Song Q, Sheng W, Zhang X, Jiao S, Li F. ILEI drives epithelial to mesenchymal transition and metastatic progression in the lung cancer cell line A549. Tumour Biol. 2014;35(2):1377–82. https://doi.org/10.1007/s13277-013-1188-y.
Sporn MB. The war on cancer. Lancet. 1996;347(9012):1377–81. https://doi.org/10.1016/s0140-6736(96)91015-6.
Staal FJ, Luis TC, Tiemessen MM. WNT signalling in the immune system: WNT is spreading its wings. Nat Rev Immunol. 2008;8(8):581–93. https://doi.org/10.1038/nri2360.
Wang H, Vardy LA, Tan CP, Loo JM, Guo K, Li J, Zeng Q. PCBP1 suppresses the translation of metastasis-associated PRL-3 phosphatase. Cancer Cell. 2010;18(1):52–62. https://doi.org/10.1016/j.ccr.2010.04.028.
Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol. 2015;15(8):486–99. https://doi.org/10.1038/nri3862.
Xue X, Wang X, Liu Y, Teng G, Wang Y, Zang X, Pan L. SchA-p85-FAK complex dictates isoform-specific activation of Akt2 and subsequent PCBP1-mediated post-transcriptional regulation of TGFbeta-mediated epithelial to mesenchymal transition in human lung cancer cell line A549. Tumour Biol. 2014;35(8):7853–9. https://doi.org/10.1007/s13277-014-1982-1.
Zeng YA, Verheyen EM. Nemo is an inducible antagonist of Wingless signaling during Drosophila wing development. Development. 2004;131(12):2911–20. https://doi.org/10.1242/dev.01177.
Zhang HY, Dou KF. PCBP1 is an important mediator of TGF-beta-induced epithelial to mesenchymal transition in gall bladder cancer cell line GBC-SD. Mol Biol Rep. 2014;41(8):5519–24. https://doi.org/10.1007/s11033-014-3428-7.
Zhang ZZ, Shen ZY, Shen YY, Zhao EH, Wang M, Wang CJ, Xu J. HOTAIR long noncoding RNA promotes gastric cancer metastasis through suppression of poly r(C)-binding protein (PCBP) 1. Mol Cancer Ther. 2015;14(5):1162–70. https://doi.org/10.1158/1535-7163.MCT-14-0695.
Funding
This study were supported by the Key R&D Project of Jilin Province Science and Technology Department (20200404144YY) and Jilin Provincial Development and Reform Commission Project (2019C052-5). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Z.Y.Y and G.S. designed this study; W.L.Z and C.W.J carried out experiments and analyzed data; Z.Y.Y and G.S. prepared the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures were performed under the approval of the Laboratory Animal Welfare and Ethics Committee of the China-Japan Union Hospital of Jilin University.
Consent to participate
Not applicable.
Consent for publication
All authors have read and approved the final manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• PCBP1 functions as the upstream of GSK-3β/WNT signaling during epithelial to mesenchymal transition in breast cancer.
• Activation of WNT signaling by GSK3β inhibitor TWS119 results in functional impairment of Tregs and potentiates effector function of CD8+ T cells
• TWS119-induced WNT signaling-mediated inhibition of cytotoxic T cell expansion is reliant on the expression of PCBP1
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, Zy., Zhang, Wl., Jiang, Cw. et al. PCBP1-mediated regulation of WNT signaling is critical for breast tumorigenesis. Cell Biol Toxicol 39, 2331–2343 (2023). https://doi.org/10.1007/s10565-022-09722-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-022-09722-4