Abstract
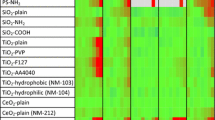
The potential mammalian hepatotoxicity of nanomaterials was explored in dose-response and structure-activity studies in human hepatic HepG2 cells exposed to between 10 and 1000 μg/ml of five different CeO2, three SiO2, and one TiO2-based particles for 3 days. Various biochemical parameters were then evaluated to study cytotoxicity, cell growth, hepatic function, and oxidative stress. Few indications of cytotoxicity were observed between 10 and 30 μg/ml. In the 100 to 300 μg/ml exposure range, a moderate degree of cytotoxicity was often observed. At 1000 μg/ml exposures, all but TiO2 showed a high degree of cytotoxicity. Cytotoxicity per se did not seem to fully explain the observed patterns of biochemical parameters. Four nanomaterials (all three SiO2) decreased glucose 6-phosphate dehydrogenase activity with some significant decreases observed at 30 μg/ml. In the range of 100 to 1000 μg/ml, the activities of glutathione reductase (by all three SiO2) and glutathione peroxidase were decreased by some nanomaterials. Decreased glutathione concentration was also found after exposure to four nanomaterials (all three nano SiO2 particles). In this study, the more responsive and informative assays were glucose 6-phosphate dehydrogenase, glutathione reductase, superoxide dismutase, lactate dehydrogenase, and aspartate transaminase. In this study, there were six factors that contribute to oxidative stress observed in nanomaterials exposed to hepatocytes (decreased glutathione content, reduced glucose 6-phosphate dehydrogenase, glutathione reductase, glutathione peroxidase, superoxide dismutase, and increased catalase activities). With respect to structure-activity, nanomaterials of SiO2 were more effective than CeO2 in reducing glutathione content, glucose 6-phosphate dehydrogenase, glutathione reductase, and superoxide dismutase activities.
Similar content being viewed by others
References
Akhtar MJ, Ahamed M, Kumar S, Siddiqui H, Patil G, Ashquin M, et al. Nanotoxicity of pure silica mediated through oxidant generation rather than glutathione depletion in human lung epithelial cells. Toxicology. 2010;276(2):95–102.
Cassee FR, van Balen EC, Singh C, Green D, Muijser H, Weinstein J, et al. Exposure, health and ecological effects review of engineered nanoscale cerium and cerium oxide associated with its use as a fuel additive. Crit Rev Toxicol. 2011;41(3):213–29.
Cheng G, Guo W, Han L, Chen E, Kong L, Wang L, et al. Cerium oxide nanoparticles induce cytotoxicity in human hepatoma SMMC-7721 cells via oxidative stress and the activation of MAPK signaling pathways. Toxicol in Vitro. 2013;27(3):1082–8.
Demokritou P, Gass S, Pyrgiotakis G, Cohen JM, Goldsmith W, McKinney W, et al. An in vivo and in vitro toxicological characterisation of realistic nanoscale CeO(2) inhalation exposures. Nanotoxicology. 2013;7(8):1338–50.
Ge Y, Bruno M, Wallace K, Winnik W, Prasad RY. Proteome profiling reveals potential toxicity and detoxification pathways following exposure of BEAS-2B cells to engineered nanoparticle titanium dioxide. Proteomics. 2011;11(12):2406–22.
Geraets L, Oomen AG, Schroeter JD, Coleman VA, Cassee FR. Tissue distribution of inhaled micro- and nano-sized cerium oxide particles in rats: results from a 28-day exposure study. Toxicol Sci. 2012;127(2):463–73.
Grulke E, Reed K, Beck M, Huang XY, Cormack A, Seal S. Nanoceria: factors affecting its pro- and anti- oxidant properties. Environ Sci Nano. 2014;1:429–44.
Hardas S, Sultana SR, Warrier G, Dan M, Florence RL, Wu P, et al. Rat brain pro-oxidant effects of peripherally administered 5 nm ceria 30 days after exposure. Neurotoxicology. 2012;33(5):1147–55.
Holsapple MP, Farland WH, Landry TD, Monteiro-Riviere NA, Carter JM, Walker NJ, et al. Research strategies for safety evaluation of nanomaterials, part II: toxicological and safety evaluation of nanomaterials, current challenges and data needs. Toxicol Sci. 2005;88(1):12–7.
Iavicoli I, Leso V, Fontana L, Bergamaschi A. Toxicological effects of titanium dioxide nanoparticles: a review of in vitro mammalian studies. Eur Rev Med Pharmacol Sci. 2011;15(5):481–508.
Khan MM, Farooq S, Al-Mayouf AA. Metal oxides as photocatalysts. J Saudi Chem Soc. 2015;19:462–4.
Kitchin KT, Prasad RY, Wallace KA. Oxidative stress studies of six TiO(2) and two CeO(2) nanomaterials: immuno-spin trapping results with DNA. Nanotoxicology. 2011;5(4):546–56.
Kitchin KT, Grulke EA, Robinette BL, Castellon BT. Metabolomic effects in HepG2 cells exposed to four TiO2 and two CeO2 nanomaterials. Environ Sci Nano. 2014;1:466–77.
Kitchin KT, Robinette BL, Richards J, Coates NH, Castellon BT. Biochemical effects in HepG2 cells exposed to six TiO2 and four CeO2 nanomaterials. J Nanosci Nanotechnol. 2016;16(9):9505–34.
Kumar A, Pandey AK, Singh SS, Shanker R, Dhawan A. Engineered ZnO and TiO(2) nanoparticles induce oxidative stress and DNA damage leading to reduced viability of Escherichia coli. Free Radic Biol Med. 2011;51(10):1872–81.
Lin W, Huang YW, Zhou XD, Ma Y. Toxicity of cerium oxide nanoparticles in human lung cancer cells. Int J Toxicol. 2006;25(6):451–7.
Merrifield RC, Wang ZW, Palmer RE, Lead JR. Synthesis and characterization of polyvinylpyrrolidone coated cerium oxide nanoparticles. Environ Sci Technol. 2013;47(21):12426–33.
Mishchuk NA. The model of hydrophobic attraction in the framework of classical DLVO forces. Adv Colloid Interf Sci. 2011;168(1–2):149–66.
Monteiller C, Tran L, MacNee W, Faux S, Jones A, Miller B, et al. The pro-inflammatory effects of low-toxicity low-solubility particles, nanoparticles and fine particles, on epithelial cells in vitro: the role of surface area. Occup Environ Med. 2007;64(9):609–15.
Monteiro-Riviere NA, Inman AO, Zhang LW. Limitations and relative utility of screening assays to assess engineered nanoparticle toxicity in a human cell line. Toxicol Appl Pharmacol. 2009;234(2):222–35.
Nel AE, Xia T, Madler L, Li N. Toxic potential of materials at the nanolevel. Science. 2006;311(5761):622–7.
Nel AE, Madler L, Velegol D, Xia T, Hoek EM, Somasundaran P, et al. Understanding biophysicochemical interactions at the nano-bio interface. Nat Mater. 2009;8(7):543–57.
Nemmar A, Yuvaraju P, Beegam S, Yasin J, Kazzam EE, Ali BH. Oxidative stress, inflammation, and DNA damage in multiple organs of mice acutely exposed to amorphous silica nanoparticles. Int J Nanomedicine. 2016;11:919–28.
Park EJ, Park K. Oxidative stress and pro-inflammatory responses induced by silica nanoparticles in vivo and in vitro. Toxicol Lett. 2009;184(1):18–25.
Polimeni M, Gazzano E, Ghiazza M, Fenoglio I, Bosia A, Fubini B, et al. Quartz inhibits glucose 6-phosphate dehydrogenase in murine alveolar macrophages. Chem Res Toxicol. 2008;21(4):888–94.
Porter D, Shiram K, Wolfarth M, Jefferson A, Schwegler-Berry D, Andrew M, et al. A biocompatible medium for nanoparticle dispersion. Nanotoxicology. 2008;2(3):144–54.
Price C, Alberti K. Biochemical assessment of liver function. In: Wright RM, Alberti K, Karran S, Millward-Sadler G, editors. Liver and Biliary Disease-Pathophysiology, Diagnosis, Management. London: W. B. Saunders; 1979. p. 381–416.
Ramesh R, Kavitha P, Kanipandian N, Arun S, Thirumurugan R, Subramanian P. Alteration of antioxidant enzymes and impairment of DNA in the SiO2 nanoparticles exposed zebra fish (Danio rerio). Environ Monit Assess. 2013;185(7):5873–81.
Shi H, Magaye R, Castranova V, Zhao J. Titanium dioxide nanoparticles: a review of current toxicological data. Part Fibre Toxicol. 2013;10:15. https://doi.org/10.1186/1743-8977-10-15.
Shvedova AA, Pietroiusti A, Fadeel B, Kagan VE. Mechanisms of carbon nanotube-induced toxicity: focus on oxidative stress. Toxicol Appl Pharmacol. 2012;261(2):121–33.
Stanca L, Petrache SN, Serban AI, Staicu AC, Sima C, Munteanu MC, et al. Interaction of silicon-based quantum dots with gibel carp liver: oxidative and structural modifications. Nanoscale Res Lett. 2013;8(1):254. https://doi.org/10.1186/1556-276X-8-254.
Thai SF, Wallace KA, Jones CP, Ren H, Castellon BT, Crooks J, et al. Differential genomic effects on signaling pathways by two different CeO2 nanoparticles in HepG2 cells. J Nanosci Nanotechnol. 2015a;15(12):9925–37.
Thai SF, Wallace KA, Jones CP, Ren H, Grulke EA, Castellon BT, et al. Differential genomic effects of six different TiO2 nanomaterials on human liver HepG2 cells. J Biochem Mol Toxicol. 2015b;30(7):331–41.
Thompson TL, Yates JT Jr. Surface science studies of the photoactivation of TiO2--new photochemical processes. Chem Rev. 2006;106(10):4428–53.
Tseng MT, Lu X, Duan X, Hardas SS, Sultana R, Wu P, et al. Alteration of hepatic structure and oxidative stress induced by intravenous nanoceria. Toxicol Appl Pharmacol. 2012;260(2):173–82.
Walker NJ, Bucher JR. A 21st century paradigm for evaluating the health hazards of nanoscale materials? Toxicol Sci. 2009;110(2):251–4.
Warheit DB, Borm PJ, Hennes C, Lademann J. Testing strategies to establish the safety of nanomaterials: conclusions of an ECETOC workshop. Inhal Toxicol. 2007;19(8):631–43.
Xu Y, Zhang Z, Hu J, Stillman IE, Leopold JA, Handy DE, et al. Glucose-6-phosphate dehydrogenase-deficient mice have increased renal oxidative stress and increased albuminuria. FASEB J. 2010;24(2):609–16.
Yokel RA, Hussain S, Garantziotis S, Demokritou P, Castranova V, Cassee FR. The Yin: an adverse health perspective of nanoceria: uptake, distribution, accumulation, and mechanisms of its toxicity. Environ Sci Nano. 2014;1(5):406–28.
Yu Y, Duan J, Li Y, Jin M, Li C, Wang Y, et al. Combined toxicity of amorphous silica nanoparticles and methylmercury to human lung epithelial cells. Ecotoxicol Environ Saf. 2015;112:144–52. https://doi.org/10.1096/fj.09-135731.
Acknowledgments
We are grateful for the participation of many individuals in this study. Particularly, we thank Drs. Carl Blackman, Michael F. Hughes, and Urmila Kodavanti for reviewing this manuscript as part of EPA clearance procedures. Dr. Xinhua Liang of Missouri University of Science and Technology performed the atomic layer deposition which produced nano SiO2 K1 and SiO2 N2.
Funding
US EPA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Disclaimer
The information in this document has been funded wholly by the U. S. Environmental Protection Agency. It has been subjected to review by the National Health and Environmental Effects Research Laboratory and approved for publication. Approval does not signify that the contents necessarily reflect the views of the Agency, nor does mention of trade names or commercial products that constitute endorsement or recommendation for use.
Electronic supplementary material
Supplementary Table 1
(DOCX 94 kb)
Supplementary Figure 1
% LDH released following metal oxide treatment of HepG2 cells (DOCX 155 kb)
Supplementary Figure 2
% AST released following metal oxide treatment of HepG2 cells (DOCX 165 kb)
Supplementary Figure 3
% ALT released following metal oxide treatment of HepG2 cells (DOCX 215 kb)
Supplementary Figure 4
Effects of nine metal oxides on T BIL concentration (DOCX 172 kb)
Supplementary Figure 5
Effects of nine metal oxides on HepG2 protein content (DOCX 201 kb)
Supplementary Figure 6
Effects of nine metal oxides on GPx activity (DOCX 178 kb)
Supplementary Figure 7
Effects of nine metal oxides on SOD activity (DOCX 199 kb)
Supplementary Figure 8
Effects of nine metal oxides on GSH concentration (DOCX 133 kb)
Supplementary Figure 9
Effects of nine metal oxides on GRD activity (DOCX 204 kb)
Supplementary Figure 10
Effects of nine metal oxides on G6PDH activity (DOCX 189 kb)
Rights and permissions
About this article
Cite this article
Kitchin, K.T., Richards, J.A., Robinette, B.L. et al. Biochemical effects of some CeO2, SiO2, and TiO2 nanomaterials in HepG2 cells. Cell Biol Toxicol 35, 129–145 (2019). https://doi.org/10.1007/s10565-018-9445-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-018-9445-x