Abstract
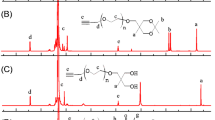
A new polymer-supported highly functionalized dendritic polyether with peripheral NHC–Pd complex was developed by the simple anionic ring opening polymerization of glycidol on bis(3-hydroxypropyl) amine grafted Merrifield resin and its subsequent functional modification of hydroxyl group to NHC–Pd complex. The polymeric material had high stability and swelling capacity in a wide range of solvents, even in water. Similar to supported higher generation dendrimer, the newly developed system showed high palladium content with uniformly distributed and well isolated metal nanocentres. The palladium content was found to be 2.02 mmol/g which was comparatively higher than other heterogeneous NHC–Pd based catalysts and the polymer was used as heterogeneous palladium catalyst for Suzuki coupling reaction. Various factors affecting the catalyst’s performance were studied. The high availability and increased accessibility of catalytic sites, stability and dendrimer like property of the polymeric support, reusability, better resistance to metal leaching, easy synthesis etc. are the important features of this newly synthesized catalytic system.
Graphical Abstract











Similar content being viewed by others
References
Bhagiyalakshmi M, Park SD, Cha WS, Jang HT (2010) Appl Surf Sci 256:6660
Wang X, Gao H (2017) Polymers 9:188
Hirao A, Yoo HS (2011) Polym J 43:2
Schüll C, Frey H (2012) ACS Macro Lett 1:461
Gao C, Yan D (2004) Prog Polym Sci 29:183
Voit BI, Lederer A (2009) Chem Rev 109:5924
Wilms D, Stiriba S, Frey H (2010) Acc Chem Res 43:129
Oikawa Y, Lee S, Kim DH, Kang DH, Kim BS, Saito K, Sasaki S, Oishi Y, Shibasaki Y (2013) Biomacromol 14:2171
Turk H, Haag R, Alban S (2004) Bioconjugate Chem 15:162
Kainthan RK, Janzen J, Levin E, Devin DV, Brooks DE (2006) Biomacromol 7:703
Frey H, Haag R (2002) Rev Mol Biotechnol 90:257
Siegers C, Biesalski M, Haag R (2004) Chem Eur J 10:2831
Venkatakrishnan S, Thengarai V, Keilitz J, Haag R (2014) Inorg Chim Acta 409:179
Ferruti P, Knobloch S, Ranucci E, Duncan R, Gianasi E (1998) Macromol. Chem Phys 199:2565
Wroblewski S, Kopeckova J (1998) Macromol Chem Phys 199:2601
Mammen M, Choi SK, Whitesides GM (1998) Angew Chem Int Ed 37:2754
Li H, Jo JK, Zhang LD, Ha CS, Suh H, Kim I (2010) Langmuir 23:18442
Pecchioli T, Kumar MM, Haag R, Christmann M (2015) Beilstein J Org Chem 11:730
Berkessel A, Kramer J, Mummy F, Neudçrfl JM, Haag R (2013) Angew Chem Int Ed 52:739
Hebel A, Haag R (2002) J Org Chem 67:9452
King ASH, Twyman LJ (2002) J Chem Soc 1:2209
Schüll C, Frey H (2013) Polymer 54:5443
Roller S, Turk H, Stumbe JF, Rapp W, Haag R (2006) J Comb Chem 8:350
Dong Z, Ye Z (2015) Appl Catal A 489:61
Murugan E, Shanmugam P (2016) J Nanosci Nanotechnol 16(1):426
Rezaei SJT, Shamseddin A, Ramazani A, Malekzadeh AM, Asiabi PA (2017) Appl Organomet Chem 31:e3707
He Y, Cai C (2011) Chem Commun 47:12319
Tamami B, Nezhad MM, Ghasemi S, Farjadian F (2016) Phosphorus, Sulfur Silicon Relat Elem 191:123
He Y, Lv MF, Cai C (2012) Dalton Trans 41:12428
Schwarz J, Böhm VPW, Gardiner MG, Grosche M, Herrmann WA, Hieringer W, Raudaschl-Sieber G (2000) Chem Eur J 6:1773
Díez-González S, Marion N, Nolan SP (2009) Chem Rev 109:3612
Peris E, Loch JA, Mata J, Crabtree RH (2001) Chem Commun. https://doi.org/10.1039/B008038L
Kang T, Feng Q, Luo M (2005) Synlett 15:2305
Lázaro G, Fernández-Alvarez FJ, Iglesias M, Horna C, Vispe E, Sancho R, Lahoz FJ, Iglesias M, Pérez-Torrente JJ, Oro LA (2014) Catal Sci Technol 4:62
Byun JW, Lee YS (2004) Tetrahedron Lett 45:1837
Kim JH, Jun BH, Byun JW, Lee YS (2004) Tetrahedron Lett 45:5827
Kim JW, Kim JH, Lee DH, Lee YS (2006) Tetrahedron Lett 47:4745
Jadhav SN, Kumbhar AS, Mali SS, Hongc CK, Salunkhe RS (2015) New J Chem 39:2333
Ranganath KVS, Onitsuka S, Kiran Kumar A, Inanaga J (2013) Catal. Sci Technol 3:2161
Rostamnia S, Hossieni HG, Doustkhah E (2015) J Organomet Chem 791:18
Haag R, Sunder A, Stumbe JF (2000) J Am Chem Soc 122:2954
Jisha KA, Sreekumar K (2017) Catal Lett 147:964
Siyad MA, Nair ASV, Kumar GSV (2010) J Comb Chem 12:298
Wang X, Hu P, Xue F, Wei Y (2014) Carbohydr Polym 114:476
Meise M, Haag R (2008) Chemsuschem 1:637
Kale D, Rashinkar G, Kumbhar A, Salunkhe R (2017) React Funct Polym 116:9
Rajesh KG, Sreekumar K (2010) Soft Mater 2:114
Bukhryakov KV, Mugemana C, Vu KB, Rodionov VO (2015) Org Lett 17:4826
Acknowledgements
The authors gratefully acknowledge STIC-CUSAT, IISc- Bangalore, and NCESS-TVM, for the assistance in various spectral analysis. One of the authors (K. J. A) acknowledges the CSIR, Govt. of India for financial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Anjali, J.K., Sreekumar, K. Heterogeneous High-Loading Hyperbranched Polyglycidol with Peripheral NHC–Pd Complex: Synthesis and Application as Catalyst in Suzuki Coupling Reaction. Catal Lett 149, 1952–1964 (2019). https://doi.org/10.1007/s10562-019-02784-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-019-02784-w