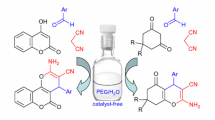
Abstract
A simple, highly efficient and green route for the synthesis of 2-amino-4H-benzo[b]pyrans and 1,4-dihydropyridines has been developed by using γ-cyclodextrin (γ-CD) as catalyst in deep eutectic solvent (DES) of urea-choline chloride (urea-ChCl), for the first time, accomplished via a one-pot, three-component strategy. All the reactions were successfully carried out under mild conditions and gave good to excellent yields (86–98%) in 8–28 min in the presence of 5 mol% of the γ-CD catalyst. The short reaction time, green reaction medium, moderate reaction conditions, simple work-up procedure and use of easily available reagents are the attractive features of this novel synthetic method. More importantly, the urea-ChCl-γ-CD catalytic system was regenerated and reused up to 6 times with a slight drop in the product yields.
Graphical Abstract






Similar content being viewed by others
References
Wang JL, Liu D, Zhang ZJ, Shan S, Han X, Srinivasula SM, Croce CM, Alnemri ES, Huang Z (2000) Proc Natl Acad Sci USA 97:7124
Zhang G, Zhang Y, Yan J, Chen R, Wang S, Ma Y, Wang R (2012) J Org Chem 77:878
Stout DM, Meyers AI (1982) Chem Rev 82:223
Gourdeau H, Leblond L, Hamelin B, Desputeau C, Dong K, Kianicka I, Custeau D, Bourdeau C, Geerts L, Cai SX, Drewe J, Labrecque D, Kasibhatla S, Tseng B (2004) Mol Cancer Ther 3:1375
Kemnitzer W, Drewe J, Jiang S, Zhang H, Wang Y, Zhao J, Jia S, Herich J, Labreque D, Storer R, Meerovitch K, Bouffard D, Rej R, Denis R, Blais C, Lamothe S, Attardo G, Gourdeau H, Tseng B, Kasibhatla S, Cai SX (2004) J Med Chem 47:6299
Chandra KS, Ramesh G (2013) Indian Heart J 65:691
Sambongi Y, Nitta H, Ichihashi K, Futai M, Ueda I (2002) J Org Chem 67:3499
Valente S, Mellini P, Spallotta F, Carafa V, Nebbioso A, Polletta L, Carnevale I, Saladini S, Trisciuoglio D, Gabellini C, Tardugno M, Zwergel C, Cencioni C, Atlante S, Moniot S, Steegborn C, Budriesi R, Tafani M, Bufalo DD, Altucci L, Gaetano C, Mai A (2016) J Med Chem 59:1471
Moghaddas M, Davoodnia A (2015) Res Chem Intermed 41:4373
Elnagdi NMH, Hokbany NS (2012) Molecules 17:4300
Devi I, Bhujyan PJ (2004) Tetrahedron Lett 45:8625
Azath IA, Puthiaraj P, Pitchumani K (2013) ACS Sustain Chem Eng 1:174
Sharma P, Gupta M (2015) Green Chem 17:1100
Khurana JM, Nand B, Saluja P (2010) Tetrahedron 66:5637
Heravi MM, Jani BA, Derikvand F, Bamoharram FF, Oskooie HA (2008) Catal Commun 10:272
Albadi J, Mansournezhad A (2016) Res Chem Intermed 42:5739
Ahad A, Farooqui M (2017) Res Chem Intermed 43:2445
Ko S, Sastry MN, Lin VC, Yao CF (2005) Tetrahedron Lett 46:5771
Sun J, Xia EY, Wu Q, Yan CG (2010) Org Lett 12:3678
Otokesh S, Koukabi N, Kolvari E, Amoozadeh A, Malmir M, Azhari S (2015) S Afr J Chem 68:15
Pal S, Singh V, Das P, Choudhury LH (2013) Bioorg Chem 48:8
Maleki A, Firouzi-Haji R, Hajizadeh Z (2018) Inter J Bio Macro 116:320
Safari J, Banitaba SH, Khalili SD (2011) J Mol Catal A: Chem 335:46
Bamoniri A, Fouladgar S (2015) RSC Adv 5:78483
Safari J, Azizi F, Sadeghi M (2015) New J Chem 39:1905
Rekunge DS, Khatri CK, Chaturbhuj GU (2017) Tetrahedron Lett 58:1240
Shabalala S, Maddila S, Van Zyl WE, Jonnalagadda SB (2016) Catal Commun 79:21
Bhaskaruni SVHS, Maddila S, van Zyl WE, Jonnalagadda SB (2018) Catal Today 309:276
Moradi L, Zare M (2018) Green Chem Lett Rev 11:197
Amoli T, Baghbanian SM (2018) Res Chem Intermed 44:3389
Nasrollahi SMH, Ghasemzadeh MA, Zolfaghari MR (2018) Acta Chim Slov 65:199
Safaei-Ghomi J, Ziarati A, Teymuri R (2012) Bull Korean Chem Soc 33:2679
Viswanath IVK, Murthy YLN (2013) Chem Sci Trans 2:227
Abbott AP, Boothby D, Capper G, Davies DL, Rasheed RK (2004) J Am Chem Soc 126:9142
Paiva A, Craveiro R, Aroso I, Martins M, Reis RL, Duarte ARC (2014) ACS Sustain Chem Eng 2:1063
María P, Zaira M (2011) Curr Opin Chem Biol 15:220
Smith EL, Abbott AP, Ryder KS (2014) Chem Rev 114:11060
Gu YL, Jerome F (2013) Chem Soc Rev 42:9550
Liu P, Hao JW, Mo LP, Zhang ZH (2015) RSC Adv 5:48675
Xiong XQ, Han Q, Shi L, Xiao SY, Bi C (2016) Chin J Org Chem 36:480
Zhang ZH, Zhang XN, Mo LP, Li YX, Ma FP (2012) Green Chem 14:1502
Zhang M, Liu YH, Shang ZR, Hu HC, Zhang ZH (2017) Catal Commun 88:39
Zhang M, Liu P, Liu YH, Shang ZR, Hu HC, Zhang ZH (2016) RSC Adv 6:106160
Perrone S, Capua M, Messa F, Salomone A (2017) Tetrahedron 73:6193
Kafle A, Handy ST (2017) Tetrahedron 73:7024
Shaibuna M, Theresa LV, Sreekumar K (2018) Catal Lett 148:2359
Fihri A, Bouhrara M, Nekoueishahraki B, Basset JM, Polshettiwar V (2011) Chem Soc Rev 40:5181
Abbasi M (2017) J Chin Chem Soc 64:896
Natarajan M, Kasi P (2018) Chemistryselect 3:10886
Kanagaraj K, Pitchumani K (2013) J Org Chem 78:744
Kanagaraj K, Pitchumani K (2010) Tetrahedron Lett 51:3312
Ren YF, Zhang W, Lu J, Gao K, Liao XL, Chen XZ (2015) RSC Adv 5:79405
Ren YF, Yang B, Liao XL (2016) RSC Adv 6:22034
Ren YF, Yang B, Liao XL (2016) Catal Sci Tech 12:4283
Xiong XQ, Yi C, Liao X, Lai SL (2019) Tetrahedron Lett 60:402
Xiong XQ, Chen HX, Liao X, Lai SL, Gao LZ (2018) ChemistrySelect 3:8819
Xiong XQ, Tang ZK, Sun ZH, Meng XQ, Song SD, Quan ZL (2018) Appl Organometal Chem 32:e3946
Xiong XQ, Yi C, Han Q, Shi L, Li SZ (2015) Chin J Catal 36:237
Han Q, Xiong XQ, Li SZ (2015) Catal Commun 58:85
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (21004024), the Natural Science Foundation of Fujian province (2016J01063), the Program for New Century Excellent Talents in University of Fujian province (2012FJ-NCET-ZR03) and the Promotion Program for Young and Middle-aged Teacher in Science and Technology Research of Huaqiao University (ZQN-YX103).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xiong, X., Yi, C., Liao, X. et al. An Effective One-Pot Access to 2-Amino-4H-benzo[b]pyrans and 1,4-Dihydropyridines via γ-Cyclodextrin-Catalyzed Multi-Component Tandem Reactions in Deep Eutectic Solvent. Catal Lett 149, 1690–1700 (2019). https://doi.org/10.1007/s10562-019-02767-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-019-02767-x