Abstract
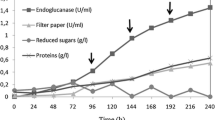
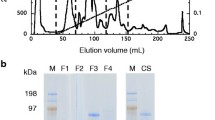
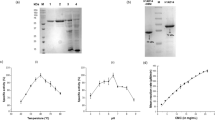
Two neutral endoglucanases (EG1 and EG2) from Stachybotrys microspora were purified and studied for their hydrolysis capacity and product profiles. Contrary to most of cellulases, especially the fungal endoglucanases such as those from Trichoderma reesei the most studied fungus, both EG1 and EG2 are more active on β-glucan from barely (β-1,3; β-1,4) than on carboxymethyl cellulose (CMC) (β-1,4). This result is much researched. Both enzymes are non specific. They hydrolyze less efficiently (30%) laminarin and only EG1 was able to attack xylan at 38%. The effectiveness of both cellulases was compared using these carbohydrates as substrates. EG1 had greater activity than EG2 on both substrates. Additionally, maximal activity (~fourfold) was observed in the presence of β-glucan from barely. The distinguishing properties of EG1 and EG2 are presented in the kinetic parameters (km, Vmax), turnover number (kcat) and catalytic efficiency (kcat/Km). EG1 catalyzed the hydrolysis of CMC with a Km of 8.048 mg/mL and a Vmax of 16.58 U/min/mg, more efficiently than EG2 with a Km of 13.71 mg/mL and a Vmax of 2.524 U/min/mg. Both purified cellulases have two distinct catalytic mechanisms. EG1 was able to hydrolyze CMC and β-glucan into soluble cellooligosaccharides (cellobiose, cellotriose) and (cellotriose, cellotetraose, cellobiose) as dominants products respectively, with a tiny amounts of glucose. In contrast, EG2 was able to efficiently hydrolyze both substrates into glucose. The relative amount of each product was dependent on the particular endoglucanase, substrate, kinetic constants and reaction time. Thus, these tow enzymatic hydrolysis behaviours should provide two alternatives for biotechnological applications.
Graphical Abstract





Similar content being viewed by others
References
Bhat MK (2000) Cellulases and related enzymes in biotechnology. Biotechnol Adv 18:355–383
Himmel ME, Ding SY, Johnson DK, Adney WS, Nimlos MR, Brady JW, Foust TD (2007) Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 315:804–807
Berlin A, Balakshin M (2014) Industrial lignins, analysis, properties, and applications. In: Gupta VK et al (eds) Bioenergy research: advances and applications. Elsevier, Amsterdam, pp 315–336
Beckham GT, Bomble YJ, Bayer EA, Himmel ME, Crowley MF (2011) Applications of computational science for understanding enzymatic deconstruction of cellulose. Curr Opin Biotechnol 22:231–238
Gupta VK, Kubicek CP, Berrin JG, Wilson DW, Couturier M, Berlin A, Filho EXF, Ezeji T (2016) Fungal enzymes for bio-products from sustainable and waste biomass. Trends Biochem Sci 41:633–645
Nidetzky B, Steiner W, Hayn M, Claeyssens M (1994) Cellulose hydrolysis by the cellulases from Trichoderma reesei: a new model for synergistic interaction. Biochem J 298:705–710
Mood SH, Golfeshan AH, Tabatabaei M, Jouzani GS, Najafi GH, Gholami M, Ardjmand M (2013) Lignocellulosic biomass to bioethanol; a comprehensive review on pretreatment. Renew Sustain Energy Rev 27:77–93
Sindhu R, Binod R, Pandey A (2016) Biological pretreatment of lignocellulosic biomass—an overview. Bioresour Technol 199:76–82
Singh A, Bishnoi NR (2012) Enzymatic hydrolysis optimization of microwave alkali pretreated wheat straw and ethanol production by yeast. Bioresour Technol 108:94–101
Araujo R, Casal M, Cavaco-paulo A (2008) Application of enzymes for textile fibres processing. Biocatal Biotransform 26:332–349
Singhania RR, Saini JK, Saini R, Adsul M, Mathur A, Gupta R, Tuli DK (2014) Bioethanol production from wheat straw via enzymatic route employing Penicillium janthinellum cellulases. Bioresour Technol 169:490–495
Kleman-Leyer KM, Siika-Aho M, Teeri TT, Kirk TK (1996) The cellulases endoglucanase I and cellobiohydrolase II of Trichoderma reesei act synergistically to solubilize native cotton cellulose but not to decrease its molecular size. Appl Environ Microbiol 62:2883–2887
Hosseini SA, Shah N (2011) Modelling enzymatic hydrolysis of cellulose part I: population balance modelling of hydrolysis by endoglucanase. Biomass Bioenergy 35:3841–3885
Karmakar M, Ray RR (2011) Current trends in research and application of microbial cellulases. Res J Microbiol 6:41–53
Ben Hmad I, Gargouri A (2017) Neutral and alkaline cellulases: production, engineering, and applications. J Basic Microbiol 9999:1–6. https://doi.org/10.1002/jobm.201700111
Juturu V, Wu JC (2012) Microbial xylanases: engineering, production and applications. Biotechnol Adv 30:1219–1227
Zheng F, Ding S (2013) Processivity and enzymatic mode of a glycoside hydrolase family 5 endoglucanase from Volvariella volvacea. Appl Environ Microbiol 76:989–996.
Venditto I, Najmudin S, Luís AS, Ferreira LMA, Sakka K, Knox JP, Gilbert HJ, Fontes CM (2015) Family 46 carbohydrate-binding modules contribute to the enzymatic hydrolysis of xyloglucan and β-1,3–1,4-glucans through distinct mechanisms. J Biol Chem 290:10572–10586
Sahasrabudhe NM, Tian L, Van Den Berg M, Bruggeman G, Bruininx E, Schols HA, Faas MM, De Vos P (2016) Endo-glucanase digestion of oat β-glucan enhances dectin-1 activation in human dendritic cells. J Funct Foods 21:104–112
Kumar AK, Sharma S (2017) Recent updates on different methods of pretreatment of lignocellulosic feedstocks: a review. Bioresour Bioprocess 4:1–19
Payne Ch M, Knott BC, Mayes HB, Hansson H, Himmel ME, Sandgren M, Stahlberg J, Beckham GT (2015) Fungal cellulases. Chem Rev 115:1308–1448
Miettinen-Oinonen A, Londesborough J, Joutsjoki V, Lantto R, Vehmaanperä J, Biotecd PL (2004) Three cellulases from Melanocarpus albomyces for textile treatment at neutral pH. Enzym Microb Technol 34:332–341
Amouri B, Gargouri A (2006) Characterization of a novel β-glucosidase from a Stachybotrys strain. Biochem Eng J 32:191–197
Saibi W, Amouri B, Gargouri A (2007) Purification and biochemical characterization of a transglucosilating β-glucosidase of Stachybotrys strain. Appl Microbiol Biotechnol 77:293–300
Ben Hmad I, Abdeljalil S, Saibi W, Amouri B, Gargouri A (2014) Medium initial pH and carbon source stimulate differential alkaline cellulase time course production in Stachybotrys microspora. Appl Biochem Biotechnol 172:2640–2649
Ben Hmad I, Boudabbous M, Belghith H, Gargouri A (2017) A novel ionic liquid-stable halophilic endoglucanase from Stachybotrys microspora. Process Biochem 54:59–66
Ben Hmad I, Boudabbous M, Yaîch A, Rebai M, Gargouri A (2016) A novel, neutral, halophile Stachybotrys microspora-based endoglucanase active impact on β-glucan. Bioprocess Biosyst Eng 39:685–693
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
De Almeida MN, Falkoski DL, Guimarâes VM, Ramos HJO, Visser EM, Maitan-Alfenas GP, De Rezende ST (2013) Characteristics of free endoglucanase and glycosidases multienzyme complex from Fusarium verticillioides. Bioresour Technol 143:413–422
Zhao J, Shi P, Li Z, Yang P, Luo H, Bai V, Wang Y, Yao B (2012) Two neutral thermostable cellulases from Phialophora sp. G5 act synergistically in the hydrolysis of filter paper. Bioresour Technol 121:404–410
Miotto LS, Alves de Rezende C, Bernardes A, Serpa VI, Tsang A, Polikarpov I (2014) The characterization of the endoglucanase Cel12A from Gloeophyllum trabeum reveals an enzyme highly active on β-glucan. PLoS ONE 9:1–9
Sims RE, Mabee W, Saddler JN, Taylor M (2010) An overview of second generation biofuel technologies. Bioresour Technol 101:1570–1580
Annamalai N, Mayavan VR, Sivaramasamy E, Thangavel B (2013) Thermostable, haloalkaline cellulase from Bacillus halodurans CAS 1 by conversion of lignocellulosic wastes. Carbohydr Polym 94:409–415
Li X, Yu HY (2012) Purification and characterization of an organic-solvent-tolerant cellulase from a halotolerant isolate, Bacillus sp. L1. J Ind Microbiol Biotechnol 39:1117–1124
Kuhad RC, Gupta R, Singh A (2011) Microbial cellulases and their industrial applications. Enzym Res. https://doi.org/10.4061/2011/280696
Morrison JF (1982) The slow-binding and slow, tight-binding inhibition of enzyme-catalysed reactions. Trends Biochem Sci 7:102–105
Rahimizadeh P, Najavand S, Pazhang M (2015) A comparative study of activity and stability of the free and the immobilized endoglucanase from Alicyclobacillus acidocaldarius. Biomacromol J 1:167–176
Park T, Seo S, Shin T, Cho BW, Cho S, Kim B, Lee S, Ha JK, Seo J (2018) Molecular cloning, purification, expression, and characterization of β-1,4-endoglucanase gene (Cel5A) from Eubacterium cellulosolvens sp. isolated from Holstein steers’ rumen. Asian-Australas J Anim Sci 31:607–615
Várnai A, Siika-aho M, Viikari L (2013) Carbohydrate-binding modules (CBMs) revisited: reduced amount of water counterbalances the need for CBMs. Biotechnol Biofuels 6:1–30
Palonen H, Tjerneld F, Zacchi G, Tenkanen M (2004) Adsorption of Trichoderma reesei CBH I and EG II and their catalytic domains on steam pretreated softwood and isolated lignin. J Biotechnol 107:65–72
Carthy TM, Hanniffy O, Lalor E, Savage AV, Tuohy MG (2005) Evaluation of three thermostable fungal endo-β-glucanases from Talaromyces emersonii for brewing and food applications. Process Biochem 40:1741–1748
Karnchanatat A, Petsoma A, Sangvanich P, Piapukiew J, Whalley AJS, Reynolds CD, Gadd GM, Sihanonth P (2008) A novel thermostable endoglucanase from the wood-decaying fungus Daldinia eschscholzii (Ehrenb.:Fr.) Rehm. Enzym Microb Technol 42:404–413
Gupta V, Prasanna R, Chaudhary V, Nain L (2012) Biochemical, structural and functional characterization of two novel antifungal endoglucanases from Anabaena laxa. Biocatal Agric Biotechnol 1:338–347
Picart P, Goedegebuur F, Diaz P, Pastor FIJ (2012) Expression of novel β-glucanase Cel12A from Stachybotrys atra in bacterial and fungal hosts. Fungal Biol 116:443–451
Karlsson J, Siika-aho M, Tenkanen M, Tjerneld F (2002) Enzymatic properties of the low molecular mass endoglucanases Cel12A (EG III) and Cel45A (EG V) of Trichoderma reesei. J Biotechnol 99:63–78
Rye CS, Withers SG (2000) Glycosidase mechanisms. Curr Opin Chem Biol 4:573–580
Fierobe HP, Bagnara-Tardif C, Gaudin C, Guerlesquin F, Sauve P, Belaich A, Belaich JP (1993) Purification and characterization of endoglucanase C from Clostridium cellulolyticum. Catalytic comparison with endoglucanase A. Eur J Biochem 217:557–565
Parry NJ, Beever DE, Owen E, Nerinckx W, Claeyssens M, Van Beeumen J, Bhat MK (2002) Biochemical characterization and mode of action of a thermostable endoglucanase purified from Thermoascus aurantiacus. Arch Biochem Biophys 404:243–253
Ghatge SS, Telke AA, Kang SH, Arulalapperumal V, Lee KW, Govindwar SP, Um Y, Oh DB, Shin HD, Kim SW (2014) Characterization of modular bifunctional processive endoglucanase Cel5 from Hahella chejuensis KCTC 2396. Appl Microbiol Biotechnol 98:4421–4435
Tamura K, Hemsworth GR, De´ jean G, Rogers TE, Pudlo NA, Urs K, Jain N, Davies GJ, Martens EC, Brumer H (2017) Molecular mechanism by which prominent human gut bacteroidetes utilize mixed-linkage beta-glucans, major health-promoting cereal polysaccharides. Cell Rep 21:417–430
Leentjens J, Quintin J, Gerretsen J, Kox M, Pickkers P, Netea MG (2014) The effects of orally administered beta-glucan on innate immune responses in humans, a randomized open-label intervention pilot-study. PLoS ONE 9:1–6
Acknowledgements
The authors of this research work would like to thank Mrs. Fatma Rezgui and Mr. Kamel Walha for their technical help in the HPLC analysis. This study was conducted thanks to grants supported by the Ministry of Higher Education and Scientific Research, Tunisia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflict of interest.
Rights and permissions
About this article
Cite this article
Ben Hmad, I., Gargouri, A. Two Distinct Hydrolysis Mechanisms of the Neutral Endoglucanases EG1 and EG2 of Stachybotrys microspora. Catal Lett 148, 2190–2199 (2018). https://doi.org/10.1007/s10562-018-2410-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-018-2410-8