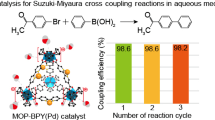
Abstract
The establishment of structure–property relationships in heterogeneous catalysis is of prime importance but remains a formidable challenge. Metal–organic frameworks (MOFs) featuring excellent chemical tunability are emerging as an auspicious platform for the atomic-level control of heterogeneous catalysis. Herein, we encapsulate palladium nanoparticles (Pd NPs) in a series of isoreticular mixed-linker MOFs, and the obtained MOF-Pd NPs catalysts were used to unveil the electronic and steric effects of linker substitution on the activity of these catalysts in the Suzuki–Miyaura cross-coupling reactions. Significantly, m-6,6′-Me2bpy-MOF-Pd exhibits a remarkable enhancement in the activity compared to non-functionalized m-bpy-MOF-Pd and m-4,4′-Me2bpy-MOF-Pd. This study unambiguously demonstrates that the stereoelectronic properties of linker units are crucial to the catalytic activity of nanoparticles encapsulated in MOFs. More interestingly, the trend of activity change is consistent with our previous work on catalytic sites generated in situ from Pd(II) coordinated in MOFs bearing the same functional groups, which suggests that both MOF-Pd NPs and MOF-Pd(II) catalysts generate similar active centers during Suzuki–Miyaura coupling reactions. This work paves a new avenue to the fabrication of advanced and tunable MOF-based catalysts through rational linker engineering.
Graphical Abstract
We encapsulate palladium nanoparticles in a series of isoreticular mixed-linker MOFs, and the obtained Pd-doped MOFs catalysts were used to unveil the electronic and steric effects of linker substitution on the activity of these catalysts in the context of Suzuki–Miyaura cross-coupling reactions. Impressively, m-6,6′-Me2bpy-MOF-Pd exhibits a remarkable enhancement in the activity compared to non-functionalized m-bpy-MOF-Pd and m-4,4′-Me2bpy-MOF-Pd, thus implementing atomic-level controls of heterogeneous catalysis.





Similar content being viewed by others
References
Furukawa H, Cordova KE, O’Keeffe M, Yaghi OM (2013) Science 341:1230444
Chughtai AH, Ahmad N, Younus HA, Laypkov A, Verpoort F (2015) Chem Soc Rev 44:6804–6849
Dhakshinamoorthy A, Garcia H (2012) Chem Soc Rev 41:5262–5284
Kim M, Cahill JF, Fei HH, Prather KA, Cohen SM (2012) J Am Chem Soc 134:18082–18088
Hendon CH, Tiana D, Fontecave M, Sanchez C, D’arras L, Sassoye C, Rozes L, Mellot-Draznieks C, Walsh A (2013) J Am Chem Soc 135:10942–10945
Burtch NC, Jasuja H, Walton KS (2014) Chem Rev 114:10575–10612
Bauer CA, Timofeeva TV, Settersten TB, Patterson BD, Liu VH, Simmons BA, Allendorf MD (2007) J Am Chem Soc 129:7136–7144
Tan JC, Cheetham AK (2011) Chem Soc Rev 40:1059–1080
Vermoortele F, Vandichel M, Van de Voorde B, Ameloot R, Waroquier M, Van Speybroeck V, De Vos DE (2012) Angew Chem Int Ed 51:4887–4890
Goh TW, Xiao C, Maligal-Ganesh RV, Li XL, Huang WY (2015) Chem Eng Sci 124:45–51
Li XL, Goh TW, Li L, Xiao CX, Guo ZY, Zeng XC, Huang WY (2016) ACS Catal 6:3461–3468
Choi KM, Na K, Somorjai GA, Yaghi OM (2015) J Am Chem Soc 137:7810–7816
Kotha S, Lahiri K, Kashinath D (2002)Tetrahedron 58:9633–9695
Maegawa T, Kitamura Y, Sako S, Udzu T, Sakurai A, Tanaka A, Kobayashi Y, Endo K, Bora U, Kurita T (2007) Chem Eur J 13:5937–5943
Artok L, Bulut H (2004) Tetrahedron Lett 45:3881–3884
Sahu D, Das P (2015) RSC Adv 5:3512–3520
Sydnes MO (2017) Catalysts 7:35–49
Yamamoto SI, Kinoshita H, Hashimoto H, Nishina Y (2014)Nanoscale 6:6501–6505
Hardy JJE, Hubert S, Macquarrie DJ, Wilson AJ (2004) Green Chem 6:53–56
Chen LY, Rangan S, Li J, Jiang HF, Li YW (2014) Green Chem 16:3978–3985
Chen LY, Gao Z, Li YW (2015) Catal Today 245:122–128
Pascanu V, Yao Q, Bermejo Gómez A, Gustafsson M, Yun Y, Wan W, Samain L, Zou XD, Martín-Matute B (2013) Chem Eur J 19:17483–17493
Yuan BZ, Pan YY, Li YW, Yin B, Jiang HF (2010) Angew Chem Int Ed 49:4054–4058
Carson F, Pascanu V, Bermejo Gómez A, Zhang Y, Platero -Prats AE, Zou XD, Martín-Matute B (2015) Chem Eur J 21:10896–10902
Saha D, Sen R, Maity T, Koner S (2013) Langmuir 29:3140–3151
Pascanu V, Hansen PR, Bermejo Gómez A, Ayats C, Platero-Prats AE, Johansson MJ, Pericàs M, Martín-Matute B (2015) ChemSusChem 8:123–130
Manna K, Zhang T, Lin WB (2014) J Am Chem Soc 136:6566–6569
Lim D, Yoon J, Ryu K, Suh M (2012) Angew Chem Int Ed 124:9952–9955
Chen LY, Chen HR, Luque R, Li YW (2014) Chem Sci 5:3708–3714
Li XL, Guo ZY, Xiao CX, Goh TW, Tesfagaber D, Huang WY (2014) ACS Catal 4:3490–3497
Li XL, Van Zeeland R, Maligal-Ganesh RV, Pei YC, Power G, Stanley L, Huang WY (2016) ACS Catal 6:6324–6328
Phan NTS, Van Der Sluys M, Jones CW (2006) Adv Synth Catal 348:609–679
Elias WC, Signori AM, Zaramello L, Albuquerque BL, de Oliveira DC, Domingos JB (2017) ACS Catal 7:1462–1469
Balanta A, Godard C, Claver C (2011) Chem Soc Rev 40:4973–4985
Astruc D (2007) Inorg Chem 46:1884–1894
Morabito J, Chou LY, Li Z, Manna C, Petroff C, Kyada R, Palomba J, Byers J, Tsung CK (2014) J Am Chem Soc 136:12540–12543
Sanhes D, Raluy E, Retory S, Saffon N, Teuma E, Gomez M (2010) Dalton Trans 39:9719–9726
Pacardo DB, Slocik JM, Kirk KC, Naik R, Knecht MR (2011) Nanoscale 3:2194–2201
Acknowledgements
This work is partially supported by NSF Grant CHE-1566445. We also thank Ames Laboratory (Royalty Account) and Iowa State University for startup funds. The Ames Laboratory is operated for the U.S. Department of Energy by Iowa State University under Contract No. DE-AC02-07CH11358. We thank Gordon J. Miller for the use of PXRD in his group.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, X., Zhang, B., Van Zeeland, R. et al. Unveiling the Effects of Linker Substitution in Suzuki Coupling with Palladium Nanoparticles in Metal–Organic Frameworks. Catal Lett 148, 940–945 (2018). https://doi.org/10.1007/s10562-017-2289-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-017-2289-9