Abstract
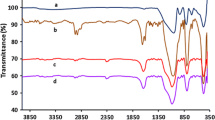
In this work, a hydroxyl functionalized Lewis acidic ionic liquid on silica is found to be an efficient heterogeneous catalyst for the C-3 Friedel-Crafts benzylation of indoles with different benzyl alcohols as benzylation agents under mild conditions. This catalytic system benefits from simple operation, work-up and reutilization procedures, wide substrate tolerance, low cost, high catalytic activity and excellent chemo-selectivity. The hybrid combination of hydroxyl functionalized ionic liquid and silica through the formation of hydrogen bond endows this catalyst with high stability in the Friedel-Crafts reaction system, and the catalyst could be easily recycled and reutilized.
Graphical Abstract
A hydroxyl functionalized Lewis acid ionic liquid supported on silica was found to be an efficient catalyst for the C-3 Friedel-Crafts benzylation reaction of iodole with alcohols. This catalyst benefits from low cost, feasible preparation and reutilization, high catalytic activity and selectivity, and the operation and work-up procedures are very simple.





Similar content being viewed by others
References
Sundberg RJ (1970) The chemistry of indoles, 189. Academic Press, New York, p 176–182
Shiri M, Zolfigol MA, Kruger GH, Tanbakouchian Z (2010) Chem Rev 110:2250–2293
Bandini M, Eichholzer A (2009) Angew Chem Int Ed 48:9608–9644
Sunaba H, Kamata K, Mizuno N 2014 Chemcatchem 6:2333–2338
Wang Z-X, Kochanowska-Karamyan AJ, Hamann MT (2010) Chem Rev 110:4489–4497
Contractor R, Samudio IJ, Estrov Z, Harris D, McCubrey JA, Safe SH, Andreeff M, Konopleva M (2005) Cancer Res 65:2890–2898
Wu X, See JWT, Xu K, Hirao H, Roger J, Hierso JC, Zhou JS (2014) Angew Chem Int Ed 53:13573–13577
Khalil A, Fihri A, Jouiad M, Hashaikeh R (2014) Tetrahedron Lett 55:5973–5975
Lewis JC, Bergman RG, Ellman JA (2008) Acc Chem Res 41:1013–1025
Hamid MHSA, Allen CL, Lamb GW, Maxwell AC, Maytum HC, Watson AJA, Williams JMJ (2009) J Am Chem Soc 131:1766–1774
Dobereiner GE, Crabtree RH (2010) Chem Rev 110:681–703
Pingen D, MKller C, Vogt D (2010) Angew Chem Int Ed 49:8130–8133
Imm S, Bahn S, Neubert L (2010) Angew Chem Int Ed 49:8126–8129
Savara A, Chan-Thaw CE, Rossetti I, Villa A, Prati L (2014) Chemcatchem 6:3464–3473
Koppolu SR, Naveen N, Balamurugan R (2014) J Org Chem 79:6069–6078
Huo C, Sun C, Wang C, Jia X, Chang W (2013) ACS Sustainable Chem Eng 1:549–553
Naredla RR, Klumpp DA (2013) Chem Rev 113:6905–6948
Baeza A, Najera C (2014) Synthesis 46:25–34
Kumar R, Eycken EV (2013) Chem Soc Rev 42:1121–1146
Bandini M, Bottoni A, Chiarucci M, Cera G, Miscione GP (2012) J Am Chem Soc 134:20690–20700
Bandini M, Eichholzer A (2009) Angew Chem Int Ed 48:9533–9537
Hikawa H, Suzuki H, Azumaya I (2013) J Org Chem 78:12128–12135
Yasuda M, Somyo T, Baba A (2006) Angew Chem Int Ed 45:793–796
Fan GP, Liu Z, Wang GW (2013) Green Chem 15:1659–1664
Jana U, Maiti S, Biswas S (2007) Tetrahedron Lett 48:7160–7163
Das D, Roy S (2013) Adv Synth Catal 355:1308–1314
Hikawa H, Suzuki H, Yokoyama Y, Azumaya I (2013) Catalysts 3:486–500
Hikawa H, Yokoyama Y (2013) RSC Adv 3:1061–1064
Kimura M, Futamata M, Mukai R, Tamaru Y (2005) J Am Chem Soc 127:4592–4593
Siddiki SMAH, Kon K, Shimizu K (2013) Chem Eur J 19:14416–14419
Tao YS, Wang BM, Zhao JF, Song YM, Qu LH, Qu JP (2012) J Org Chem 77:2942–2946
Zaitsev AB, Gruber S, Pregosin PS (2007) Chem Commun 44:4692–4693
Zaitsev AB, Gruber S, Pluss PA, Pregosin PS, Veiros LF, Worle M (2008) J Am Chem Soc 130:11604–11605
Baehn S, Imm S, Mevius K (2010) Chem Eur J 16:3590–3593
Han F, Yang L, Li Z, Xia CG (2012) Adv Synth Catal 354:1052–1060
Minakata S, Komatsu M (2009) Chem Rev 109:711–724
Mehnert CP, Cook RA, Dispenziere NC, Afeworki M (2002) J Am Chem Soc 124:12932–12933
Huang J, Jiang T, Gao HX, Han BX, Wu WZ, Chang YH, Zhao GY (2004) Angew Chem Int Ed 43:1397–1399
Dragoni D, Manini N, Ballone P (2012) Chemphyschem 13:1772–1780
Perkin S (2012) Phys Chem Chem Phys 14:5052–5062
Ballone P, Del Popolo MG, Vovio S, Podesta A, Milani P, Manini N (2012) Phys Chem Chem Phys 14:2475–2482
Chrobok A, Baj S, Pudlo W, Jarzebski A (2010) Appl Catal A 389:179–185
Zhang P, Wu TB, Han BX (2014) Adv Mater 26:6810–6827
Jakuttis M, Schönweiz A, Werner S, Franke R, Wiese KD, Haumann M, Wasserscheid P (2011) Angew Chem Int Ed 50:4492–4495
Fehrmann R, Riisager A, Haumann M (2014) Supported ionic liquids. Wiley, Weinheim, p 1-496
Aprile C, Giacalone F, Agrigento P, Liotta LF, Martens JA, Pescarmona PP, Gruttadauria M (2011) Chemsuschem 4:1830–1837
Meyer C, Hager V, Schwieger W, Wasserscheid P (2012) J Catal 292:157–165
Zhu AL, Li LJ, Wang JJ, Zhuo KL (2011) Green Chem 13:1244–1250
Doetterl M, Alt HG (2011) Chemcatchem 3:1799–1804
Deshmukh KM, Qureshi ZS, Dhake KP, Bhanage BM (2010) Catal Commun 12:207–211
Zimanis A, Katkevica S, Mekss P (2009) Catal Commun 10:614–619
Hu SQ, Zhang ZF, Song JL, Zhou YX, Han BX (2009) Green Chem 11:1746–1749
Morales RC, Tambyrajah V, Jenkins PR, Davies DL, Abbott AP (2004) Chem Commun 2:158–159
McClelland RA, Kanagasabapathy VM, Steenken S (1988) J Am Chem Soc 110:6913–6914
Avalos M, Babiano R, Cintas P, Jimenez JL, Palacios JC (2006) Angew Chem Int Ed 45:3904–3908
Hertl W, Hair ML (1968) J Phys Chem 72:4676–4682
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21373079) and Program for Science & Technology Innovation Talents in Universities of Henan Province (14HASTIT015).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhu, A., Feng, W., Li, L. et al. Hydroxyl Functionalized Lewis Acidic Ionic Liquid on Silica: An Efficient Catalyst for the C-3 Friedel-Crafts Benzylation of Indoles with Benzyl Alcohols. Catal Lett 147, 261–268 (2017). https://doi.org/10.1007/s10562-016-1918-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-016-1918-z