Abstract
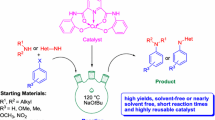
5-(N,N-bis-3-(triethoxysilyl) propyl) ureyl-1,10-phenanthroline was prepared and successively grafted onto mesoporous silica of SBA-15 to get SBA-15 anchored ligand which was then reacted with Cu(I) to get SBA-15 anchored complex as the heterogeneous copper catalyst. This catalyst was characterized by FT-IR, XRD, CHN, AAS, TGA, EDAX, BET, SEM and TEM. The activities of the catalyst were tested in C–N cross-coupling of amides and N–H heterocycle compounds with aryl halides. The catalyst showed high catalytic activities for these cross-coupling reactions providing good to excellent yields of desired products. Moreover, the catalyst can be easily recovered by simple filtration and reused several times without significant loss of its catalytic activity.
Graphical Abstract












Similar content being viewed by others
References
Fagan PJ, Hauptman E, Shapiro R, Casalnuovo A (2000) J Am Chem Soc 122:5043–5051
Montalbetti CAG, Falque NV (2005) Tetrahedron 61:10827–10842
Huang ZB, Kim SK, Chang SH (2006) Synlett 17:1707–1710
Monnier F, Taillefer M (2009) Angew Chem Int Ed 48:6954–6971
Goldberg I (1906) Ber Dtsch Chem Ges 39:1691–1692
Shakespeare WC (1999) Tetrahedron Lett 40:2035–2038
Yin J, Buchwald SL (2000) Org Lett 2:1101–1104
Ullmann F (1903) Ber Dtsch Chem Ges 36:2382–2384
Arterburn JB, Pannala M, Gonzalez AM (2001) Tetrahedron Lett 42:1475
Lang F, Zewge D, Houpis IN, Volante RP (2001) Tetrahedron Lett 42:3251–3254
Lindley J (1984) Tetrahedron 40:1433–1456
Sawyer JS (2000) Tetrahedron 56:5045–5065
Hosseinzadeh R, Tajbakhsh M, Mohadjerani M, Mehdinejad H (2004) Synlett 15:1517–1520
Hosseinzadeh R, Golchoubian H, Masoudi M (2008) J Chin Chem Soc 55:649–653
Hosseinzadeh R, Tajbakhsh M, Mohadjerani M, Ghorbani E (2008) Chin J Chem 26:2120–2124
Wang C, Liu L, Wang W, Ma DS, Zhang H (2010) Molecules 15:1154–1160
Jiang L (2014) Molecules 19:13448–13460
Hosseinzadeh R, Tajbakhsh M, Alikarami M (2006) Synlett 17:2124–2126
Hosseinzadeh R, Tajbakhsh M, Alikarami M (2006) Tetrahedron Lett 47:5203–5205
Hosseinzadeh R, Tajbakhsh M, Alikarami M, Mohadjerani M (2008) J Heterocyclic Chem 45:1815–1818
Altman RA, Koval ED, Buchwald SL (2007) J Org Chem 72:6190–6199
Verma AK, Singh J, Sankar VK, Chaudhary R, Chandra R (2007) Tetrahedron Lett 48:4207–4210
Mao J, Guo J, Song H, Ji SJ (2008) Tetrahedron 64:1383–1387
Choudary BM, Sridhar C, Kantam ML, Venkanna GT, Sreedhar B (2005) J Am Chem Soc 127:9948–9949
Kantam ML, Rao BPC, Choudary BM, Reddy RS (2006) Synlett 17:2195–2198
Reddy KR, Kumar NS, Sreedhar B, Kantam ML (2006) J Mol Cat A: Chem 252:136–141
Likhar PR, Roy S, Roy M, Kantam ML, De RL (2007) J Mol Catal A: Chem 271:57–62
Gopiramana M, Babu SG, Khatri Z, Kai W, Kim YA, Endo M, Karvembu R, Kim IS (2013) Carbon 62:135–148
Rout L, Jammi S, Punniyamurthy T (2007) Org Lett 9:3397–3399
Jammi S, Sakthivel S, Rout L, Mukherjee T, Mandal S, Mitra R, Saha P, Punniyamurthy T (2009) J Org Chem 74:1971–1976
Babu SG, Karvembu R (2011) Ing Eng Chem Res 50:9594–9600
Kantama ML, Roy M, Roy S, Sreedhar B, De RL (2008) Catal Commun 9:2226–2230
Huang YZ, Miao H, Zhang QH, Chen C, Xu J (2008) Catal Lett 122:344–348
Jammi S, Krishnamoorthy S, Saha P, Kundu DS, Sakthivel S, Ali MA, Paul R, Punniyamurthy T (2009) Synlett 20:3323–3327
Sreedhar B, Arundhathi R, Reddy PL, Reddy MA, Kantam ML (2009) Synthesis 41:2517–2522
Son SU, Park IK, Park J, Hyeon T (2004) Chem Commun 40:778–779
Yao Z, Wei X (2010) Chin J Chem 28:2260–2268
Islam M, Mondal S, Mondal P, Roy AS, Tuhina K, Salam N, Paul S, Hossain D, Mobarok M (2011) Transit Met Chem 36:447–458
Islam M, Mondal S, Mondal P, Roy AS, Tuhina K, Mobarok M, Paul S, Salam N, Hossain D (2011) Catal Lett 141:1171–1181
Xiao R, Zhao H, Cai M (2013) Tetrahedron 69:5444–5450
Maity T, Saha D, Koner S (2014) ChemCatChem 6:2373–2383
Kumar A, Bishnoi AK (2014) RSC Adv 4:41631–41635
Neouze MA (2010) J Mater Chem 20:9593–9607
Sauvage JP (1990) Acc Chem Res 23:319–327
Binnemans K, Lenaerts P, Driesen K, Gorller-Walrand C (2004) J Mater Chem 14:191–195
Zhao DY, Feng JL, Huo QS, Melosh N, Fredrickson GH, Chmelka BF, Stucky GD (1998) Science 279:548–552
Beck JS, Vartuli JC, Roth WJ, Leonowics ME, Kresge CT, Schmitt KD, Chu CTW, Olson DH, Sheppard EW, Mccullen SB, Higgins JB, Schlenker JL (1992) J Am Chem Soc 114:10834–10843
Brunauer S (1938) mmett E P, Teller E. J Am Chem Soc 60:309–319
Barrett EP, Joyner LG, Halenda PP (1951) J Am Chem Soc 73:373–380
Klapars A, Huang X, Buchwald SL (2002) J Am Chem Soc 124:7421–7428
Wang J, Yin X, Wu J, Wu D, Pan Y (2013) Tetrahedron 69:10463–10469
Quan ZJ, Xia HD, Zhang Z, Da YX, Wang XC (2014) Appl Organometal Chem 28:81–85
Huang M, Lin X, Zhu X, Peng W, Xie J, Wan Y (2011) Eur J Org Chem 2011:4523–4527
Jha RR, Singh J, Tiwari RK, Verma AK (2013) ARKIVOC(ii) 2013:228–248
Murthy SN, Madhav B, Reddy VP, Nageswar YVD (2010) Adv Synth Catal 352:3241–3245
Yang H, Xi C, Miao Z, Chen R (2011) Eur J Org Chem 2011:3353–3360
Wang YL, Luo J, Liu ZL (2013) J Chin Chem Soc 60:1–7
Lempers HEB, Sheldon RA (1998) J Catal 175:62–69
Cheraghali R, Tavakoli H, Sepehrian H (2013) Sci Iran 20:1028–1034
Acknowledgments
Financial support of this work from the Research Council of the University of Mazandaran is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hosseinzadeh, R., Aghili, N. & Tajbakhsh, M. SBA-15 Immobilized Phenanthroline–Copper(I) Complex as a Recyclable Efficient Catalyst for N-Arylation of Amides and N–H Heterocycles with Aryl Halides. Catal Lett 146, 193–203 (2016). https://doi.org/10.1007/s10562-015-1622-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-015-1622-4