Abstract
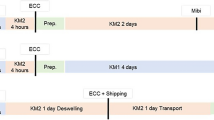
To assess corneal endothelial cell (CEC) quantity and quality in eye-bank prepared lamellar grafts for Descemet Membrane Endothelial Keratoplasty (DMEK) from organ cultured donor corneas that have not undergone de-swelling by media supplementation with dextran. Prior to graft preparation, corneas that had not undergone de-swelling (n = 30) were placed into fresh storage medium without dextran (KMI). Corneas in the control group (n = 30) were placed in dehydration medium containing 5% dextran (KMII). Subtotal stripping of Descemet’s membrane (DM) was performed manually. Following graft preparation, 10 corneas of each group were cultured further in their respective medium for 24 h, 72 h, or 120 h, respectively. Before and after DM stripping, as well as at the end of culture, CEC numbers were obtained. At the end of culture, CEC morphology was graded using a scoring system and CEC metabolism was assessed by detection of adenosine triphosphate. At 24 h after DM stripping, mean CEC counts (in cells/mm2) were 2204 in corneas stored in KMII, and 2391 in corneas stored in KMI (p = 0.003). This corresponds to a mean relative CEC loss of 12.4% with dextran versus 9.7% without dextran (p = 0.04). At 72 h, CEC counts were 1946 in KMII, and 2289 in KMI (p = 0.004). This corresponds to CEC loss of 23% with dextran versus 14% without dextran (p = 0.009). At 120 h, CEC counts were 2047 in KMII, and 2230 in KMI (p = 0.14). This corresponds to CEC loss of 22.7% with dextran versus 17.2% without dextran (p = 0.14). Also, at 120 h after DM stripping, 6/10 corneas fell below a threshold of 2000 cells/mm2 if stored in medium containing dextran, versus 1/9 corneas if stored without dextran (p = 0.003). Morphological assessment of CEC quality revealed equal scores for cell polymorphism (median = 1), granulation (median = 0) and segmentation (median = 1) in all groups. Lower ATP/protein ratios were observed in corneas stored in medium without dextran at 24 h (p < 0.001), 72 h (p < 0.001), and 120 h (p = 0.02). Abandoning the use of dextran in corneas destined for DMEK surgery leads to increased CEC counts and thereby serves to reduce tissue loss.






Similar content being viewed by others
References
Abdin A, Daas L, Pattmöller M, Suffo S, Langenbucher A, Seitz S (2018) Negative impact of dextran in organ culture media for pre-stripped tissue preservation on DMEK (Descemet membrane endothelial keratoplasty) outcome. Graefes Arch Clin Exp Ophthalmol 256:2135–2142
Armitage WJ, Easty DL (1997) Factors influencing the suitability of organ-cultured corneas for transplantation. Invest Ophthalmol Vis Sci 38:16–24
Bhogal M, Matter K, Balda MS, Allan B (2016) Organ culture storage of pre-prepared corneal donor material for Descemet’s membrane endothelial keratoplasty. Br J Ophthalmol 100:1576–1583
Borderie VM, Baudrimont M, Lopez M et al (1997) Evaluation of the deswelling period in dextran-containing medium after corneal organ culture. Cornea 16:215–223
Borderie VM, Scheer S, Touzeau O et al (1998) Donor organ cultured corneal tissue selection before penetrating keratoplasty. Br J Ophthalmol 82:382–388
Bundesärztekammer (2018) Richtlinien zum Führen einer Hornhautbank. Dtsch Arztebl 115:262
Heinzelmann S, Böhringer D, Eberwein P et al (2017) Graft dislocation and graft failure following Descemet membrane endothelial keratoplasty (DMEK) using precut tissue: a retrospective cohort study. Graefes Arch Clin Exp Ophthalmol 255:127–133
Hermel M, Salla S, Fuest M, Walter P (2016) The role of corneal endothelial morphology in graft assessment and prediction of endothelial cell loss during organ culture of human donor corneas. Acta Ophthalmol 95:205–210
Kruse FE, Schrehardt US, Tourtas T (2014) Optimizing outcomes with Descemet’s membrane endothelial keratoplasty. Curr Opin Ophthalmol 25:325–334
Menzel-Severing J, Kruse FE, Tourtas T (2017) Organ-cultured, prestripped donor tissue for DMEK surgery: clinical outcomes. Br J Ophthalmol 101:1124–1127
Nakagawa H, Inatomi T, Hieda O et al (2014) Clinical outcomes in Descemet stripping automated endothelial keratoplasty with internationally shipped precut donor corneas. Am J Ophthalmol 157:50–55.e1
Palioura S, Colby K (2017) Outcomes of Descemet stripping endothelial keratoplasty using eye bank-prepared preloaded grafts. Cornea 36:21–25
Parekh M, Ruzza A, Ferrari S, Ponzin D (2016) Preservation of preloaded DMEK lenticules in dextran and non-dextran-based organ culture medium. J Ophthalmol 2016:5830835
Petty RD, Sutherland LA, Hunter EM, Cree IA (1995) Comparison of MTT and ATP-based assays for the measurement of viable cell number. J Biolumin Chemilumin 10:29–34
Ragunathan S, Ivarsen A, Nielsen K, Hjortdal J (2014) Comparison of organ cultured precut corneas versus surgeon-cut corneas for Descemet’s stripping automated endothelial keratoplasty. Cell Tissue Bank 15:573–578
Redbrake C, Salla S, Nilius R et al (1997) A histochemical study of the distribution of dextran 500 in human corneas during organ culture. Curr Eye Res 16:405–411
Romano V, Parekh M, Ruzza A et al (2018) Comparison of preservation and transportation protocols for preloaded Descemet membrane endothelial keratoplasty. Br J Ophthalmol 102:549–555
Salla S, Redbrake C, Becker J, Reim M (1995) Remarks on the vitality of the human cornea after organ culture. Cornea 14:502–508
Schlögl A, Tourtas T, Kruse FE, Weller JM (2016) Long-term clinical outcome after Descemet membrane endothelial keratoplasty. Am J Ophthalmol 169:218–226
Yoeruek E, Hofmann J, Bartz-Schmidt KU (2013) Comparison of swollen and dextran deswollen organ-cultured corneas for Descemet membrane dissection preparation: histological and ultrastructural findings. Invest Ophthalmol Vis Sci 54:8036–8040
Zhu L, Zha Y, Cai J, Zhang Y (2018) Descemet stripping automated endothelial keratoplasty versus descemet membrane endothelial keratoplasty: a meta-analysis. Int Ophthalmol 38:897–905
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors. However, this article does contain studies using human tissue. Informed consent was obtained from the relatives of all post-mortem tissue donors included in the study. All study procedures were performed in accordance with the principles laid down in the Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Salla, S., Kruse, F.E., Walter, P. et al. Supplementation of organ culture medium with dextran is not required in pre-stripped human donor tissue for DMEK surgery. Cell Tissue Bank 20, 193–200 (2019). https://doi.org/10.1007/s10561-019-09757-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-019-09757-8