Abstract
Purpose
Although a number of studies involving small-vessel de novo coronary disease showed clinical benefits of drug-coated balloons (DCB), the role of DCB in large vessel lesions is still unclear.
Methods
We searched main electronic databases for randomized controlled trials (RCTs) comparing DCB with stents for large vessel de novo coronary artery disease. The primary endpoint was major cardiovascular adverse events (MACE), composite cardiovascular death (CD), myocardial infarction (MI), or target lesion revascularization (TLR).
Results
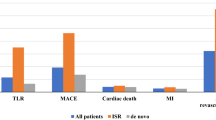
This study included 7 RCTs with 770 participants. DCB were associated with a marked risk reduction in MACE [Risk Ratio (RR): 0.48; 95% confidence interval [CI]: 0.24 to 0.97; P = 0.04], TLR (RR: 0.53; 95% CI: 0.25 to 1.14; P = 0.10), and late lumen loss [standard mean difference (SMD): -0.57; 95% CI: -1.09 to -0.05; P = 0.03] as compared with stents. There is no significant difference in MI (RR: 0.58; 95% CI: 0.21 to 1.54; P = 0.27), CD (RR: 0.33; 95% CI: 0.06 to 1.78; P = 0.19), and minimal lumen diameter (SMD: -0.34; 95% CI: -0.72 to 0.05; P = 0.08) between groups. In subgroup analyses, the risk reduction of MACE persisted in patients with chronic coronary syndrome (RR: 0.25; 95% CI: 0.07 to 0.89; P = 0.03), and patients receiving DCB vs. bare metal stent (RR: 0.19; 95% CI: 0.05 to 0.73; P = 0.01). In addition, there was no significant difference between the DCB group and the drug eluting stent group for MACE (RR: 0.69; 95% CI: 0.30 to 1.60; P = 0.38).
Conclusion
DCB may be an effective therapeutic option in patients with large vessel de novo coronary artery disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coronary artery disease (CAD) is still a major cause of mortality despite major improvements in primary and secondary prevention strategies [1, 2]. Drug-coated balloons (DCB) are a novel treatment strategy for CAD, based on combination therapy of balloon and drug, dilating narrowed coronary lesions and achieving lower rates of restenosis by leaving no metal behind [3]. In fact, the clinical efficacy of DCB has been confirmed in small-vessel disease and in-stent restenosis [4, 5]. However, the role of DCB in large vessel de novo coronary artery disease remains uncertain.
DCB have been investigated for more than ten years in cardiovascular disease [6]. The technique of DCB is direct contact of the antiproliferative drug with the vessel wall via a semicompliant balloon, which could inhibit the proliferation of smooth muscle cells. Compared with stents, DCB have several advantages [7, 8]: 1) DCB may be used in subsets of lesions where stents cannot be delivered or where stents do not perform well, such as in torturous vessels, long diffuse calcified lesions, or bifurcated lesions; 2) The DCB angioplasty requires a short dual antiplatelet therapy duration of only 4 weeks and 3) absence of a stent allows the artery’s original anatomy to remain intact, thereby diminishing abnormal flow patterns.
Recently, several studies comparing DCB with stents in large vessel lesions have been reported but no updated meta-analyses are available [9, 10]. In addition, the higher risk of cardiovascular events in DCB for patients with acute coronary syndrome (ACS) has raised concern in two observational studies [11, 12]. It is still unknown whether this concern extends to randomized controlled trials (RCT). Two previous meta-analyses were performed in large vessels, but data relating to the risk of clinical outcomes are scant [13, 14]. Moreover, these meta-analyses included only a small number of studies and included observational studies, which are prone to ascertainment and selection biases.
The aim of this meta-analysis was to compare efficacy and safety between patients undergoing DCB vs. stents for large de novo coronary lesions in RCT. In addition, to explore a target population that could benefit the most from DCB strategy, subgroup analyses were conducted according to the type of CAD [chronic coronary syndrome (CCS) vs. ACS], and the type of stents [drug eluting stent (DES) or bare metal stent (BMS)].
Methods
Search for Relevant Research
A systematic literature search was performed to identify randomized clinical trials (RCTs) that compared DCB with stents in patients with CAD and published between January 2000 and November 2022 in English. Two independent reviewers (Ma and Liu) searched databases, including Cochrane Library, Pubmed, and Embase. Disagreements were discussed with another author (Li) and resolved by consensus. The search strategy included both Medical Subject Headings terms (MeSH-terms) and keywords. The following keywords were used: “drug-coated balloon”, “percutaneous coronary intervention”, “myocardial infarction”, “coronary artery disease” and “randomized”. The meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) Statement. The study protocol was submitted to PROSPERO (ID: CRD42022372350).
Study Selection
There will be no restrictions on stent or DCB type. Pre-specified inclusion criteria were: 1) RCTs of patients receiving DCB versus stents; 2) percutaneous coronary intervention (PCI) for de novo lesions with mean reference lumen diameters ≥ 2.75 mm (This value follows the previous study [20]); 3) The study reported at least one of the following outcomes: myocardial infarction (MI), target lesion revascularization (TLR), cardiovascular death (CD), minimal lumen diameter (MLD) and late lume loss (LLL). Excluded criteria included: 1) Reviews, meta-analyses, case reports, and observational studies; 2) Studies of patients without CAD; 3) Studies that did not report endpoints were excluded; 4) data insufficient. The DCB group included any coronary vessels that underwent only drug-coated balloon angioplasty. Patients in the stent group underwent balloon pre-dilation before stent implantation.
Outcomes and Definition
The primary endpoint was major cardiovascular adverse events (MACE), as defined by the individual trials (Table S1). Secondary endpoints included individual components of MACE, MLD, and LLL. CD was defined as any death that was not clearly of extracardiac origin, and myocardial infarction. MI was defined according to the fourth universal definition of MI [21]. TLR was defined as any repeat revascularization within the stented or DCB-treated segment. MLD was defined as minimal lumen diameter at the end of follow-up. LLL was defined as the MLD immediately after the procedure minus the MLD at the longest follow-up.
Data Extraction
Relevant information from eligible studies was extracted using prespecified data collection forms. Two reviewers (Ma and Liu) extracted the following data from each eligible study: author, year, sample size, age, gender, underlying disease, vessel diameter, type of DCB and stents, clinical outcomes (MACE or components of the MACE), angiographic outcomes (MLD or LLL), and duration of follow-up. For the Vos et al. study, the 2-year-follow-up data were used to obtain additional information [22]. If data were reported only as median and interquartile range or confidence interval, we followed the Cochrane’s recommendation to approximate the values of mean and standard deviation.
Risk of Bias Assessment
A risk-of-bias assessment was conducted using the Cochrane Collaboration tool for assessing risk of bias [23]. Risk of bias was assessed independently by some authors (Ma, Liu and Li), and disagreement was resolved by discussion. Each study was evaluated for adequacy of randomization (selection bias), blinding for participants and personnel and statistician responsible for analysis (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), and other bias (conflict of interest). Each risk of bias was rated as either low, unclear, moderate, or high for the RCT.
Trial Sequential Analysis
To determine whether the evidence in our meta-analysis is reliable, we performed a trial sequential analysis (TSA) for MACE. When the cumulative z curve crosses the trial sequential monitoring boundary, a sufficient level of evidence for the anticipated intervention effect may have been reached and no further trials need be included. If the z curve crosses none of the boundaries, and the required information size has not been reached, there is insufficient evidence to reach a conclusion. TSA was undertaken with type I error of 5% and type II error of 20% by using a random-effects model for those trials. The information size (n = 917) was calculated using an anticipated intervention effect of RR = 0.48 (the intervention effect obtained from our meta-analysis), and the control event proportion of 7%. TSA was performed using TSA Version 0.9.5.10 Beta.
Statistical Analyses
A meta-analysis was performed on the basis of aggregate data in eligible studies reporting comparisons of DCB and stents. Risk ratio (RR) or risk difference with a 95% confidence interval was used as a measure of relative risk for the categorical data, such as TLR, mortality of CD, and mortality of MI. Mean difference (MD) with the 95% CI was calculated as the effect size for endpoint with continuous data, such as MLD or LLL. The Cochrane risk-of-bias tool was used for RCTs. The Cochrane Q test and I2 index were used to quantify the heterogeneity of each study. We considered heterogeneity to be significant when the p-value of the Q-test was < 0.1 or the I2 statistic was ≥ 50%. Random‐effects model was adopted if there was evidence of heterogeneity. Otherwise, fixed‐effects model was used. Subgroup analyses for the primary and secondary outcomes were performed based on age, sex, diabetes status, hypertension status, and comparators (DCB vs. DES and DCB vs. BMS). The results were displayed as a forest plot. Statistical analyses were conducted following the recommendations of the Cochrane Handbook for Systematic Reviews of Interventions and the PRISMA statement. All data analyses were performed by R software (Version R-4.1.2 for Windows).
Results
Study Population
We identified 2077 studies according to the search strategy. After excluding duplicates, another 1395 studies were excluded according to titles and abstracts. Finally, 7 RCTs were included in our analysis (Fig. 1). Among the 7 included studies, all studies reported clinical outcomes (MACE or components of the MACE). The study of Rissanen et al. was powered for MACE. In the study by Rissanen et al., patients were eligible if they had an ischaemic de-novo lesion in a coronary artery and at least one risk factor for bleeding. In the study by Rissanen et al., the average reference vessel diameter was over 2.75 mm in approximately 80% of patients. However, the study by Rissanen et al. did not report angiographic outcomes (MLD or LLL).
The cochrane tool was used to assess the quality of all 7 studies. Qualitative evaluation of the included trials revealed overall moderate risk of bias, which was attributable to incomplete description of random sequence generation and assignment concealment, the infeasibility of operator blinding due to manufacturing differences in devices, and the rate of patients lost at clinical and angiography follow-up (Figure S1).
Overall, 770 patients (384 patients randomized to the balloon group and 386 patients randomized to the stent group) were included. The mean age of patients ranged from 49 to 77 years. 512 patients (66%) underwent PCI for ACS. The average reference vessel diameter was over 2.75 mm. More than half of the patients reported hypertension. Follow-up time ranged from 6 to 12 months. In type of stents, two studies (Rissanen et al. and Shin et al.) including 248 patients used BMS as controls [17, 18]. All types of balloon were paclitaxel-coated or iopromide balloon. In the 7 studies we included, most studies excluded left main coronary artery lesion, severe calcification lesion, or chronic total occlusion. Baseline characteristics of the patients are summarized in Table 1 and Table S2.
Main Outcomes
MACE
MACE were reported in 7 trials (Fig. 2). Patients assigned to DCB showed a 52% risk reduction as compared with those receiving stents (12 vs 28 events; RR: 0.48; 95% CI: 0.24 to 0.97; P = 0.04). The statistical heterogeneity was low (I2 = 6%). Among these studies, the study of Rissanen et al. had the highest weight (38.2% of total). Sequential removal of each trial, one at a time, showed that the result became nonsignificant by removing the study by Rissanen et al. (P = 0.16). Aside from this, other results are stable: the RR ranged from 0.34 (without the study of Vos et al.; P < 0.01) to 0.47 (without the study of Shin et al.; P = 0.03) (Figure S2). Overall, visual inspection of the contour-enhanced funnel plot for MACE revealed a roughly symmetric distribution (Figure S2).
Subgroup analysis of the MACE rate was performed to evaluate the effect of type of patient status (ACS vs. CCS). Compared to stents, DCB showed a significant clinical effect only in patients with CCS (2 vs. 14 events; RR: 0.25; 95% CI: 0.07 to 0.89; P = 0.03; I2 = 0%), but not the patients with ACS (9 vs. 18 events; RR: 0.60; 95% CI: 0.24 to 1.49; P = 0.26; I2 = 28%) (Fig. 3). Stratifying the trials according to the type of stents used, we observed a significant clinical effect in the DCB group compared with the BMS group (2 vs. 14 events; RR: 0.19; 95% CI: 0.05 to 0.73; P = 0.01; I2 = 0%), while the trials using the DCB vs. DES were associated with a nonsignificant treatment effect (10 vs. 14 events; RR: 0.69; 95% CI: 0.30 to 1.60; P = 0.38; I2 = 0%) (Fig. 3).
TSA
As shown in Fig. 4, TSA for MACE was conducted using random effects, and the cumulative Z-curve crossed the trial sequential monitoring boundary, but not the RIS boundary. The result of TSA showed the stability of our meta-analysis results.
Trial sequential analysis of the included studies. A one-sided graph is plotted by TSA, where the dotted lines represent the conventional significance boundaries, the blue line indicates the cumulative Z-score, and the red lines shows the trial sequential monitoring boundary. TSA, Trial sequential analysis; RIS, Required information size
TLR
Overall, there were 10 events among 405 patients randomized to DCB and 24 events among 410 patients randomized to stents (Fig. 5). Based on a random-effect meta-analysis, the RR was 0.53 (10 vs. 24 events; 95% CI:0.25 to 1.14; P = 0.10; I2 = 0%).
MI and CD
MI event was reported in 4 trials (Fig. 5). Analysis of the pooled data revealed that DCB did not reduce the risk of MI compared with stents (6 vs. 14 events; RR: 0.58; 95% CI: 0.21 to 1.54; P = 0.27; I2 = 0%). In addition, only two studies reported CD events (Fig. 5). Similarly, there was no significant effect observed in CD events (2 vs. 7 events; RR: 0.33; 95% CI: 0.06 to 1.78; P = 0.19; I2 = 0%).
LLL
LLL event was reported in 6 trials (Fig. 6). Overall, the results showed that LLL in DCB group was smaller than stent group (SMD: -0.57; 95% CI: -1.09 to -0.05; P = 0.03; I2 = 85%). To evaluate the stability of the results, we conducted a sensitivity analysis. We excluded each article in order, but the summary results did not significantly change (I2 from 79 to 88%). Subsequently, we investigated the possible combinations of trials resulted in an I2 value below the threshold of low heterogeneity (< 50%). Only by removing the combination of the study by Gobić et al., the study by Vos et al. and the study by Shin et al., was heterogeneity low (I2 = 0%), suggesting that this cluster introduced relevant differences in term of LLL.
MLD
Results of the meta-analysis for the MLD are illustrated in Fig. 6. The pooled risk of MLD was similar between the 2 treatments (SMD: -0.34; 95% CI: -0.72 to 0.05; P = 0.08; I2 = 73%;). To analyze the individual impact on heterogeneity, a single trial was removed on at a time, and the individual influence on I2 was estimated. Using this method, we identified that the study of Shin et al. was the main origin of heterogeneity. After exclusion of this study, the heterogeneity was significantly decreased, and a significant clinical effect was observed in DCB group compared with stents (SMD: -0.30, 95% CI -0.69 to -0.01; P = 0.02; I2 = 20%).
Discussion
In this meta-analysis, we observed 5 key findings: 1) DCB would contribute to a decreased risk in MACE compared with stents and this benefit appears to persist in patients with CCS; 2) DCB were associated with a lower risk of MACE and TLR compared with BMS and a similar effect compared with DES; 3) the LLL in the DCB group was lower than the stent group, but there was high heterogeneity between trials; 4) there was a trend but no significant difference in MLD between the DCB and stents; and 5) DCB were associated with no difference in the incidence of CD and MI as compared with stents.
Our meta-analysis differs from prior meta-analyses in several aspects [13, 14]: 1) we found, for the first time, that DCB could reduce the risk of MACE and TLR compared with stents in large vessel de novo coronary lesions; 2) Unlike the previous meta-analysis by Sun et al., a significant difference in MLD between DCB and stents was not observed; and 3) it provides novel insights regarding the available evidence on DCB vs. stents in large vessel de novo lesions.
Although the use of DCB in patients with small vessel disease has been extensively investigated, evidence for the optimal method of revascularization for large coronary lesions remains inadequate. It has been suggested previously that large coronary arteries have more smooth muscle fibers than small vessel arteries and are more prone to recoil and dissection, which may lead to acute occlusion or restenosis of blood vessels [24]. In a retrospective study, although optimal lesion preparation for the use of DCB has been taken, 10% of patients remain need to be bailout stenting due to recoil and dissection [11]. The safety of DCB compared with stents in large vessel disease remain a concern. In this context, by pooling data from 7 studies, our meta-analysis provided some preliminary results for the potential clinical application of DCB in large coronary vessels.
From the subgroup analysis of the MACE according to the type of stents, we observed a significant clinical effect in the DCB group compared with the BMS group (1.9% vs. 13.7%; RR: 0.19; 95% CI: 0.05 to 0.73; P = 0.01; I2 = 0%). Although most patients undergoing PCI are treated with DES, BMS continue to be used in approximately 16% [25]. These patients tend to have high bleeding risk or are uncertain candidates for dual-antiplatelet therapy. Our results provided an important therapeutic strategy for these patients. In addition, DCB did not increase the risk of MACE compared with DES through subgroup analysis (3% vs. 5.5%; RR: 0.69; 95% CI: 0.30 to 1.60; P = 0.38; I2 = 0%). This finding is in accordance with patients with small vessel disease. In BASKET-SMALL 2 trial including 758 patients with de novo in coronary vessels < 3 mm, the rates of MACE are similar in the DCB group and the DES group (15% vs. 15%; P = 0.95) [26]. In addition, a similar result was observed in a meta-analysis including 1824 patients with small vessel disease. There was no significant difference between DCB and DES (11.0% vs. 13.5%; P = 0.57) [27]. Based on our results, the use of DCB is safe for patients with large vessel disease.
In our study, the incidence of MACE was 3% in DCB and 5.5% in DES, which was much lower than the values in the above studies. There are several possible reasons: 1) In RCTs, 36 patients were not included in the final analysis because the bail-out stenting was forced after the randomization and therefore were excluded from these trials (Table S3). If these 36 patients were included in the final analysis, the incidence of MACE would be about 10%. This result was similar to previous studies [11]; 2) the improvement of operator experience will promote the decrease of bail-out stenting. In clinical practice, secure lesion pre-dilation is absolutely necessary. In the case of unsatisfactory residual stenosis or coronary dissection of Type C or above, bail-out stenting should be performed [3].
The lower incidence of MACE was only observed in patients with CCS (1.2% VS. 8.9%; RR: 0.25; 95% CI: 0.07 to 0.89; P = 0.03; I2 = 0%), but not in patients with ACS (4.0% VS. 8.1%; RR: 0.60; 95% CI: 0.24 to 1.49; P = 0.26; I2 = 28%). Although it has been proposed that DCB in patients with ACS has many potential advantages, such as low risk of thrombosis due to less malposition and homogeneous administration of the drug [28], these advantages did not appear to translate into improved clinical outcomes. In a retrospective registry study, including 487 patients with large vessel coronary lesions, the MACE rate was higher in patients presenting with STEMI (12%) than those with CCS (7%), with p-value less than 0.05 [11]. A rather similar result was also described in a recent study [12]. A mean follow-up of 24 months in the study by Tervo et al. showed that the incidence of MACE was significantly higher in patients with STEMI than in patients with CCS (23.9% vs. 7.8%, P < 0.01). Because the number of relevant RCT reports is limited, more studies are needed to confirm the efficacy and safety of DCB in patients with CCS or ACS.
Another important finding of this meta-analysis is that the LLL in the DCB group was lower than the stents group (SMD: -0.57; 95% CI: -1.09 to -0.05; P = 0.03), with high heterogeneity (I2 = 85%). The current opinion is that unfavorable remodeling is considered the major determinant of LLL [29]. However, it is unclear whether this remodeling could lead to adverse clinical events. A patient-level meta-analysis including 2426 patients showed that an angiographic LLL ≤ 0.50 mm was not predictive of the incidence of TLR whereas a LLL > 0.50 mm was [30]. This suggested that modest lumen loss does not seem to affect coronary blood flow significantly. Most of the values of LLL in our study are less than 0.5 mm, which suggested that the capability to predict future TLR is very limited. In addition, heterogeneity was not detected by excluding the combination of studies by Gobić et al., Vos et al., and Shin et al. (I2 = 0%). In these studies, patients tend to have fewer (< 50%) risk factors such as hypertension or diabetes. It may be a reason to explain the heterogeneity.
Previous studies had raised concerns about late mortality with DCB [31, 32]. In our analysis, there were no differences in risk of adverse events (MI or CD). This could be for two reasons. 1) the median follow-up considered in this meta-analysis was only 9 months. In such a short amount of time, the exploration of strong but rarer endpoints such as CD or MI is less effective. Longer follow‐up is needed to confirm the efficacy and safety of DCB; 2) patients in this meta-analysis were not only STEMI (57%) but also CCS (43%) on optimal medical therapy with a lower cardiovascular risk, so it is not surprising that in many studies only few death events were registered. As things stand, DCB may be a safe therapeutic option in patients with large vessel de novo coronary artery disease.
Our meta-analysis has several limitations: 1) in this study, different types of DCB were used, such as SeQuent Please DCB, Pantera Lux DCB, and Vasoguard™ DCB. Due to the limited number of included studies, subgroup analysis could not be conducted; 2) there were differences in the core laboratory assessment of the angiographic outcomes (LLL or MLD) between trials. In addition, several studies used different definitions of clinical endpoints in their studies-MACE, TLR, CD, and MI. These may be the reason for the heterogeneity; 3) the lack of patient-level data precluded a careful evaluation for the patient and lesion characteristics that would benefit most from DCBs; 4) This study was limited by the small sample size. Thus, TSA was used to correct these random errors and test for more reliable and conclusive evidence. 5) The majority of the included studies in our analysis excluded left main coronary artery lesion, severe calcification lesion, or chronic total occlusion. Therefore, our results may not be generalizable to these lesions.
Conclusions
In this meta-analysis of 7 studies comprising 770 patients with large vessel de novo coronary artery disease, DCB were associated with a lower risk of MACE, TLR and LLL compared with stents. In addition, there was no significant difference in the risk of MI and CD between DCB and stents. According to subgroup analysis, the reduction of MACE appears to persist in patients with CCS, and patients receiving DCB vs. BMS. More high-quality research is needed to provide high-quality evidence for further support of DCB efficacy.
Data availability
All data generated or analyzed during this study are included in this article.
References
Benjamin EJ, Muntner P, Alonso A, et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association [published correction appears in Circulation. 2020 Jan 14;141(2):e33]. Circulation. 2019;139(10):e56-e528. https://doi.org/10.1161/CIR.0000000000000659
Liberale L, Badimon L, Montecucco F, Lüscher TF, Libby P, Camici GG. Inflammation, Aging, and Cardiovascular Disease: JACC Review Topic of the Week. J Am Coll Cardiol. 2022;79(8):837–47. https://doi.org/10.1016/j.jacc.2021.12.017.
Jeger RV, Eccleshall S, Wan Ahmad WA, et al. Drug-Coated Balloons for Coronary Artery Disease: Third Report of the International DCB Consensus Group. JACC Cardiovasc Interv. 2020;13(12):1391–402. https://doi.org/10.1016/j.jcin.2020.02.043.
Latib A, Ruparelia N, Menozzi A, et al. 3-Year Follow-Up of the Balloon Elution and Late Loss Optimization Study (BELLO). JACC Cardiovasc Interv. 2015;8(8):1132–4. https://doi.org/10.1016/j.jcin.2015.04.008.
Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization [published correction appears in Eur Heart J. 2019 Oct 1;40(37):3096]. Eur Heart J. 2019;40(2):87–165. https://doi.org/10.1093/eurheartj/ehy394
Scheller B, Hehrlein C, Bocksch W, et al. Treatment of coronary in-stent restenosis with a paclitaxel-coated balloon catheter. N Engl J Med. 2006;355(20):2113–24. https://doi.org/10.1056/NEJMoa061254.
Waksman R, Pakala R. Drug-eluting balloon: the comeback kid? Circ Cardiovasc Interv. 2009;2(4):352–8. https://doi.org/10.1161/CIRCINTERVENTIONS.109.873703.
Axel DI, Kunert W, Göggelmann C, et al. Paclitaxel inhibits arterial smooth muscle cell proliferation and migration in vitro and in vivo using local drug delivery. Circulation. 1997;96(2):636–45. https://doi.org/10.1161/01.cir.96.2.636.
Yu X, Wang X, Ji F, et al. A Non-inferiority, Randomized Clinical Trial Comparing Paclitaxel-Coated Balloon Versus New-Generation Drug-Eluting Stents on Angiographic Outcomes for Coronary De Novo Lesions [published correction appears in Cardiovasc Drugs Ther. 2021 Apr 12;:]. Cardiovasc Drugs Ther. 2022;36(4):655–664. https://doi.org/10.1007/s10557-021-07172-4
Wang Z, Yin Y, Li J, et al. New Ultrasound-Controlled Paclitaxel Releasing Balloon vs. Asymmetric Drug-Eluting Stent in Primary ST-Segment Elevation Myocardial Infarction- A Prospective Randomized Trial. Circ J. 2022;86(4):642–650. https://doi.org/10.1253/circj.CJ-21-0315
Uskela S, Kärkkäinen JM, Eränen J, et al. Percutaneous coronary intervention with drug-coated balloon-only strategy in stable coronary artery disease and in acute coronary syndromes: An all-comers registry study. Catheter Cardiovasc Interv. 2019;93(5):893–900. https://doi.org/10.1002/ccd.27950.
Tervo J, Kärkkäinen JM, Rissanen TT. Technical success, clinical efficacy, and insight into the causes of restenosis after the percutaneous coronary intervention of de novo coronary artery lesions using a paclitaxel-coated balloon with citrate ester excipient. Front Cardiovasc Med. 2022;9:1012473. Published 2022 Oct 31. https://doi.org/10.3389/fcvm.2022.1012473.
Sun K, Liu Z, Wang H. Drug-Coated Balloon vs. Stent for de novo Non-small Coronary Artery Disease: A Systematic Review and Meta-Analysis. Front Cardiovasc Med. 2021;8:700235. Published 2021 Dec 10. https://doi.org/10.3389/fcvm.2021.700235.
Lin Y, Sun X, Liu H, Pang X, Dong S. Drug-coated balloon versus drug-eluting stent for treating de novo coronary lesions in large vessels: a meta-analysis of clinical trials. Medikamentenbeschichteter Ballon vs. medikamentenfreisetzender Stent für die Behandlung von De-novo-Koronarläsionen in großen Gefäßen – eine Metaanalyse klinischer Studien. Herz. 2021;46(3):269–276. https://doi.org/10.1007/s00059-020-04938-8.
Nishiyama N, Komatsu T, Kuroyanagi T, et al. Clinical value of drug-coated balloon angioplasty for de novo lesions in patients with coronary artery disease. Int J Cardiol. 2016;222:113–8. https://doi.org/10.1016/j.ijcard.2016.07.156.
Gobić D, Tomulić V, Lulić D, et al. Drug-Coated Balloon Versus Drug-Eluting Stent in Primary Percutaneous Coronary Intervention: A Feasibility Study. Am J Med Sci. 2017;354(6):553–60. https://doi.org/10.1016/j.amjms.2017.07.005.
Rissanen TT, Uskela S, Eränen J, et al. Drug-coated balloon for treatment of de-novo coronary artery lesions in patients with high bleeding risk (DEBUT): a single-blind, randomised, non-inferiority trial [published correction appears in Lancet. 2019 Jul 20;394(10194):218]. Lancet. 2019;394(10194):230–239. https://doi.org/10.1016/S0140-6736(19)31126-2.
Shin ES, Lee JM, Her AY, et al. Prospective randomized trial of paclitaxel-coated balloon versus bare-metal stent in high bleeding risk patients with de novo coronary artery lesions. Coron Artery Dis. 2019;30(6):425–31. https://doi.org/10.1097/MCA.0000000000000755.
Vos NS, Fagel ND, Amoroso G, et al. Paclitaxel-Coated Balloon Angioplasty Versus Drug-Eluting Stent in Acute Myocardial Infarction: The REVELATION Randomized Trial. JACC Cardiovasc Interv. 2019;12(17):1691–9. https://doi.org/10.1016/j.jcin.2019.04.016.
Jeger RV, Farah A, Ohlow MA, et al. Drug-coated balloons for small coronary artery disease (BASKET-SMALL 2): an open-label randomised non-inferiority trial. Lancet. 2018;392(10150):849–56. https://doi.org/10.1016/S0140-6736(18)31719-7.
Thygesen K, Alpert JS, Jaffe AS, et al. Fourth Universal Definition of Myocardial Infarction (2018). J Am Coll Cardiol. 2018;72(18):2231–64. https://doi.org/10.1016/j.jacc.2018.08.1038.
Niehe SR, Vos NS, Van Der Schaaf RJ, et al. Two-Year Clinical Outcomes of the REVELATION Study: Sustained Safety and Feasibility of Paclitaxel-Coated Balloon Angioplasty Versus Drug-Eluting Stent in Acute Myocardial Infarction. J Invasive Cardiol. 2022;34(1):E39–42.
Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. Published 2019 Aug 28. https://doi.org/10.1136/bmj.l4898.
Wagenseil JE, Mecham RP. Vascular extracellular matrix and arterial mechanics. Physiol Rev. 2009;89(3):957–89. https://doi.org/10.1152/physrev.00041.2008.
Moussa ID, Mohananey D, Saucedo J, et al. Trends and Outcomes of Restenosis After Coronary Stent Implantation in the United States. J Am Coll Cardiol. 2020;76(13):1521–31. https://doi.org/10.1016/j.jacc.2020.08.002.
Jeger RV, Farah A, Ohlow MA, et al. Long-term efficacy and safety of drug-coated balloons versus drug-eluting stents for small coronary artery disease (BASKET-SMALL 2): 3-year follow-up of a randomised, non-inferiority trial [published correction appears in Lancet. 2020 Nov 7;396(10261):1490]. Lancet. 2020;396(10261):1504–1510. https://doi.org/10.1016/S0140-6736(20)32173-5.
Megaly M, Rofael M, Saad M, et al. Outcomes with drug-coated balloons in small-vessel coronary artery disease. Catheter Cardiovasc Interv. 2019;93(5):E277–86. https://doi.org/10.1002/ccd.27996.
Yerasi C, Case BC, Forrestal BJ, et al. Drug-Coated Balloon for De Novo Coronary Artery Disease: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75(9):1061–73. https://doi.org/10.1016/j.jacc.2019.12.046.
Yamamoto T, Ishii T, Ishida A. Impact of post physiological assessment after treatment for de novo coronary lesions using drug-coated balloons. Int J Cardiol. 2022;363:11–9. https://doi.org/10.1016/j.ijcard.2022.06.009.
Asano T, Serruys PW, Collet C, et al. Angiographic late lumen loss revisited: impact on long-term target lesion revascularization. Eur Heart J. 2018;39(36):3381–9. https://doi.org/10.1093/eurheartj/ehy436.
Katsanos K, Spiliopoulos S, Kitrou P, Krokidis M, Karnabatidis D. Risk of Death Following Application of Paclitaxel-Coated Balloons and Stents in the Femoropopliteal Artery of the Leg: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J Am Heart Assoc. 2018;7(24):e011245. https://doi.org/10.1161/JAHA.118.011245.
Milojevic M, Head SJ, Parasca CA, et al. Causes of Death Following PCI Versus CABG in Complex CAD: 5-Year Follow-Up of SYNTAX. J Am Coll Cardiol. 2016;67(1):42–55. https://doi.org/10.1016/j.jacc.2015.10.043.
Acknowledgements
We gratefully acknowledge our colleagues for their cooperation and assistance throughout the course of this study.
Funding
There is no fund support for this work.
Author information
Authors and Affiliations
Contributions
MZQ and LKL wrote the manuscript. MZQ, LKL and HYH conducted the literature search, data extraction, and data analysis. MZQ, HXW and HYH contributed to the data analysis and to writing the manuscript. LZY and LWH reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ma, Z., Liu, K., Hu, Y. et al. Comparison Between Drug-Coated Balloon and Stents in Large De Novo Coronary Artery Disease: A Systematic Review and Meta-Analysis of RCT Data. Cardiovasc Drugs Ther (2024). https://doi.org/10.1007/s10557-024-07548-2
Accepted:
Published:
DOI: https://doi.org/10.1007/s10557-024-07548-2