Abstract
Purpose
This study was designed to explore the effects of interleukin 33 (IL-33) on the progression of atherosclerosis and the possible mechanism.
Methods
The adhesion assay was performed on isolated peripheral blood mononuclear cells (PBMCs) and human umbilical vein endothelial cells (HUVEC). The expression of proteins and messenger RNA (mRNA) were detected by western blot and quantitative real-time polymerase chain reaction (PCR), including intercellular cell adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), and P-selectin. The effect of IL-33 on the interaction of growth stimulation expressed gene 2 (ST2) with myeloid differentiation factor 88 (MyD88) and interleukin-1 receptor-associated kinase (IRAK) 1/4 were investigated using co-immunoprecipitation assay. An apolipoprotein (Apo) E-/- mice model was used to confirm the effect of IL-33 on atherosclerosis progression. Area of plaques was recorded by hematoxylin-eosin (H&E) staining. The severity of atherosclerosis plaque was evaluated using immunohistochemistry assay, and lipid accumulation was measured by an oil red O staining. In contrast, western blot was performed to detect the expression levels of VCAM-1, extracellular signal-regulated kinase (ERK) 1/2, and interferon regulatory factor 1 (IRF1).
Results
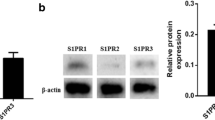
Our study observed that IL-33 suppressed cell adhesion and the expression of VCAM-1 in tumor necrosis factor-α (TNF-α) exposed HUVEC. Moreover, the addition of IL-33 significantly inhibited the expression of IRF1 and the binding level of IRF1 to VCAM-1 and also promoted the phosphorylation level of IRAK1/4 and ERK1/2 compared to TNF-α-stimulated HUVEC. The ST2 neutralizing antibody or ERK pathway inhibitor SCH772984 reversed the regulatory effects of IL-33 on HUVEC, suggesting that IL-33 suppressed IRF1 and VCAM-1 dependent on binding to ST2 and activating the ERK1/2 signaling pathway. Further investigation in vivo confirmed that IL-33 decreased the expressions of IRF1 and VCAM-1 by activating the phosphorylation of ERK1/2 in the thoracic aorta of Apo E-/- mice.
Conclusion
In conclusion, our results demonstrated that IL-33 plays a protective role in the progression of atherosclerosis by inhibiting cell adhesion via the ERK1/2-IRF1-VCAM-1 pathway. This study may provide a potential therapeutic way to prevent the development of atherosclerosis.





Similar content being viewed by others
References
Marijon E, Narayanan K, Smith K, et al. The Lancet Commission to reduce the global burden of sudden cardiac death: a call for multidisciplinary action. Lancet. 2023;402(10405):883–936.
Deng P, Fu Y, Chen M, Wang D, et al. Temporal trends in inequalities of the burden of cardiovascular disease across 186 countries and territories. Int J Equity Health. 2023;22(1):164.
Besedovsky L, Lange T, Haack M. The Sleep-Immune Crosstalk in Health and Disease. Physiol Rev. 2019;99(3):1325–80.
Yin C, Ackermann S, Ma Z, et al. ApoE attenuates unresolvable inflammation by complex formation with activated C1q. Nat Med. 2019;25(3):496–506.
Wojtasińska A, Frąk W, Lisińska W, et al. Atherosclerosis and inflammation. New therapeutic approaches. Int J Mol Sci. 2023;24(17):13434.
Bobryshev YV. Monocyte recruitment and foam cell formation in atherosclerosis. Micron. 2006;37(3):208–22.
Ríos-Navarro C, de Pablo C, Collado-Diaz V, et al. Differential effects of anti-TNF-α and anti-IL-12/23 agents on human leukocyte-endothelial cell interactions. Eur J Pharmacol. 2015;765:355–65.
Khodabandehlou K, Masehi-Lano JJ, Poon C, et al. Targeting cell adhesion molecules with nanoparticles using in vivo and flow-based in vitro models of atherosclerosis. Exp Biol Med (Maywood). 2017;242(8):799–812.
Ley K, Laudanna C, Cybulsky MI, et al. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol. 2007;7(9):678–89.
Diaz Sanchez L, Sanchez-Aranguren L, Wang K, et al. TNF-α-mediated endothelial cell apoptosis is rescued by hydrogen sulfide. Antioxidants (Basel). 2023;12(3):734.
Schmitz J, Owyang A, Oldham E, et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity. 2005;23(5):479–90.
Lamkanfi M, Dixit VM. IL-33 raises alarm. Immunity. 2009;31(1):5–7.
Cayrol C, Girard JP. Interleukin-33 (IL-33): a nuclear cytokine from the IL-1 family. Immunol Rev. 2018;281(1):154–68.
Park SB, Kim SJ, Cho SW, et al. Blocking of the IL-33/ST2 signaling axis by a single-chain antibody variable fragment (scFv) specific to IL-33 with a defined epitope. Int J Mol Sci. 2020;21(18):6953.
Griesenauer B, Paczesnpsy S. The ST2/IL-33 axis in immune cells during inflammatory diseases. Front Immunol. 2017;8:475.
Aimo A, Migliorini P, Vergaro G, et al. The IL-33/ST2 pathway, inflammation and atherosclerosis: trigger and target? Int J Cardiol. 2018;267:188–92.
Liew FY, Pitman NI, McInnes IB. Disease-associated functions of IL-33: the new kid in the IL-1 family. Nat Rev Immunol. 2010;10(2):103–10.
Miller AM, Xu D, Asquith DL, et al. IL-33 reduces the development of atherosclerosis. J Exp Med. 2008;205(2):339–46.
Ha SJ, Lee J, Song KM, et al. Ultrasonicated Lespedeza cuneata extract prevents TNF-α-induced early atherosclerosis in vitro and in vivo. Food Funct. 2018;9(4):2090–101.
Zhou Y, Cao ZQ, Wang HY, et al. The anti-inflammatory effects of Morin hydrate in atherosclerosis is associated with autophagy induction through cAMP signaling. Mole Nutri Food Res. 2017;61(9):1600966.
Liu X, Pan L, Wang X, et al. Leonurine protects against tumor necrosis factor-α-mediated inflammation in human umbilical vein endothelial cells. Atherosclerosis. 2012;222(1):34–42.
Jian D, Wang Y, Jian L, et al. METTL14 aggravates endothelial inflammation and atherosclerosis by increasing FOXO1 N6-methyladeosine modifications. Theranostics. 2020;10(20):8939.
Lee J, Ha SJ, Park J, et al. Arctium lappa root extract containing L-arginine prevents TNF-α-induced early atherosclerosis in vitro and in vivo. Nutri Res. 2020;77:85–96.
Stojkovic S, Kaun C, Heinz M, et al. Interleukin-33 induces urokinase in human endothelial cells―possible impact on angiogenesis. J Thrombosis Haemostasis. 2014;12(6):948–57.
Chalubinski M, Wojdan K, Luczak E, et al. IL-33 and IL-4 impair barrier functions of human vascular endothelium via different mechanisms. Vasc Pharmacol. 2015;73:57–63.
Choi YS, Choi HJ, Min JK, et al. Interleukin-33 induces angiogenesis and vascular permeability through ST2/TRAF6-mediated endothelial nitric oxide production. Blood J Am Soc Hematol. 2009;114(14):3117–26.
Demyanets S, Konya V, Kastl SP, et al. Interleukin-33 induces expression of adhesion molecules and inflammatory activation in human endothelial cells and in human atherosclerotic plaques. Arteriosclerosis Thrombosis Vasc Biol. 2011;31(9):2080–9.
Liu Y, Fang X, Yuan J, et al. The role of corticotropin-releasing hormone receptor 1 in the development of colitis-associated cancer in mouse model. Endocr Relat Cancer. 2014;21(4):639–51.
Zaki MH, Vogel P, Malireddi RK, et al. The NOD-like receptor NLRP12 attenuates colon inflammation and tumorigenesis. Cancer Cell. 2011;20(5):649–60.
Fan CS, Chen CC, Chen LL, et al. Extracellular HSP90α induces MyD88-IRAK complex-associated IKKα/β-NF-κB/IRF3 and JAK2/TYK2-STAT-3 signaling in macrophages for tumor-promoting M2-polarization. Cells. 2022;11(2):229.
Ohto-Ozaki H, Kuroiwa K, Mato N, et al. Characterization of ST2 transgenic mice with resistance to IL-33. Eur J Immunol. 2010;40(9):2632–42.
Demyanets S, Konya V, Kastl SP, et al. Interleukin-33 induces expression of adhesion molecules and inflammatory activation in human endothelial cells and in human atherosclerotic plaques. Arterioscler Thromb Vasc Biol. 2011;31(9):2080–9.
Tembhre MK, Sriwastva MK, Hote MP, et al. Interleukin-33 induces neutrophil extracellular trap (NET) formation and macrophage necroptosis via enhancing oxidative stress and secretion of proatherogenic factors in advanced atherosclerosis. Antioxidants (Basel, Switzerland). 2022;11(12):2343.
Altara R, Ghali R, Mallat Z, et al. Conflicting vascular and metabolic impact of the IL-33/sST2 axis. Cardiovasc Res. 2018;114(12):1578–94.
Lin CC, Yang CC, Wang CY, et al. NADPH uxidase/ROS-dependent VCAM-1 induction on TNF-α-challenged human cardiac fibroblasts enhances monocyte adhesion. Front Pharmacol. 2015;6:310.
Neish AS, Read MA, Thanos D, Pine R, Maniatis T, Collins T. Endothelial interferon regulatory factor 1 cooperates with NF-kappa B as a transcriptional activator of vascular cell adhesion molecule 1. Mol Cell Biol. 1995;15(5):2558–69.
Du M, Wang X, Mao X, et al. Absence of interferon regulatory factor 1 protects against atherosclerosis in apolipoprotein E-deficient mice. Theranostics. 2019;9(16):4688–703.
Hsieh HL, Lin CC, Shih RH, Hsiao LD, Yang CM. NADPH oxidase-mediated redox signal contributes to lipoteichoic acid-induced MMP-9 upregulation in brain astrocytes. J Neuroinflammation. 2012;9:110.
Palomer X, Salvadó L, Barroso E, Vázquez-Carrera M. An overview of the crosstalk between inflammatory processes and metabolic dysregulation during diabetic cardiomyopathy. Int J Cardiol. 2013;168(4):3160–72.
Buckley ML, Williams JO, Chan YH, et al. The interleukin-33-mediated inhibition of expression of two key genes implicated in atherosclerosis in human macrophages requires MAP kinase, phosphoinositide 3-kinase and nuclear factor-κB signaling pathways. Sci Rep. 2019;9(1):11317.
Yamamoto M, Umebashi K, Tokito A, et al. Interleukin-33 induces growth-regulated oncogene-α expression and secretion in human umbilical vein endothelial cells. Am J Physiol Regul Integr Comp Physiol. 2017;313(3):R272–9.
Umebashi K, Tokito A, Yamamoto M, et al. Interleukin-33 induces interleukin-8 expression via JNK/c-Jun/AP-1 pathway in human umbilical vein endothelial cells. PLoS One. 2018;13(1):e0191659.
Author information
Authors and Affiliations
Contributions
Zhang Qian conceived, designed and performed the experiments, analyzed data, wrote and revised the manuscript. Feng Shaofang and Shi chunhua conducted the research and analyzed the data. Chen Chen helped us revise the manuscript. Wang Nan and Liu Chao conceived, supervised, funded, and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflicts of Interest
The authors report no conflicts of interest in this work.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

Fig. S1
Determination of the concentrations of TNF-α and IL-33 in HUVEC. (A) VCAM-1 expression in HUVEC after a gradient periods of TNF-α exposure. (B) VCAM-1 expression in HUVEC treated with different concentrations of IL-33 (with or without 10 ng/mL TNF-α). Notes: Data represented mean ± SD; n = 3; **P < 0.01. Abbreviations: IL, interleukin; VCAM-1, vascular cell adhesion molecule-1; TNF, tumor necrosis factor; HUVEC, human umbilical vein endothelial cells (PNG 422 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qian, Z., Shaofang, F., Chen, C. et al. IL-33 Suppresses the Progression of Atherosclerosis via the ERK1/2-IRF1-VCAM-1 Pathway. Cardiovasc Drugs Ther (2023). https://doi.org/10.1007/s10557-023-07523-3
Accepted:
Published:
DOI: https://doi.org/10.1007/s10557-023-07523-3