Abstract
Purpose
Given the beneficial effects of sacubitril/valsartan on blood pressure generally, this study investigates its antihypertension effects in diabetes mellitus (DM) patients with primary hypertension specifically, and the effect of sacubitril/valsartan on glycolipid metabolism.
Methods
We conducted a randomized, open-label, active-controlled study to compare the antihypertension effects of sacubitril/valsartan in DM individuals with primary hypertension. The primary end point was reduction in mean systolic blood pressure (SBP) from baseline with sacubitril/valsartan vs. olmesartan at week 8. The secondary endpoints included the changes in diastolic blood pressure (DBP), daytime SBP/DBP, nighttime SBP/DBP, BP achievement (office sitting BP < 130/80 mmHg), and lipid profile. The trial was registered with chictr.org.cn (ChiCTR2200066428) on Dec 22, 2022.
Results
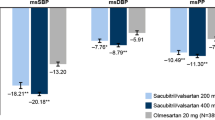
A total of 124 patients were included in the final analysis. SBP decreased to a greater extent in the sacubitril/valsartan group from baseline to 8 weeks [between-treatment difference: 3.51 mm Hg, 95% confidence interval (95% CI) 0.41 to 6.62 mm Hg, P = 0.03]. Furthermore, more patients achieved the blood pressure goal with sacubitril/valasartan (74.60% vs. 54.70%, P = 0.03). Multiple logistical regression analysis showed that sacubitril/valsartan was associated with BP achievement [odds ratio (OR) 0.33, 95% CI 0.14–0.73, P = 0.007], but the difference in SBP, DBP, day time SBP/DBP, and night time SBP/DBP reduction did not approach statistical significance. HbA1C1, total cholesterol, and low-density lipoprotein-cholesterol were lower than baseline in both groups (P < 0.05); however, there was no difference in the effects on glucose and lipid metabolism from sacubitril/valsartan compared to olmesartan.
Conclusions
Sacubitril/valsartan not only provided superior BP reduction compared to olmesartan, it did so without adverse effects on glycemic control and lipid parameters in DM patients with primary hypertension.




Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
The codes used in the analyses can be made available by the corresponding author on request.
References
Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018;14(2):88–98. https://doi.org/10.1038/nrendo.2017.151.
Mills KT, Bundy JD, Kelly TN, et al. Global disparities of hypertension prevalence and control: a systematic analysis of population-based studies from 90 countries. Circulation. 2016;134(6):441–50. https://doi.org/10.1161/circulationaha.115.018912.
Zhou B, Perel P, Mensah GA, Ezzati M. Global epidemiology, health burden and effective interventions for elevated blood pressure and hypertension. Nat Rev Cardiol. 2021;18(11):785–802. https://doi.org/10.1038/s41569-021-00559-8.
Yamazaki D, Hitomi H, Nishiyama A. Hypertension with diabetes mellitus complications. Hypertens Res. 2018;41(3):147–56. https://doi.org/10.1038/s41440-017-0008-y.
Kotseva K, De Backer G, De Bacquer D, et al. Lifestyle and impact on cardiovascular risk factor control in coronary patients across 27 countries: results from the European Society of Cardiology ESC-EORP EUROASPIRE V registry. Eur J Prev Cardiol. 2019;26(8):824–35. https://doi.org/10.1177/2047487318825350.
Weinberger MH. Mechanisms of diuretic effects on carbohydrate tolerance, insulin sensitivity and lipid levels. Eur Heart J. 1992;13 Suppl G:5–9. https://doi.org/10.1093/eurheartj/13.suppl_g.5.
Cooper-DeHoff RM, Pacanowski MA, Pepine CJ. Cardiovascular therapies and associated glucose homeostasis: implications across the dysglycemia continuum. J Am Coll Cardiol. 2009;53(5 Suppl):S28-34. https://doi.org/10.1016/j.jacc.2008.10.037.
Katsi V, Skalis G, Pavlidis AN, et al. Angiotensin receptor neprilysin inhibitor LCZ696: a novel targeted therapy for arterial hypertension? Eur Heart J Cardiovasc Pharmacother. 2015;1(4):260–4. https://doi.org/10.1093/ehjcvp/pvv031.
McMurray JJ, Packer M, Desai AS, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371(11):993–1004. https://doi.org/10.1056/NEJMoa1409077.
Cheung DG, Aizenberg D, Gorbunov V, et al. Efficacy and safety of sacubitril/valsartan in patients with essential hypertension uncontrolled by olmesartan: a randomized, double-blind, 8-week study. J Clin Hypertens (Greenwich). 2018;20(1):150–8. https://doi.org/10.1111/jch.13153.
Huo Y, Li W, Webb R, et al. Efficacy and safety of sacubitril/valsartan compared with olmesartan in Asian patients with essential hypertension: a randomized, double-blind, 8-week study. J Clin Hypertens (Greenwich). 2019;21(1):67–76. https://doi.org/10.1111/jch.13437.
Seferovic JP, Claggett B, Seidelmann SB, et al. Effect of sacubitril/valsartan versus enalapril on glycaemic control in patients with heart failure and diabetes: a post-hoc analysis from the PARADIGM-HF trial. Lancet Diabetes Endocrinol. 2017;5(5):333–40. https://doi.org/10.1016/s2213-8587(17)30087-6.
Armentaro G, D’Arrigo G, Miceli S, et al. Long term metabolic effects of sacubitril/valsartan in non-diabetic and diabetic patients with heart failure reduced ejection fraction: a real life study. Front Physiol. 2022;13:897109. https://doi.org/10.3389/fphys.2022.897109.
Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the american college of cardiology/american heart association task force on clinical practice guidelines. J Am Coll Cardiol. 2018;71(19):e127-248. https://doi.org/10.1016/j.jacc.2017.11.006.
Schmieder RE, Wagner F, Mayr M, et al. The effect of sacubitril/valsartan compared to olmesartan on cardiovascular remodelling in subjects with essential hypertension: the results of a randomized, double-blind, active-controlled study. Eur Heart J. 2017;38(44):3308–17. https://doi.org/10.1093/eurheartj/ehx525.
Kario K. The sacubitril/valsartan, a first-in-class, angiotensin receptor neprilysin inhibitor (ARNI): potential uses in hypertension, heart failure, and beyond. Curr Cardiol Rep. 2018;20(1):5. https://doi.org/10.1007/s11886-018-0944-4.
Seki T, Goto K, Kansui Y, et al. Angiotensin II receptor-neprilysin inhibitor sacubitril/valsartan improves endothelial dysfunction in spontaneously hypertensive rats. J Am Heart Assoc. 2017;6(10):e006617. https://doi.org/10.1161/jaha.117.006617.
Geng Q, Yan R, Wang Z, Hou F. Effects of LCZ696 (Sacubitril/Valsartan) on blood pressure in patients with hypertension: a meta-analysis of randomized controlled trials. Cardiology. 2020;145(9):589–98. https://doi.org/10.1159/000507327.
Supasyndh O, Sun N, Kario K, Hafeez K, Zhang J. Long-term (52-week) safety and efficacy of Sacubitril/valsartan in Asian patients with hypertension. Hypertens Res. 2017;40(5):472–6. https://doi.org/10.1038/hr.2016.151.
Rakugi H, Kario K, Yamaguchi M, et al. Efficacy of sacubitril/valsartan versus olmesartan in Japanese patients with essential hypertension: a randomized, double-blind, multicenter study. Hypertens Res. 2022;45(5):824–33. https://doi.org/10.1038/s41440-021-00819-7.
Pereira GME, Duarte GS, Katerenchuk V, et al. Safety and tolerability of sacubitril-valsartan: a systematic review and meta-analysis. Expert Opin Drug Saf. 2021;20(5):577–88. https://doi.org/10.1080/14740338.2021.1877658.
Li Q, Li L, Wang F, et al. Effect and safety of LCZ696 in the treatment of hypertension: a meta-analysis of 9 RCT studies. Medicine (Baltimore). 2019;98(28):e16093. https://doi.org/10.1097/md.0000000000016093.
Chua SK, Lai WT, Chen LC, Hung HF. The antihypertensive effects and safety of lcz696 in patients with hypertension: a systemic review and meta-analysis of randomized controlled trials. J Clin Med. 2021;10(13):2824. https://doi.org/10.3390/jcm10132824.
Ettehad D, Emdin CA, Kiran A, et al. Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet. 2016;387(10022):957–67. https://doi.org/10.1016/s0140-6736(15)01225-8.
Eid S, Sas KM, Abcouwer SF, et al. New insights into the mechanisms of diabetic complications: role of lipids and lipid metabolism. Diabetologia. 2019;62(9):1539–49. https://doi.org/10.1007/s00125-019-4959-1.
Oparil S, Acelajado MC, Bakris GL, et al. Hypertension Nat Rev Dis Primers. 2018;4:18014. https://doi.org/10.1038/nrdp.2018.14.
Luther JM, Brown NJ. The renin-angiotensin-aldosterone system and glucose homeostasis. Trends Pharmacol Sci. 2011;32(12):734–9. https://doi.org/10.1016/j.tips.2011.07.006.
Oktay AA, Akturk HK, Jahangir E. Diabetes mellitus and hypertension: a dual threat. Curr Opin Cardiol. 2016;31(4):402–9. https://doi.org/10.1097/hco.0000000000000297.
Henriksen EJ, Prasannarong M. The role of the renin-angiotensin system in the development of insulin resistance in skeletal muscle. Mol Cell Endocrinol. 2013;378(1–2):15–22. https://doi.org/10.1016/j.mce.2012.04.011.
Seferovic JP, Solomon SD, Seely EW. Potential mechanisms of beneficial effect of sacubitril/valsartan on glycemic control. Ther Adv Endocrinol Metab. 2020;11:2042018820970444. https://doi.org/10.1177/2042018820970444.
Engeli S, Stinkens R, Heise T, et al. Effect of sacubitril/valsartan on exercise-induced lipid metabolism in patients with obesity and hypertension. Hypertension. 2018;71(1):70–7. https://doi.org/10.1161/hypertensionaha.117.10224.
Barman HA, Tanyolaç S, Dogan O, et al. Impact of sacubitril/valsartan on lipid parameters in patients with heart failure with reduced ejection fraction. Clin Drug Investig. 2022;42(6):533–40. https://doi.org/10.1007/s40261-022-01161-7.
Funding
This research was funded by Cardiovascular Health Alliance Nuo-Chuang Hypertension Research and Innovation Fund (2021-CCA-HTN-10).
Author information
Authors and Affiliations
Contributions
N.-Q.W.: conception/design. W.-J.Z., Y.-G.S., Y.-L.X., H.-T.Z., X.-N.L., H.Q., J.-L.Z., J.-J.L., K.-F.D., and J.Q.: provision of study materials. S.Z., Z.Y., and Z.-F.L.: collection and/or assembly of data. S.Z.: data analysis, interpretation, and manuscript writing. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of Fuwai Hospital.
Consent to Participate
Written informed consent has been obtained from the patient(s) to publish this paper.
Consent for Publication
The authors affirm that human research participants provided informed consent for publication of all tables and figures.
Conflicts of Interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, S., Yin, Z., Li, ZF. et al. The Effects of Sacubitril/Valsartan Compared to Olmesartan on the Blood Pressure and Glucolipid Metabolism in DM Patients with Primary Hypertension. Cardiovasc Drugs Ther (2023). https://doi.org/10.1007/s10557-023-07509-1
Accepted:
Published:
DOI: https://doi.org/10.1007/s10557-023-07509-1