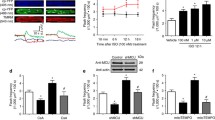
Abstract
Purpose
β-Adrenergic receptors (βAR) are essential targets for the treatment of heart failure (HF); however, chronic use of βAR agonists as positive inotropes to increase contractility in a Gs protein-dependent manner is associated with increased mortality. Alternatively, we previously reported that allosteric modulation of β2AR with the pepducin intracellular loop (ICL)1-9 increased cardiomyocyte contractility in a β-arrestin (βarr)-dependent manner, and subsequently showed that ICL1-9 activates the Ras homolog family member A (RhoA). Here, we aimed to elucidate both the proximal and downstream signaling mediators involved in the promotion of cardiomyocyte contractility in response to ICL1-9.
Methods
We measured adult mouse cardiomyocyte contractility in response to ICL1-9 or isoproterenol (ISO, as a positive control) alone or in the presence of inhibitors of various potential components of βarr- or RhoA-dependent signaling. We also assessed the contractile effects of ICL1-9 on cardiomyocytes lacking G protein-coupled receptor (GPCR) kinase 2 (GRK2) or 5 (GRK5).
Results
Consistent with RhoA activation by ICL1-9, both Rho-associated protein kinase (ROCK) and protein kinase D (PKD) inhibition were able to attenuate ICL1-9-mediated contractility, as was inhibition of myosin light chain kinase (MLCK). While neither GRK2 nor GRK5 deletion impacted ICL1-9-mediated contractility, pertussis toxin attenuated the response, suggesting that ICL1-9 promotes downstream RhoA-dependent signaling in a Gi protein-dependent manner.
Conclusion
Altogether, our study highlights a novel signaling modality that may offer a new approach to the promotion, or preservation, of cardiac contractility during HF via the allosteric regulation of β2AR to promote Gi protein/βarr-dependent activation of RhoA/ROCK/PKD signaling.






Similar content being viewed by others
References
Francis GS, Bartos JA, Adatya S. Inotropes. J Am Coll Cardiol. 2014;63:2069–78.
Bristow MR. Treatment of chronic heart failure with beta-adrenergic receptor antagonists: a convergence of receptor pharmacology and clinical cardiology. Circ Res. 2011;109:1176–94.
Abdel-Magid AF. Allosteric modulators: an emerging concept in drug discovery. ACS Med Chem Lett. 2015;6:104–7.
Ahn S, Pani B, Kahsai AW, Olsen EK, Husemoen G, Vestergaard M, et al. Small-molecule positive allosteric modulators of the beta2-adrenoceptor isolated from DNA-encoded libraries. Mol Pharmacol. 2018;94:850–61.
Okyere AD, Tilley DG. Self-made allostery: endogenous COMP antagonizes pathologic AT1AR signaling. Cell Res. 2021. https://doi.org/10.1038/s41422-021-00493-x.
Wang J, Gareri C, Rockman HA. G-protein-coupled receptors in heart disease. Circ Res. 2018;123:716–35.
Zhang P, Leger AJ, Baleja JD, Rana R, Corlin T, Nguyen N, et al. Allosteric activation of a G protein-coupled receptor with cell-penetrating receptor mimetics. J Biol Chem. 2015;290:15785–98.
Carr R 3rd, Benovic JL. From biased signalling to polypharmacology: unlocking unique intracellular signalling using pepducins. Biochem Soc Trans. 2016;44:555–61.
Carr R 3rd, Schilling J, Song J, Carter RL, Du Y, Yoo SM, et al. beta-Arrestin-biased signaling through the beta2-adrenergic receptor promotes cardiomyocyte contraction. Proc Natl Acad Sci USA. 2016;113:E4107–16.
Wang J, Hanada K, Staus DP, Makara MA, Dahal GR, Chen Q, et al. Galphai is required for carvedilol-induced beta1 adrenergic receptor beta-arrestin biased signaling. Nat Commun. 2017;8:1706.
Grisanti LA, Thomas TP, Carter RL, de Lucia C, Gao E, Koch WJ, et al. Pepducin-mediated cardioprotection via beta-arrestin-biased beta2-adrenergic receptor-specific signaling. Theranostics. 2018;8:4664–78.
Lauriol J, Keith K, Jaffre F, Couvillon A, Saci A, Goonasekera SA, et al. RhoA signaling in cardiomyocytes protects against stress-induced heart failure but facilitates cardiac fibrosis. Sci Signal. 2014;7:ra100.
Raake PW, Vinge LE, Gao E, Boucher M, Rengo G, Chen X, et al. G protein-coupled receptor kinase 2 ablation in cardiac myocytes before or after myocardial infarction prevents heart failure. Circ Res. 2008;103:413–22.
Gainetdinov RR, Bohn LM, Walker JK, Laporte SA, Macrae AD, Caron MG, et al. Muscarinic supersensitivity and impaired receptor desensitization in G protein-coupled receptor kinase 5-deficient mice. Neuron. 1999;24:1029–36.
Grisanti LA, Schumacher SM, Tilley DG, Koch WJ. Designer approaches for G protein-coupled receptor modulation for cardiovascular disease. JACC Basic Transl Sci. 2018;3:550–62.
Pfleger J, Gresham K, Koch WJ. G protein-coupled receptor kinases as therapeutic targets in the heart. Nat Rev Cardiol. 2019;16:612–22.
Daaka Y, Luttrell LM, Lefkowitz RJ. Switching of the coupling of the beta2-adrenergic receptor to different G proteins by protein kinase A. Nature. 1997;390:88–91.
Anthony DF, Sin YY, Vadrevu S, Advant N, Day JP, Byrne AM, et al. beta-Arrestin 1 inhibits the GTPase-activating protein function of ARHGAP21, promoting activation of RhoA following angiotensin II type 1A receptor stimulation. Mol Cell Biol. 2011;31:1066–75.
Barnes WG, Reiter E, Violin JD, Ren XR, Milligan G, Lefkowitz RJ. beta-Arrestin 1 and Galphaq/11 coordinately activate RhoA and stress fiber formation following receptor stimulation. J Biol Chem. 2005;280:8041–50.
Godin CM, Ferguson SS. The angiotensin II type 1 receptor induces membrane blebbing by coupling to Rho A, Rho kinase, and myosin light chain kinase. Mol Pharmacol. 2010;77:903–11.
Tocci P, Cianfrocca R, Di Castro V, Rosano L, Sacconi A, Donzelli S, et al. beta-arrestin1/YAP/mutant p53 complexes orchestrate the endothelin A receptor signaling in high-grade serous ovarian cancer. Nat Commun. 2019;10:3196.
Xiang SY, Vanhoutte D, Del Re DP, Purcell NH, Ling H, Banerjee I, et al. RhoA protects the mouse heart against ischemia/reperfusion injury. J Clin Invest. 2011;121:3269–76.
Yung BS, Brand CS, Xiang SY, Gray CB, Means CK, Rosen H, et al. Selective coupling of the S1P3 receptor subtype to S1P-mediated RhoA activation and cardioprotection. J Mol Cell Cardiol. 2017;103:1–10.
Martin-Garrido A, Biesiadecki BJ, Salhi HE, Shaifta Y, Dos Remedios CG, Ayaz-Guner S, et al. Monophosphorylation of cardiac troponin-I at Ser-23/24 is sufficient to regulate cardiac myofibrillar Ca(2+) sensitivity and calpain-induced proteolysis. J Biol Chem. 2018;293:8588–99.
Ryba DM, Li J, Cowan CL, Russell B, Wolska BM, Solaro RJ. Long-term biased beta-arrestin signaling improves cardiac structure and function in dilated cardiomyopathy. Circulation. 2017;135:1056–70.
Tarigopula M, Davis RT 3rd, Mungai PT, Ryba DM, Wieczorek DF, Cowan CL, et al. Cardiac myosin light chain phosphorylation and inotropic effects of a biased ligand, TRV120023, in a dilated cardiomyopathy model. Cardiovasc Res. 2015;107:226–34.
Thomsen ARB, Plouffe B, Cahill TJ 3rd, Shukla AK, Tarrasch JT, Dosey AM, et al. GPCR-G protein-beta-arrestin super-complex mediates sustained G protein signaling. Cell. 2016;166:907–19.
Nguyen AH, Thomsen ARB, Cahill TJ 3rd, Huang R, Huang LY, Marcink T, et al. Structure of an endosomal signaling GPCR-G protein-beta-arrestin megacomplex. Nat Struct Mol Biol. 2019;26:1123–31.
Song J, Li J, Lulla A, Evers BM, Chung DH. Protein kinase D protects against oxidative stress-induced intestinal epithelial cell injury via Rho/ROK/PKC-delta pathway activation. Am J Physiol Cell Physiol. 2006;290:C1469–76.
Okamoto R, Kato T, Mizoguchi A, Takahashi N, Nakakuki T, Mizutani H, et al. Characterization and function of MYPT2, a target subunit of myosin phosphatase in heart. Cell Signal. 2006;18:1408–16.
Chang AN, Battiprolu PK, Cowley PM, Chen G, Gerard RD, Pinto JR, et al. Constitutive phosphorylation of cardiac myosin regulatory light chain in vivo. J Biol Chem. 2015;290:10703–16.
Chang AN, Kamm KE, Stull JT. Role of myosin light chain phosphatase in cardiac physiology and pathophysiology. J Mol Cell Cardiol. 2016;101:35–43.
Homan KT, Tesmer JJ. Molecular basis for small molecule inhibition of G protein-coupled receptor kinases. ACS Chem Biol. 2015;10:246–56.
Tressel SL, Koukos G, Tchernychev B, Jacques SL, Covic L, Kuliopulos A. Pharmacology, biodistribution, and efficacy of GPCR-based pepducins in disease models. Methods Mol Biol. 2011;683:259–75.
Gurbel PA, Bliden KP, Turner SE, Tantry US, Gesheff MG, Barr TP, et al. Cell-penetrating pepducin therapy targeting PAR1 in subjects with coronary artery disease. Arterioscler Thromb Vasc Biol. 2016;36:189–97.
Availability of Data and Materials
The data underlying this article will be shared upon reasonable request to the corresponding authors.
Funding
This work was supported by National Institutes of Health (R01 HL136219 to DGT and JLB; P01 HL147841 to WJK and DGT; F31 HL154814 to ADO; K99 HL132882 to SMS) and the American Heart Association (17POST33660942 to CdL).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection and analysis were performed by Ama Okyere, Jianliang Song, Viren Patwa, Rhonda Carter, Nitya Enjamuri, Anna Maria Lucchese, Jessica Ibetti, Claudio de Lucia, and Sarah Schumacher. The first draft of the manuscript was written by Ama Okyere and Douglas Tilley and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All animal experiments were conducted under the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee (IACUC) at Temple University under Animal Care and Use Protocols #4902 and #5017.
Consent for Publication
Not applicable
Informed Consent
Not applicable
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Okyere, A.D., Song, J., Patwa, V. et al. Pepducin ICL1-9-Mediated β2-Adrenergic Receptor-Dependent Cardiomyocyte Contractility Occurs in a Gi Protein/ROCK/PKD-Sensitive Manner. Cardiovasc Drugs Ther 37, 245–256 (2023). https://doi.org/10.1007/s10557-021-07299-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-021-07299-4