Abstract
Purpose
To assess real-world data on the clinical implementation of sodium-glucose cotransporter 2 inhibitors (SGLT2i) and glucagon-like peptide-1 receptor agonists (GLP-1RA) in cardiovascular patients and to investigate barriers to prescribe these agents.
Methods
Patients presenting with coronary artery disease (CAD) and type 2 diabetes mellitus (T2DM) between 01/2014 and 04/2020 were included in the present analysis and followed prospectively. All first-time prescriptions of SGLT2i and GLP-1RA were identified.
Results
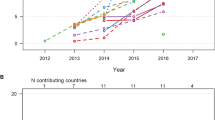
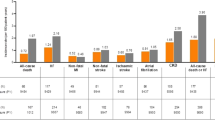
Among 1498 patients with CAD and T2DM, 17.6% of patients received an SGLT2i and 5.5% a GLP-1RA. The prescription of SGLT2i (+38.7%; p < 0.001) and GLP-1RA (+8%; p = 0.007) significantly increased during the observation period. Considering remuneration criteria for SGLT2i therapy, lowering the GFR cut-off to 30 ml/min/1.73 m2 would allow additional 26.6% of patients to qualify for an SGLT2i therapy. While SGLT2i therapy was inversely associated with CV mortality (adjusted hazard ratio of 0.18 [95% CI: 0.05–0.76]; p = 0.019), GLP-1RA therapy showed a trend for risk reduction.
Conclusion
The present analysis revealed an infrequent prescription of SGLT2i and GLP-1RAs in patients with T2DM and CAD in clinical practice. Remuneration regulations that better reflect the inclusion criteria of the CV outcome trials would allow more patients at high risk to receive these CV protective drugs. Most importantly, while GLP-1RA therapy showed a trend for risk reduction of cardiovascular mortality, the use of SGLT2i had a strong inverse impact on cardiovascular mortality from a long-term perspective.


Similar content being viewed by others
Data Availability
Data will be provided upon request.
References
Saeedi P, Petersohn I, Salpea P, Malanda B, Karuranga S, Unwin N, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract. Ireland. 2019;157:107843.
Leon BM, Maddox TM. Diabetes and cardiovascular disease: epidemiology, biological mechanisms, treatment recommendations and future research. World J Diabetes. 2015;6:1246–58.
Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117–28.
Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380:347–57.
Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644–57.
Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JFE, Nauck MA, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311–22.
Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink HJL, Charytan DM, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med. 2019;380:2295–306.
Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Bonaca MP, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Lond Engl. 2019;393:31–9.
Chahine N, Al-Kindi S. Under-prescription of SGLT2 inhibitors in patients with diabetes and cardiovascular disease in the United States. J Am Coll Cardiol. 2020;75:1915.
Cosentino F, Grant PJ, Aboyans V, Bailey CJ, Ceriello A, Delgado V, et al. 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD: The Task Force for diabetes, pre-diabetes, and cardiovascular diseases of the European Society of Cardiology (ESC) and the European Association for the Study of Diabetes (EASD). Eur Heart J. 2020;41:255–323.
Knuuti J, Wijns W, Saraste A, Capodanno D, Barbato E, Funck-Brentano C, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur Heart J. 2020;41:407–77.
Heerspink HJLH. DAPA-CKD trial meets primary endpoint in patients with chronic kidney disease [Internet]. Available from: https://www.escardio.org/The-ESC/Press-Office/Press-releases/DAPA.
McMurray JJV, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381:1995–2008.
Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N Engl J Med United States. 2020;383:1413–24.
Anandhakrishnan A, Korbonits M. Glucagon-like peptide 1 in the pathophysiology and pharmacotherapy of clinical obesity. World J Diabetes. 2016;7:572–98.
Author information
Authors and Affiliations
Contributions
AN, PS, and FH contributed to the conception or design of the work. FH, AH, RS, and FJ contributed to the acquisition, analysis, or interpretation of data for the work. FH drafted the manuscript. AN, NK, LK, and CH critically revised the manuscript. All gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Corresponding author
Ethics declarations
Ethical Approval
This article does not contain any studies with human participants performed by any of the authors.
Due to the observational character of the study, patient informed consent was not required.
The study protocol complies with the declaration of Helsinki and was approved by the local ethics committee of the Medical University of Vienna (EK 2267/2019).
Conflict of Interest
Felix Hofer: none
Niema Kazem: none
Ronny Schweitzer: none
Andreas Hammer: none
Friedrich Jakse: none
Lorenz Koller: none
Christian Hengstenberg: none
Patrick Sulzgruber: grants from Daiichi Sankyo, grants from AstraZeneca, and grants from Boehringer-Ingelheim outside the submitted work
Alexander Niessner: personal fees from Bayer, personal fees from BMS, grants and personal fees from Boehringer Ingelheim, grants and personal fees from Daiichi Sankyo, and personal fees from Pfizer outside the submitted work
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Hofer, F., Kazem, N., Schweitzer, R. et al. Prescription Patterns of Sodium-Glucose Cotransporter 2 Inhibitors and Glucagon-Like Peptide-1 Receptor Agonists in Patients with Coronary Artery Disease. Cardiovasc Drugs Ther 35, 1161–1170 (2021). https://doi.org/10.1007/s10557-021-07160-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-021-07160-8