Abstract
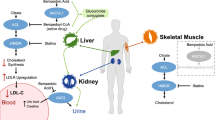
An elevated plasma low-density lipoprotein cholesterol (LDL-C) level is a well-established atherosclerotic cardiovascular disease (ACSVD) risk factor. Randomized studies with statins (alone or in combination with other lipid-lowering drugs) have demonstrated their clinical efficacy in lowering LDL-C. Several classes of new, non-statin agents have been successfully studied and used (e.g., ezetimibe and inhibitors of proprotein convertase subtilisin/kexin type 9 [i-PSCK9]). However, many high ACSVD risk patients remain at a high residual cardiovascular risk, with at least 10% being statin intolerant. Bempedoic acid (ETC-1002) is a new inhibitor of cholesterol synthesis that targets ATP citrate lyase (ACL). Importantly, ETC-1002 is only converted into an active form in the liver and is free of muscle side effects.
Area Covered: Mechanism of action of ETC-1002, clinical pharmacology, completed clinical studies with bempedoic acid, lipid-lowering efficacy/safety issues, and recent meta-analyses of trials with ETC-1002.
Expert Opinion: ETC-1002 has been extensively studied in phase I–III clinical studies in over 4000 individuals from different patient populations (statin intolerance, familial hypercholesterolemia, and high ACSVD risk patients), ETC-1002 has been demonstrated to have moderate cholesterol-lowering efficacy and a good safety profile at a dose of 180 mg/day as a monotherapy and in combination with statins and ezetimibe. The ongoing study CLEAR Outcomes, with composite cardiovascular endpoints, will elucidate the role of bempedoic acid in the management of high ACSVD risk and statin-intolerant patients with hypercholesterolemia. Long-term safety data on bempedoic acid are needed to fully establish this agent in evidence-informed guidelines for managing of patients with dyslipidemias.


Similar content being viewed by others
References
Ference BA, Ginsberg HN, Graham I, et al. Low density lipoprotein cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic and clinical studies. A consensus statement from the European atherosclerosis society consensus panel. Eur Heart J. 2017;38(32):2459–72.
Borén J, Chapman MJ, Krauss RM, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease: pathophysiological, genetic, and therapeutic insights: a consensus statement from the European atherosclerosis society consensus panel. Eur Heart J. 2020;41(24):2313–30.
Authors/Task Force Members; ESC Committee for Practice Guidelines (CPG). ESC National Cardiac Societies 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Atherosclerosis. 2019;290:140–205.
Grundy SM, Stone NJ, Bailey AL, et al. A report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. J Am Coll Cardiol. 2019;73(24):e285–350.
Cannon CP, Blazing MA, Giugliano RP, et al. For IMPROVE-IT investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372(25):2387–97.
Sabatine MS, Giugliano RP, Keech AC, Honarpour N, Wiviott SD, Murphy SA, et al. FOURIER steering committee and investigators. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376(18):1713–22.
Ray KK, Colhoun HM, Szarek M, et al. For ODYSSEY OUTCOMES committees and investigators. Effects of alirocumab on cardiovascular and metabolic outcomes after acute coronary syndrome in patients with or without diabetes: a prespecified analysis of the ODYSSEY OUTCOMES randomized controlled trial. Lancet Diabetes Endocrino. 2019;7(8):618–28.
Ridker PM, JG MF, Glynn RJ, et al. Inhibition of interleukin-1β by Canakinumab and cardiovascular outcomes in patients with chronic kidney disease. J Am Coll Cardiol. 2018;71(21):2405–14.
Bhatt DL, Steg PG, Miller M, Brinton EA, Jacobson TA, Ketchum SB, et al. REDUCE-IT investigators. Cardiovascular risk reduction with Icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380(1):11–22.
Fruchart JC, Santos RD, Aguilar-Salinas C, et al. The selective peroxisome proliferator-activated receptor alpha modulator (SPPARMα) paradigm: conceptual framework and therapeutic potential: a consensus statement from the international atherosclerosis society (IAS) and the residual risk reduction initiative (R3i) foundation. Cardiovacular Diabetol. 2019;18(1):71.
Bloom DJ, Raal FJ, Santos RD, Marais AD. Lomitapide and mipomersen-inhibiting microsomal triglyceride transfer protein (MTP) and apoB100 synthesis. Curr Atheroscler Rep. 2019;21(12):48.
Bilen O, Ballantyne C. Bempedoic acid (ETC-1002): an investigational inhibitor of ATP citrate Lyase. Curr Atheroscler Rep. 2016;18:61.
Pinkosky SL, Filippov S, Srivastava RA, et al. AMP-activated protein kinase and ATP-citrate lyase are two distinct molecular targets for ETC-1002, a novel small molecule regulator of lipid and carbohydrate metabolism. J Lipid Res. 2013;54(1):134–51.
Pinkosky SL, Newton RS, Day EA, Ford RJ, Lhotak S, Austin RC, et al. Liver-specific ATP-citrate lyase inhibition by bempedoic acid decreases LDL-C and attenuates atherosclerosis. Nat Commun. 2016;7:13457.
Saeed A, Ballantyne CM. Bempedoic acid (ETC-1002): a current review. Cardiol Clin. 2018;36(2):257–64.
Gutierres M, Rosenberg N, DE MD, et al. Efficacy and safety of ETC-1002, a novel investigational low-density lipoprotein-cholesterol-lowering therapy for the treatment of patients with hypercholesterolemia and type 2 diabetes mellitus. Arterioscler Thromb Vasc Biol. 2014;34:676–83.
Filippov S, Pinkosky SL, Lister RJ, Pawloski C, Hanselman JC, Cramer CT, et al. Newton RS. ETC-1002 regulates immune response, leukocyte homing, and adipose tissue inflammation via LKB1-dependent activation of macrophage AMPK. J Lipid Res. 2013;54(8):2095–108.
Cramer CT, Goetz B, Hopson KL, et al. Effects of a novel dual lipid synthesis inhibitor and its potential utility in treating dyslipidemia and metabolic syndrome. J Lipid Res. 2004;45(7):1289–301.
Ruscica M, Banach M, Sahebkar A, et al. ETC-1002 (Bempedoic acid) for the management of hyperlipidemia: from preclinical studies to phase 3 trials. Expert Opin Pharmacother. 2019;20(7):791–803.
Ference BA, Ray KK, Catapano AL, et al. Mendelian randomization study of ACLY and cardiovascular disease. N Engl J Med. 2019;380(11):1033–42.
Feng Х, Zhanga L, Xu S, et al. ATP-citrate lyase (ACLY) in lipid metabolism and atherosclerosis: an updated review. Prog Lipid Res. 2020;77:101006.
https://www.esperion.com/product/nexletol. Accessed 22 November 2020
Chu KY, Lin Y, Hendel A, et al. ATP-citrate lyase reduction mediates palmitate-induced apoptosis in pancreatic beta cells. J Biol Chem. 2010;285(42):32606–15.
Penson P, McGowan M, Banach M. Evaluating bempedoic acid for the treatment of Hyperlipidaemia. Expert Opin Investig Drugs. 2017;26(2):251–9.
Burke AC, Huff MW. ATP-citrate lyase: genetics, molecular biology and therapeutic target for dyslipidemia. Curr Opin Lipidol. 2017;28(2):193–200.
Parker BA, Capizzi JA, Grimaldi AS, et al. Effect of statins on skeletal muscle function. Circulation. 2013;127(1):96–103.
Souich P, Roederer G, Duofour R. Myotoxicity of statins: mechanism of action. Pharmacol Ther. 2017;175:1–16.
Ballantyne CM, Davidson MH, Macdougall DE, et al. Efficacy and safety of a novel dual modulator of adenosine triphosphate -citrate lyase and adenosine monophosphate-activated protein kinase in patients with hypercholesterolemia:results of a multicenter, randomized, double-blinde, placebo-controlled , parallel-group trial. J Am Coll Cardiol. 2013;62:1154–62.
Molusky MM, Hsieh J, Lee SX, et al. Metformin and AMP kinase activation increase expression of the sterol transporters ABCG5/8 (ATP-binding cassette transporter G5/G8) with potential antiatherogenic consequences. Arterioscler Thromb Vasc Biol. 2018;38(7):1493–503.
Katsiki N, Mikhailidis DP, Banach M. Lipid-lowering agents for concurrent cardiovascular and chronic kidney disease. Expert Opin Pharmacother. 2019;20(16):2007–17.
Ballantyne C, McKeney J. MacDougall et al. Effect of ETC-1002 on Serum Low-Density Lipoprotein Cholesterol in Hypercholesterolemic Patients Receiving Statin Therapy. Am J Cardiol. 2016;117:1928–33.
Lalwani ND, Hanselman JC, MacDougal DF. Complementary low-density lipoprotein-cholesterol lowering and pharmacokinetics of adding bempedoic acid (ETC-1002) to high-dose atorvastatin background therapy in hypercholesterolemic patients: a randomized placebo-controlled trial. J Clin Lipidol. 2019;13(4):568–79.
Li MN, Guo X, Bao PJ, et al. Association of genetic variations in the ACLY gene with growth traits in Chinese beef cattle. Genet Mol Res. 2016;15(2):gmr.15028250. https://doi.org/10.4238/gmr.15028250.
Bruckert E, Hayem G, Dejager S, et al. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients –the PRIMO Study. Cardiovasc. Drugs Ther. 2005;19(6):403–14.
Patel J, Marin SS, Banach M, et al. Expert opinion: the therapeutic challenges faced by statin intolerance. Exp Opinion Pharmacother. 2016;17:1497–507.
Buettner C, Davis RB, Leveille SG, et al. Prevalence of musculoskeletal pain and statin use. J Gen Intern Med. 2008;23(8):1182–6. J Gen Intern Med. 2008.1182–6.
Rosenson RS, Baker SK, Jacobson TA, Kopecky SL, Parker BA, The National Lipid Association’s Muscle Safety Expert Panel. An assessment by the Statin Muscle Safety Task Force: 2014 update. J Clin Lipidol 2014;8 Suppl:S58–71. https://doi.org/10.1016/j.jacl.2014.03.004.
Banach M, Rizzo M, Toth PP, et al. Statin intolerance: an attempt at a unified definition. Position paper from an International Lipid Expert Panel. Arch Med Sci. 2015;11(1):1–23. doi: https://doi.org/10.5114/aoms.2015.49807.
Mancini GB, Baker S, Bergeron J, et al. Diagnosis, prevention, and management of statin adverse effects and intolerance: Canadian Consensus Working Group Update. Can J Cardiol. 2016;32(7Suppl):S35–65.
Stroes ES, Thompson PD, Corsini A, et al. European Atherosclerosis Society Consensus Panel. Statin-associated muscle symptoms: impact on statin therapy-European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur Heart J. 2015;36:1012–22.
Thompson PD, Rubino J, Janik MJ, MacDougall DE, McBride SJ, Margulies JR, et al. Use of ETC-1002 to treat hypercholesterolemia in patients with statin intolerance. J Clin Lipidol. 2015;9(3):295–304.
Ballantyne CM, Banach M, Mancini GBJ, et al. Efficacy and safety of bempedoic acid added to ezetimibe in statin-intolerant patients with hypercholesterolemia: a randomized, placebo-controlled study. Atherosclerosis. 2018;277:195–203.
Laufs U, Banach M, Mancini GBJ, et al. Efficacy and safety of bempedoic acid in patients with hypercholesterolemia and statin intolerance. J Am Heart Assoc. 2019;8(7):e011662.
Ray KK, Bays HE, Catapano AL, et al. CLEAR Harmony Trial. Safety and efficacy of bempedoic acid to reduce LDL cholesterol. N Engl J Med. 2019;380(11):1022–32.
Goldberg AC, Leiter LA, Stroes ESG, et al. Effect of bempedoic acid vs placebo added to maximally tolerated statins on low-density lipoprotein cholesterol in patients at high risk for cardiovascular disease: the CLEAR Wisdom randomized clinical trial. JAMA. 2019;322(18):1780–8.
Ballantyne CM, Laufs U, Ray K, et al. Bempedoic acid plus ezetimibe fixed-dose combination in patients with hypercholesterolemia and high CVD risk treated with maximally tolerated statin therapy. European Journal of Preventive Cardiology. 2020;27(6):593–603.
Dai L, Zyo Y, You Q, et al. Efficacy and safety of bempedoic acid in patients with hypercholesterolemia: a systematic review and meta-analysis of randomized controlled trials. Eur J Prev Cardiol. 2020:2047487320930585. https://doi.org/10.1177/2047487320930585.
Wang X, Luo S, Gan X, et al. Safety and efficacy of ETC-1002 in hypercholesterolaemic patients: a meta-analysis of randomized controlled trials. Kardiologia polska. 2019;77(2):207–6.
Cicero AFG, Fogacci F, Hernandez AV, Banach M, on behalf of the Lipid and Blood Pressure Meta-Analysis Collaboration (LBPMC) Group and the International Lipid Expert Panel (ILEP). Efficacy and safety of bempedoic acid for the treatment of hypercholesterolemia: a systematic review and meta-analysis. PLoS Med. 2020;17(7):e1003121.
Masson W, Lobo M. Lavalle-Cobo et al. Effect of bempedoic acid on new onset or worsening diabetes: a meta-analysis. Diabetes Res Clin Pract. 2020, Oct;168:108369.
Cicero AFG, Pontremoli F, Fogacchi F, et al. Effect of bempedoic acid on serum uric acid and related outcomes: a systematic review and meta-analysis of the available phase 2 and phase 3 clinical studies. Drug Saf. 2020;43(8):727–36.
Yusuf S, Hawken S, Ounpuu S, Dans T, Avezum A, Lanas F, et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet. 2004;364(9438):937–52.
Baigent C, Keech A, Keraney PM. Efficacy and aafety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomized trials of statins. Lancet. 2005;366(9493):1267–78.
Cholesterol Treatment Trialists (CTT). Collaborators Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomized trials. Lancet. 2010;376:1670–81.
Fruchart J-C, Sacks FM, Hermans MP, et al. Residual risk reduction initiative: a call to action to reduce residual vascular risk in dyslipidaemic patients. Diab Vasc Dis Res. 2008;5(4):319–35.
Gitt AK, Lautsch D, Ferrières J, et al. Contemporary data on treatment practices for low-density lipoprotein cholesterol in 6794 patients with stable coronary heart disease across the world. Data Brief. 2018;18:1937–40.
Chiang C-E, Ferrieres J, Gotcheva N, et al. Suboptimal control of lipid levels: results from 29 countries participating in the centralized pan-regional surveys on the undertreatment of hypercholesterolaemia (CEPHEUS). J Atheroscler Thromb. 2016;23(5):567–87.
Kotseva K, De Backer G, De Bacquer D, et al. Primary prevention efforts are poorly developed in people at high cardiovascular risk: A report from the European Society of Cardiology EURObservational Research Programme EUROASPIRE V survey in 16 European countries. Eur J Prev Cardiol. 2020:2047487320908698. https://doi.org/10.1177/2047487320908698.
Perera K, Kam N, Ademi Z et al. Bempedoic acid for high-risk patients with CVD as adjunct lipid-lowering therapy: a cost-effectiveness analysis. J Clin Lipidology. 2020;S1933–2874(20)30259–2. doi: https://doi.org/10.1016/j.jacl.2020.08.013.
Bempedoic Acid (Nexletol) for lowering LDL-C cholesterol. Med Lett Drug Ther. 2020;62(1595):53–5.
Beigneux AP, Kosinski C, Gavino B, Horton JD, Skarnes WC, Young SG. ATP-citrate lyase deficiency in the mouse. J Biol Chem. 2004;279:9557–64.
Townsend N, Nichols M, Scarborough P, Rayner M. Cardiovascular disease in Europe–epidemiological update 2015. Eur Heart J. 2015;36(40):2696–705.
Danchin N, Almahmeed W, Al-Rasadi K, et al. Achievement of low-density lipoprotein cholesterol goals in 18 countries outside Western Europe: the International Cholesterol management Practice Study (ICLPS). Eur J Prev Cardiol. 2018;25(10):1087–94.
Cholesterol Treatment Trialists (CTT) Collaborators. The effects of lowering LDL cholesterol with statin therapy in people at low risk of vascular disease: meta-analysis of individual data from 27 randomized trials. Lancet. 2012;380:581–90.
Cholesterol Treatment Trialists (CTT). Collaborators. Efficacy and safety of LDL-lowering therapy among men and women: meta-analysis of individual data from 174,000 participants in 27 randomized trials. Lancet. 2015;385:1397–405.
Author information
Authors and Affiliations
Contributions
Andrey V Susekov: literature search, first draft of manuscript and lead editor of manuscript.
Ludmila A Korol: literature search, writing a segment on clinical pharmacology.
Gerald F Watts: general plan of writing overview, paper proof-reading and correction, writing a segment on clinical trials.
Proof-reading complete: 16 July 2020 by Proof-reading Services Ltd. UK.
English language (US) editing completed 20 Оct 2020 by Scribendi (http:// www.scribendi.com).
Corresponding author
Ethics declarations
Conflicts of Interest/Competing Interests
Andrey V Susekov: none for this manuscript.
Lecturer, honoraria: AstraZeneca, Abbott, KRKA, Akrikhin, Sanofi-Aventis, Amgen, Servier. Teva Pharmaceuticals. Clinical Trials: ESPERION 2018–2019, PI (Clear OUTCOMES), KOWA Research (PROMINENT, K-877-301, K-877-302).
Ludmila A Korol: none.
Gerald Watts: GFW has received honoraria for advisory boards and research grants from Amgen, Arrowhead, Gemphire, Kowa, Novartis, Pfizer, Sanofi and Regeneron.
Ethics Approval
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Susekov, A.V., Korol, L.A. & Watts, G.F. Bempedoic Acid in the Treatment of Patients with Dyslipidemias and Statin Intolerance. Cardiovasc Drugs Ther 35, 841–852 (2021). https://doi.org/10.1007/s10557-020-07139-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-020-07139-x