Abstract
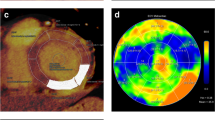
To clarify the consistency and efficiency of four methods for myocardial extracellular volume (ECV) measurement (manual method using dual-energy iodine [manual ECViodine], manual method using subtraction [manual ECVsub], automatic ECViodine, automatic ECVsub) in patients with ischaemic cardiomyopathy. Fifty patients with ischaemic cardiomyopathy who underwent coronary computed tomography angiography (CCTA) following dual-energy computed tomography (CT) with late iodine enhancement (LIE-DECT) were included. LIE with ischaemic patterns representing scarring could be detected using iodine maps in all patients. The global and remote ECVs of non-scarred myocardium were measured using four methods (manual ECViodine, automatic ECViodine, manual ECVsub, and automatic ECVsub). The consistency and time cost of the four methods were analysed. There were no significant differences in the mean global ECVs or remote ECVs among the four methods (p > 0.05). ECViodine resulted in a lower Bland–Altman limit of agreement than that of ECVsub for both global and remote measurements. Intraclass correlation coefficients of the automatic and manual ECViodine measurements demonstrated better concordance (0.804 and 0.859, respectively) than those of automatic and manual ECVsub (0.607 and 0.669, respectively) for both global and remote measurements. The measurement time for automatic ECV was less than that for manual ECV for both global and remote ECV measurements (all p < 0.001). ECV measurement using dual-energy iodine yielded good concordance, and the automatic method has the advantages of being simple and convenient, which can become a useful tool for quantification of myocardial fibrosis.




Similar content being viewed by others
Abbreviations
- CACS:
-
Coronary artery calcium scoring
- CCT:
-
Cardiac computed tomography
- CCTA:
-
Coronary computed tomography angiography
- CI:
-
Confidence interval
- CMR:
-
Cardiac magnetic resonance
- DECT:
-
Dual-energy computed tomography
- ECM:
-
Extracellular matrix
- ECV:
-
Extracellular volume
- ICC:
-
Interclass correlation coefficient
- LB:
-
Linear blending
- LGE:
-
Late gadolinium enhancement
- LIE:
-
Late iodine enhancement
- LOA:
-
Limits of agreement
References
Schelbert EB, Messroghli DR (2016) State of the art: clinical applications of cardiac T1 mapping. Radiology 278(3):658–676. https://doi.org/10.1148/radiol.2016141802
Mewton N, Liu CY, Croisille P, Bluemke D, Lima JA (2011) Assessment of myocardial fibrosis with cardiovascular magnetic resonance. J Am Coll Cardiol 57(8):891–903. https://doi.org/10.1016/j.jacc.2010.11.013
Ho CY, López B, Coelho-Filho OR et al (2010) Myocardial fibrosis as an early manifestation of hypertrophic cardiomyopathy. N Engl J Med 363(6):552–563. https://doi.org/10.1056/NEJMoa1002659
Flett AS, Sado DM, Quarta G et al (2012) Diffuse myocardial fibrosis in severe aortic stenosis: an equilibrium contrast cardiovascular magnetic resonance study. Eur Heart J Cardiovasc Imaging 13(10):819–826. https://doi.org/10.1093/ehjci/jes102
González A, Schelbert EB, Díez J, Butler J (2018) Myocardial interstitial fibrosis in heart failure biological and translational perspectives. J Am Coll Cardiol 71(15):1696–1706. https://doi.org/10.1016/j.jacc.2018.02.021
Kim RJ, Fieno DS, Parrish TB et al (1999) Relationship of MRI delayed contrast enhancement to irreversible injury, infarct age, and contractile function. Circulation 100:1992–2002. https://doi.org/10.1161/01.cir.100.19.1992
Flacke SJ, Fischer SE, Lorenz CH (2001) Measurement of the gadopentetate dimeglumine partition coefficient in human myocardium in vivo: normal distribution and elevation in acute and chronic infarction. Radiology 218:703–710. https://doi.org/10.1148/radiology.218.3.r01fe18703
Nacif MS, Kawel N, Lee JJ et al (2012) Interstitial myocardial fibrosis assessed as extracellular volume fraction with low-radiation-dose cardiac CT. Radiology 264(3):876–883. https://doi.org/10.1148/radiol.12112458
Lee HJ, Im DJ, Youn JC et al (2016) Myocardial extracellular volume fraction with dual-energy equilibrium contrast-enhanced cardiac CT in nonischemic cardiomyopathy: a prospective comparison with cardiac MR imaging. Radiology 280(1):49–57. https://doi.org/10.1148/radiol.2016151289
Emoto T, Oda S, Kidoh M et al (2021) Myocardial extracellular volume quantification using cardiac computed tomography: a comparison of the dual-energy iodine method and the standard subtraction method. Acad Radiol 28(5):e119–e126. https://doi.org/10.1016/j.acra.2020.03.019
Jablonowski R, Wilson MW, Do L, Hetts SW, Saeed M (2015) Multidetector CT measurement of myocardial extracellular volume in acute patchy and contiguous infarction: validation with microscopic measurement. Radiology 274(2):370–378. https://doi.org/10.1148/radiol.14140131
Bandula S, White SK, Flett AS et al (2013) Measurement of myocardial extracellular volume fraction by using equilibrium contrast-enhanced CT: validation against histologic findings. Radiology 269(2):396–403. https://doi.org/10.1148/radiology.13130130
Treibel TA, Bandula S, Fontana M et al (2015) Extracellular volume quantification by dynamic equilibrium cardiac computed tomography in cardiac amyloidosis. J Cardiovasc Comput Tomogr 9(6):585–592. https://doi.org/10.1016/j.jcct.2015.07.001
Han D, Tamarappoo B, Klein E et al (2021) Computed tomography angiography-derived extracellular volume fraction predicts early recovery of left ventricular systolic function after transcatheter aortic valve replacement. Eur Heart J Cardiovasc Imaging 22(2):179–185. https://doi.org/10.1093/ehjci/jeaa310
Abadia AF, van Assen M, Martin SS et al (2020) Myocardial extracellular volume fraction to differentiate healthy from cardiomyopathic myocardium using dual-source dual-energy CT. J Cardiovasc Comput Tomogr 14(2):162–167. https://doi.org/10.1016/j.jcct.2019.09.008
Qi RX, Shao J, Jiang JS et al (2021) Myocardial extracellular volume fraction quantitation using cardiac dual-energy CT with late iodine enhancement in patients with heart failure without coronary artery disease: a single-center prospective study. Eur J Radiol 140:109743. https://doi.org/10.1016/j.ejrad.2021.109743
Rockey DC, Bell PD, Hill JA (2015) Fibrosis—a common pathway to organ injury and failure. N Engl J Med 372(12):1138–1149. https://doi.org/10.1056/NEJMra1300575
Travers JG, Kamal FA, Robbins J, Yutzey KE, Blaxall BC (2016) Cardiac fibrosis: the fibroblast awakens. Circ Res 118(6):1021–1040. https://doi.org/10.1161/CIRCRESAHA.115.3065
Messroghli DR, Moon JC, Ferreira VM et al (2017) Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2*and extracellular volume: a consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imaging (EACVI). J Cardiovasc Magn Reson 19(1):75. https://doi.org/10.1186/s12968-017-0389-8
Ohta Y, Kishimoto J, Kitao S et al (2020) Investigation of myocardial extracellular volume fraction in heart failure patients using iodine map with rapid-kV switching dual-energy CT: segmental comparison with MRI T1 mapping. J Cardiovasc Comput Tomogr 14(4):349–355. https://doi.org/10.1016/j.jcct.2019.12.032
Emoto T, Kidoh M, Oda S et al (2020) Myocardial extracellular volume quantification in cardiac CT: comparison of the effects of two different iterative reconstruction algorithms with MRI as a reference standard. Eur Radiol 30(2):691–701. https://doi.org/10.1007/s00330-019-06418-y
van Assen M, De Cecco CN, Sahbaee P et al (2019) Feasibility of extracellular volume quantification using dual-energy CT. J Cardiovasc Comput Tomogr 13(1):81–84. https://doi.org/10.1016/j.jcct.2018.10.011
Oda S, Emoto T, Nakaura T et al (2019) Myocardial late iodine enhancement and extracellular volume quantification with dual-layer spectral detector dual-energy cardiac CT. Radiol Cardiothorac Imaging. 1(1):e180003. https://doi.org/10.1148/ryct.2019180003
Cerqueira MD, Weissman NJ, Dilsizian V et al (2002) Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 105(4):539–542. https://doi.org/10.1161/hc0402.102975
Ohta Y, Kitao S, Yunaga H et al (2018) Myocardial delayed enhancement CT for the evaluation of heart failure: comparison to MRI. Radiology 288(3):682–691. https://doi.org/10.1148/radiol.2018172523
Wong TC, Piehler K, Meier CG et al (2012) Association between extracellular matrix expansion quantified by cardiovascular magnetic resonance and short-term mortality. Circulation 126(10):1206–1216. https://doi.org/10.1161/CIRCULATIONAHA.111.089409
Schelbert EB, Fonarow GC, Bonow RO, Butler J, Gheorghiade M (2014) Therapeutic targets in heart failure: refocusing on the myocardial interstitium. J Am Coll Cardiol 63(21):2188–2198. https://doi.org/10.1016/j.jacc.2014.01.068
Gerber BL, Belge B, Legros GJ et al (2006) Characterization of acute and chronic myocardial infarcts by multidetector computed tomography: comparison with contrast-enhanced magnetic resonance. Circulation 113(6):823–833. https://doi.org/10.1161/CIRCULATIONAHA.104.529511
Ohta Y, Kitao S, Watanabe T et al (2017) Measurement of myocardial extracellular volume fraction from iodine density images using single-source, dual-energy computed tomography: a feasibility study. J Comput Assist Tomogr. 41(5):750–756. https://doi.org/10.1097/RCT.0000000000000587
Acknowledgements
This study was supported by Jiangsu Provincial Medical Association Roentgen imaging research project (SYH-3201150-0010, 2021005), Nantong Basic Science Research Project (JC2021195). We would like to thank all the patients and volunteers who participated in this study.
Funding
Jiangsu Provincial Medical Association Roentgen imaging research project, SYH-3201150–0010, Rong-Xing Qi, 2021005, Rong-Xing Qi, Nantong Basic Science Research Project, JC2021195, Rong-Xing Qi
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shao, J., Jiang, JS., Wang, XY. et al. Measurement of myocardial extracellular volume using cardiac dual-energy computed tomography in patients with ischaemic cardiomyopathy: a comparison of different methods. Int J Cardiovasc Imaging 38, 1591–1600 (2022). https://doi.org/10.1007/s10554-022-02532-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-022-02532-z