Abstract
Several studies have reported a correlation between right ventricular (RV) and left ventricular (LV) systolic dysfunction in adults with repaired tetralogy of Fallot (TOF). However, data are lacking regarding the relationship between RV and LV diastolic dysfunction assessed by 2-dimensional speckle-tracking echocardiography. We studied 69 adults with repaired TOF (mean age 34 years, 61% male) who had been regularly followed up and had routinely undergone echocardiography. In addition to conventional echocardiography, global longitudinal strain (GLS) and early diastolic strain rate (SRe) of both ventricles were assessed using 2-dimensional speckle-tracking echocardiography. Results were compared with 30 age- and sex-matched controls. RV and LV GLS were decreased in TOF patients compared with controls (− 18.4 ± 3.3% vs. −23.5 ± 4.2%, p < 0.001 and − 16.0 ± 3.8% vs. −20.0 ± 3.0%, p < 0.001, respectively). RV and LV SRe were also decreased in TOF patients compared with controls (1.22 ± 0.34 sec− 1 vs. 1.47 ± 0.41 sec− 1, p = 0.003 and 1.29 ± 0.42 sec− 1 vs. 1.63 ± 0.42 sec− 1, p < 0.001, respectively). A correlation between RV and LV SRe was found in TOF patients (r = 0.43, p < 0.001) as well as between RV and LV GLS (r = 0.45, p < 0.001). Two-dimensional speckle-tracking echocardiography reveals subclinical RV and LV diastolic dysfunction in adults with repaired TOF. A correlation is observed between RV and LV diastolic dysfunction as well as between RV and LV systolic dysfunction.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Tetralogy of Fallot (TOF) is the most common cyanotic congenital heart disease [1]. Early surgical repair has dramatically improved long-term outcomes, and more than 90% of patients reach adulthood after repair of TOF [2, 3]. The right side of the heart has historically been paid attention in the management of patients late after repair of TOF [4,5,6].
However, recent studies have found that left ventricular (LV) systolic dysfunction is also associated with impaired clinical status in patients with repaired TOF [7] and is an independent predictor of adverse clinical events [8,9,10]. Decreased LV ejection fraction has been reported in 21% of patients with repaired TOF [11], and a study using speckle-tracking echocardiography revealed that subclinical LV systolic dysfunction was present even in TOF patients with normal LV ejection fraction [12]. Several studies have reported a correlation between right ventricular (RV) and LV systolic dysfunction in patients with repaired TOF, indicating ventricular interaction in which RV systolic dysfunction adversely affects LV systolic function [7, 12,13,14].
LV diastolic dysfunction in patients with repaired TOF have also been investigated [15,16,17,18,19]; however, data are lacking regarding the relationship between RV and LV diastolic dysfunction assessed by 2-dimensional speckle-tracking echocardiography. We hypothesized that there would also be a correlation between subtle RV and LV diastolic dysfunction in adults with repaired TOF. This study investigated the relationship between RV and LV diastolic dysfunction in addition to the relationship between RV and LV systolic dysfunction in asymptomatic adults with repaired TOF using 2-dimensional speckle-tracking echocardiography.
Methods
Study population
This was a single-center, retrospective, cross-sectional study. The study included asymptomatic adults (age ≥ 15 years) with repaired TOF, who had received regular follow-up at Congenital Heart Disease Center, Tenri Hospital (Tenri, Japan) and had routinely undergone echocardiography between January 2012 and August 2018. We used an echocardiographic database to screen for eligible patients with repaired TOF. Only echocardiography performed with Vivid 7 or Vivid E9 machine (GE Healthcare, Horten, Norway) was included to avoid variability in strain measurements between different vendors. For those who had undergone several echocardiographic examinations with the GE machines during this period, the most recent one was used for analysis. Exclusion criteria were as follows: echocardiography without the entire RV free wall in the apical 4-chamber view, poor image quality for speckle-tracking analysis, and patients who were not in sinus rhythm. Echocardiographic data were compared with those of 30 age- and sex-matched normal controls.
This study was conducted in accordance with the Declaration of Helsinki. The institutional ethics committee at Tenri Hospital approved the protocol (Approval number 1072). Written informed consent was waived because of the retrospective nature of the study.
Conventional echocardiography
All echocardiography was performed by experienced sonographers according to standardized protocols using Vivid 7 or Vivid E9 machines (GE Healthcare). Images were acquired from standard parasternal and apical views and digitally stored. Measurements included RV end-diastolic and end-systolic area, RV fractional area change, right atrial end-systolic area, LV end-diastolic and end-systolic dimeter, LV ejection fraction (using biplane Simpson’s method), LV wall thickness, left atrial volume, and tricuspid annular plane systolic excursion (TAPSE). Severity of pulmonary regurgitation (PR) was graded by jet width, length, and duration using Doppler echocardiography according to the American Society of Echocardiography (ASE) and the European Association of Cardiovascular Imaging recommendations [20]. Severity of pulmonary stenosis (PS) was graded by peak velocity of the jet across the narrowing lesion. Peak velocity of more than 4 m/s and 3–4 m/s were considered severe PS and moderate PS, respectively, according to current American College of Cardiology/American Heart Association guidelines [21]. Doppler measurements also included transmitral flow velocity ratio (E/A), diastolic tricuspid (tricuspid E′) and mitral lateral annular velocity (mitral E′).
Speckle-tracking echocardiography
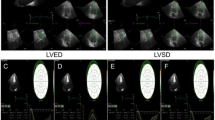
RV and LV strain and strain rate were analyzed offline using 2-dimensional speckle-tracking analysis software (EchoPAC version 113.1.0; GE Healthcare). The LV algorithm was used for both ventricles. The endocardial border was traced in the apical 4-chamber view and the width of the region of interest was adjusted. Myocardial tracking was confirmed visually and verified with the software. The endocardial border was retraced if myocardial tracking was inadequate. Peak systolic longitudinal strain was defined as the peak negative value on the curve. As in LV global longitudinal strain (GLS) analysis, RV GLS was defined as the average of peak systolic longitudinal strain of all segments of the right ventricle including the ventricular septum [22]. RV and LV early diastolic strain rate (SRe) were defined as the average of peak early diastolic longitudinal strain rate of all segments of the right and left ventricle, respectively. All images were recorded at a frame rate of more than 50 frames/sec. Representative GLS and SRe curves for both ventricles in a patient with repaired TOF are shown in Fig. 1.
Two-dimensional speckle-tracking echocardiography in the apical 4-chamber view. White dotted lines indicate GLS or global longitudinal strain rate (average of all segments). White arrow indicates RV GLS (a), LV GLS (b), RV SRe (c), LV SRe (d). GLS global longitudinal strain, LV left ventricular, RV right ventricular, SRe early diastolic strain rate
Statistical analysis
Categorical variables were presented as numbers and percentages and compared using the chi-squared test. Continuous variables were expressed as mean and standard deviation or median and interquartile range and compared using the Student's t-test or Wilcoxon rank sum test based on their distributions. Relationships between two continuous variables were assessed using Pearson’s correlation analysis. Variables which were not normally distributed were log transformed. Intra- and interobserver reproducibility for conventional and speckle-tracking echocardiographic parameters of both ventricles were assessed by calculating interclass correlation coefficient in 20 randomly selected patients with repaired TOF by the same observer and two different observers, respectively. All statistical analyses were conducted with SPSS software version 22.0 (IBM Corp., Armonk, New York). All reported p values were two-tailed, and p < 0.05 were considered statistically significant.
Results
Study population and feasibility of echocardiographic measurements
A total of 113 adult patients with repaired TOF were identified to be potentially eligible for enrollment using the echocardiographic database. Among them, RV speckle-tracking analysis was not feasible in 44 patients because the entire RV wall was not recorded in the apical 4-chamber view or image quality was too poor to perform adequate myocardial tracking. LV speckle-tracking analysis was not feasible in 21 patients because of poor image quality. The feasibility of RV speckle-tracking analysis was 61%, which was lower than that of measuring TAPSE (86%, p = 0.004) and tricuspid E′ (88%, p = 0.007). The feasibility of LV speckle-tracking analysis was 81%, which was also lower than that of measuring LV EF (88%, p < 0.001) and mitral E′ (94%, p = 0.010). This study finally included 69 TOF patients in which biventricular speckle-tracking analysis was feasible. Sixty-three patients had undergone intracardiac repair at our hospital; the remaining 6 patients had undergone this surgery at other hospitals.
Patients characteristics
Demographic and clinical characteristics are presented in Table 1. Median age at initial intracardiac repair was 39 months. Five patients (7%) underwent initial repair at the age of ≥ 15 years. Median time from initial intracardiac repair was 30 years. One patient underwent pulmonary valve replacement at initial repair and 11 patients underwent pulmonary valve replacement after repair. QRS duration was more prolonged in patients with repaired TOF than controls.
Conventional echocardiography
Conventional echocardiographic findings are presented in Table 2. RV end-diastolic area and right atrial end-systolic area were larger, and LV end-diastolic diameter was smaller in patients with repaired TOF than controls. Five patients (7%) had decreased RV fractional area change (< 35%). One patient (1.4%) had moderately decreased LV ejection fraction (< 45%), 10 patients (14%) had mildly decreased LV ejection fraction (< 55%).
Global longitudinal strain
RV and LV GLS were significantly decreased in patients with repaired TOF compared with controls (− 18.4 ± 3.3% vs. −23.5 ± 4.2%, p < 0.001 and − 16.0 ± 3.8% vs. −20.0 ± 3.0%, p < 0.001, respectively; Fig. 2a).
RV and LV GLS did not significantly correlate with age at time of imaging (r = 0.14, p = 0.25 and r = 0.16, p = 0.19, respectively) or log-transformed age at repair (r = 0.11, p = 0.38 and r = 0.02, p = 0.88, respectively). RV and LV GLS were not significantly different between TOF patients with and without moderate to severe PR (− 18.8 ± 3.0% vs. −18.1 ± 3.5%, p = 0.39 and − 15.5 ± 3.6% vs. −16.3 ± 3.9%, p = 0.43, respectively) and between those with and without moderate to severe PS (− 18.1 ± 3.7% vs. −18.4 ± 3.3%, p = 0.76 and − 16.6 ± 4.6% vs. −15.9 ± 3.6%, p = 0.57, respectively). RV and LV GLS correlated with QRS duration in patients with repaired TOF (r = 0.24, p = 0.045 and r = 0.41, p < 0.001, respectively). RV GLS correlated with TAPSE (r = − 0.43, p < 0.001) and LV GLS correlated with LV EF (r = − 0.41, p = 0.002) in patients with repaired TOF.
A correlation between RV GLS and LV GLS was found in patients with repaired TOF (r = 0.45, p < 0.001; Fig. 3a) as with controls (r = 0.55, p = 0.002).
Early diastolic strain rate
RV and LV SRe were significantly decreased in patients with repaired TOF compared with controls (1.22 ± 0.34 sec− 1 vs. 1.47 ± 0.41 sec− 1, p = 0.003 and 1.29 ± 0.42 sec− 1 vs. 1.63 ± 0.42 sec− 1, p < 0.001, respectively; Fig. 2b).
RV and LV SRe correlated with age at time of imaging (r = − 0.39, p < 0.001 and r = − 0.46, p < 0.001, respectively) and log-transformed age at repair (r = − 0.31, p = 0.012 and r = − 0.30, p = 0.012, respectively). RV and LV SRe were not significantly different between TOF patients with and without moderate to severe PR (1.29 ± 0.31% vs. 1.19 ± 0.35%, p = 0.26 and 1.32 ± 0.47% vs. 1.27 ± 0.40%, p = 0.64, respectively) and between those with and without moderate to severe PS (1.18 ± 0.31% vs. 1.24 ± 0.34%, p = 0.59 and 1.33 ± 0.40% vs. 1.28 ± 0.43%, p = 0.71, respectively). RV and LV SRe correlated with QRS duration in patients with repaired TOF (r = − 0.31, p = 0.010 and r = − 0.25, p = 0.038, respectively). RV SRe correlated with tricuspid E′ (r = 0.51, p < 0.001) and LV SRe correlated with mitral E′ (r = 0.46, p < 0.001) in patients with repaired TOF.
A correlation between RV SRe and LV SRe was found in patients with repaired TOF (r = 0.43, p < 0.001; Fig. 3b) as with controls (r = 0.59, p < 0.001).
Reproducibility
The intra- and interobserver interclass correlation coefficient were 0.92 (95% confidence interval [CI] 0.82–0.97) and 0.86 (95% CI 0.55–0.95) for TAPSE, respectively, and 0.97 (95% CI 0.92–0.99) and 0.91 (95% CI 0.79–0.96) for LV EF, respectively. Reproducibility of tricuspid and mitral E′ could not be investigated because video clips of tissue Doppler imaging are not routinely stored at our hospital. The interclass correlation coefficient for GLS and SRe of both ventricles are presented in Table 3. All parameters showed good intra- and interobserver reproducibility with interclass correlation coefficients of > 0.8.
Discussion
This study investigated subclinical RV and LV systolic and diastolic dysfunction in adults with repaired TOF using 2-dimensional speckle-tracking echocardiography. The main results were as follows: (1) 2-dimensional speckle-tracking echocardiography was able to reveal a decrease in RV and LV SRe in asymptomatic adults with repaired TOF compared with controls; (2) a correlation was observed between RV and LV SRe as well as between RV and LV GLS.
Recently, SRe has been proposed as a more accurate marker of myocardial relaxation than conventional tissue Doppler echocardiography [23, 24]. Strain rates are not angle dependent, not affected by myocardial tethering, and are less affected by loading condition [23]. Its usefulness for assessing LV diastolic function has been reported in general population [25], patients with valvular heart disease [26], and patients with coronary artery disease [27]. In our study, despite a decrease in LV SRe in patients with repaired TOF, left atrial volume, mitral E/A, and mitral E′ were not significantly different between patients with repaired TOF and controls, and mitral E/E′ in patients with repaired TOF was slightly increased but remained within normal range. A previous study also showed that Doppler measurements were not associated with elevated LV end-diastolic pressure (> 12 mmHg) in patients with repaired TOF [19]. These results suggest that LV SRe decreases at an earlier stage of LV diastolic dysfunction, in which conventional echocardiographic parameters do not yet indicate the presence of LV diastolic dysfunction. Thus, LV SRe appears to be a more sensitive marker to detect subtle LV diastolic dysfunction in patients with repaired TOF.
There are no established parameters regarding assessment of RV diastolic dysfunction in patients with repaired TOF. In our study, tricuspid E′ was decreased in patients with TOF. However, previous studies have reported that conventional parameters using tricuspid flow velocities and tricuspid annular velocities were not associated with RV end-diastolic pressure in children with repaired TOF [28, 29]. Furthermore, the ASE guidelines for imaging in repaired TOF state that Doppler parameters of tricuspid inflow are not reliable for assessment of RV diastolic function [30]. Meanwhile, speckle-tracking analysis has recently become feasible in not only the left ventricle but also the right ventricle [22]. Our study suggests that RV SRe has a potential to become a novel marker of RV diastolic function in patients with repaired TOF.
The mechanism of LV diastolic dysfunction in adults with repaired TOF is not fully understood; in particular, data on its relationship with RV diastolic dysfunction assessed by 2-dimensional speckle-tracking echocardiography are lacking. A previous study investigated RV and LV diastolic dysfunction in children (mean age 12 years) with repaired TOF using speckle-tracking echocardiography [17]. This previous study demonstrated that RV SRe in children with repaired TOF was decreased compared with controls, whereas LV SRe was not significantly different. Based on this previous study and our results, we speculate that RV diastolic dysfunction has a long-term adverse effect on LV diastolic function after repair of TOF.
Our study suggests that ventricular interaction plays a role in diastolic dysfunction, as has previously been reported for systolic dysfunction. Recent studies with cardiac magnetic resonance in patients with repaired TOF found a correlation between RV and LV extracellular volume fraction [31] and between RV and LV native T1 value [32], suggesting that the degree of LV fibrosis correlates with that of RV fibrosis. These results can explain impairment of both RV and LV contractility and relaxation, and correlation of systolic and diastolic dysfunction between the right and left ventricles, as shown in our study.
The mechanism of ventricular interaction remains unclear. Mechanical coupling may provide an explanation. The right and left ventricles share a continuous myocardial band [33]. Impaired RV myocardial performance may adversely affect LV myocardial performance through this shared myocardial band. Neurohormonal coupling may also provide an explanation for ventricular interaction. Neurohumoral activation or inhibition has been reported to alter myocardial relaxation and stiffness [34], and neurohormonal activation has been reported to exist in adults with congenital heart disease [35]. Genetic factors have been reported to be associated with RV dysfunction in patients with TOF [36]. Given that TOF is a congenital disease, genetic factors may also influence LV remodeling as with RV remodeling.
Limitations
This study has several limitations. First, this is a retrospective study, therefore there is a potential for selection bias. Not all echocardiographic images of eligible patients from the echocardiographic database could be analyzed due to the inability to track the RV myocardium. Second, this study has demonstrated low feasibility of speckle-tracking echocardiography for the assessment of RV function in patients with repaired TOF. However, this limitation may be resolved if more attention is paid to acquisition of good-quality images of the entire right ventricle. Third, all patients in the study were clinically stable and did not undergo cardiac catheterization around the time of echocardiography; therefore, we could not directly compare the accuracy of SRe and E′ using catheter-derived references such as relaxation time constant (Tau), peak negative dP/dt, or end-diastolic pressure. Fourth, RV assessment by 2-dimensional speckle-tracking echocardiography did not include RV outflow tract function. Previous studies have suggested that RV outflow tract systolic dysfunction is related to impairment of RV performance in patients with repaired TOF [37, 38]. RV outflow tract may also contribute to RV diastolic function, although RV outflow tract is a relatively small portion compared to the inlet and trabecular portion. Fifth, the subgroup analysis by PR and PS showed no significant difference in GLS and SRe. This may be due to a lack of power because of small sample size. Finally, because this is a cross-sectional study, we could not demonstrate association between subclinical diastolic dysfunction detected by 2-dimensional speckle-tracking echocardiography and long-term clinical outcomes. Further research is needed to assess whether decreased RV and LV SRe are associated with poor clinical outcomes.
Conclusions
Two-dimensional speckle-tracking echocardiography reveals subclinical RV and LV diastolic dysfunction in adults with repaired TOF. A correlation is observed between RV and LV diastolic dysfunction as well as between RV and LV systolic dysfunction in adults with repaired TOF.
Change history
23 December 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10554-021-02473-z
References
Apitz C, Webb GD, Redington AN (2009) Tetralogy of Fallot. Lancet 374:1462–1471
Murphy JG, Gersh BJ, Mair DD, Fuster V, McGoon MD, Ilstrup DM et al (1993) Long-term outcome in patients undergoing surgical repair of tetralogy of Fallot. N Engl J Med 329:593–599
Nollert G, Fischlein T, Bouterwek S, Böhmer C, Klinner W, Reichart B (1997) Long-term survival in patients with repair of tetralogy of Fallot: 36-year follow-up of 490 survivors of the first year after surgical repair. J Am Coll Cardiol 30:1374–1383
Frigiola A, Redington AN, Cullen S, Vogel M (2004) Pulmonary regurgitation is an important determinant of right ventricular contractile dysfunction in patients with surgically repaired tetralogy of Fallot. Circulation 110:II153–I157
Marie PY, Marçon F, Brunotte F, Briançon S, Danchin N, Worms AM et al (1992) Right ventricular overload and induced sustained ventricular tachycardia in operatively “repaired” tetralogy of Fallot. Am J Cardiol 69:785–789
Gatzoulis MA (2000) Risk factors for arrhythmia and sudden cardiac death late after repair of tetralogy of Fallot: a multicentre study. Lancet 356:975–981
Geva T, Sandweiss BM, Gauvreau K, Lock JE, Powell AJ (2004) Factors associated with impaired clinical status in long-term survivors of tetralogy of Fallot repair evaluated by magnetic resonance imaging. J Am Coll Cardiol 43:1068–1074
Ghai A, Silversides C, Harris L, Webb GD, Siu SC, Therrien J (2002) Left ventricular dysfunction is a risk factor for sudden cardiac death in adults late after repair of tetralogy of Fallot. J Am Coll Cardiol 40:1675–1680
Knauth AL, Gauvreau K, Powell AJ, Landzberg MJ, Walsh EP, Lock JE et al (2008) Ventricular size and function assessed by cardiac MRI predict major adverse clinical outcomes late after tetralogy of Fallot repair. Heart 94:211–216
Diller GP, Kempny A, Liodakis E, Alonso-Gonzalez R, Inuzuka R, Uebing A et al (2012) Left ventricular longitudinal function predicts life-threatening ventricular arrhythmia and death in adults with repaired tetralogy of Fallot. Circulation 125:2440–2446
Broberg CS, Aboulhosn J, Mongeon FP, Kay J, Valente AM, Khairy P et al (2011) Prevalence of left ventricular systolic dysfunction in adults with repaired tetralogy of Fallot. Am J Cardiol 107:1215–1220
Kempny A, Diller GP, Orwat S, Kaleschke G, Kerckhoff G, Bunck, ACh et al (2012) Right ventricular-left ventricular interaction in adults with Tetralogy of Fallot: a combined cardiac magnetic resonance and echocardiographic speckle tracking study. Int J Cardiol 154:259–264
Davlouros PA, Kilner PJ, Hornung TS, Li W, Francis JM, Moon JC et al (2002) Right ventricular function in adults with repaired tetralogy of Fallot assessed with cardiovascular magnetic resonance imaging: detrimental role of right ventricular outflow aneurysms or akinesia and adverse right-to-left ventricular interaction. J Am Coll Cardiol 40:2044–2052
Menting ME, van den Bosch AE, McGhie JS, Eindhoven JA, Cuypers JA, Witsenburg M et al (2015) Assessment of ventricular function in adults with repaired Tetralogy of Fallot using myocardial deformation imaging. Eur Heart J Cardiovasc Imaging 16:1347–1357
Schwartz MC, Rome JJ, Gillespie MJ, Whitehead K, Harris MA, Fogel MA et al (2012) Relation of left ventricular end diastolic pressure to right ventricular end diastolic volume after operative treatment of tetralogy of Fallot. Am J Cardiol 109:417–422
Ahmad N, Kantor PF, Grosse-Wortmann L, Seller N, Jaeggi ET, Friedberg MK et al (2012) Influence of RV restrictive physiology on LV diastolic function in children after tetralogy of Fallot repair. J Am Soc Echocardiogr 25:866–873
Friedberg MK, Fernandes FP, Roche SL, Grosse-Wortmann L, Manlhiot C, Fackoury C et al (2012) Impaired right and left ventricular diastolic myocardial mechanics and filling in asymptomatic children and adolescents after repair of tetralogy of Fallot. Eur Heart J Cardiovasc Imaging 13:905–913
Aboulhosn JA, Lluri G, Gurvitz MZ, Khairy P, Mongeon FP, Kay J et al (2013) Left and right ventricular diastolic function in adults with surgically repaired tetralogy of Fallot: a multi-institutional study. Can J Cardiol 29:866–872
Lubert AM, Cotts TB, Zampi JD, Yu S, Norris MD (2019) Echocardiographic predictors of elevated left ventricular end diastolic pressure in adolescent and adult patients with repaired tetralogy of Fallot. Cardiol Young 29:1020–1024
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L et al (2015) Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 28:1–39
Stout KK, Daniels CJ, Aboulhosn JA, Bozkurt B, Broberg CS, Colman JM et al (2019) 2018 AHA/ACC Guideline for the Management of Adults With Congenital Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 139:e698–e800
Morris DA, Krisper M, Nakatani S, Köhncke C, Otsuji Y, Belyavskiy E et al (2017) Normal range and usefulness of right ventricular systolic strain to detect subtle right ventricular systolic abnormalities in patients with heart failure: a multicentre study. Eur Heart J Cardiovasc Imaging 18:212–223
Wang J, Khoury DS, Thohan V, Torre-Amione G, Nagueh SF (2007) Global diastolic strain rate for the assessment of left ventricular relaxation and filling pressures. Circulation 115:1376–1383
Dokainish H, Sengupta R, Pillai M, Bobek J, Lakkis N (2008) Usefulness of new diastolic strain and strain rate indexes for the estimation of left ventricular filling pressure. Am J Cardiol 101:1504–1509
Lassen MCH, Biering-Sørensen SR, Olsen FJ, Skaarup KG, Tolstrup K, Qasim AN et al (2019) Ratio of transmitral early filling velocity to early diastolic strain rate predicts long-term risk of cardiovascular morbidity and mortality in the general population. Eur Heart J 40:518–525
Dahl JS, Barros-Gomes S, Videbæk L, Poulsen MK, Issa IF, Carter-Storch R et al (2016) Early Diastolic Strain Rate in Relation to Systolic and Diastolic Function and Prognosis in Aortic Stenosis. JACC Cardiovasc Imaging 9:519–528
Ersbøll M, Andersen MJ, Valeur N, Mogensen UM, Fakhri Y, Thune JJ et al (2014) Early diastolic strain rate in relation to systolic and diastolic function and prognosis in acute myocardial infarction: a two-dimensional speckle-tracking study. Eur Heart J 35:648–656
Hayabuchi Y, Sakata M, Ohnishi T, Inoue M, Kagami S (2013) Ratio of early diastolic tricuspid inflow to tricuspid lateral annulus velocity reflects pulmonary regurgitation severity but not right ventricular diastolic function in children with repaired Tetralogy of Fallot. Pediatr Cardiol 34:1112–1117
DiLorenzo M, Hwang WT, Goldmuntz E, Ky B, Mercer-Rosa L (2018) Diastolic dysfunction in tetralogy of Fallot: Comparison of echocardiography with catheterization. Echocardiography 35:1641–1648
Valente AM, Cook S, Festa P, Ko HH, Krishnamurthy R, Taylor AM et al (2014) Multimodality imaging guidelines for patients with repaired tetralogy of Fallot: a report from the American Society of Echocardiography: developed in collaboration with the Society for Cardiovascular Magnetic Resonance and the Society for Pediatric Radiology. J Am Soc Echocardiogr 27:111–141
Chen CA, Dusenbery SM, Valente AM, Powell AJ, Geva T (2016) Myocardial ECV Fraction Assessed by CMR Is Associated With Type of Hemodynamic Load and Arrhythmia in Repaired Tetralogy of Fallot. JACC Cardiovasc Imaging 9:1–10
Cochet H, Iriart X, Allain-Nicolaï A, Camaioni C, Sridi S, Nivet H et al (2019) Focal scar and diffuse myocardial fibrosis are independent imaging markers in repaired tetralogy of Fallot. Eur Heart J Cardiovasc Imaging 20:990–1003
Torrent-Guasp F, Ballester M, Buckberg GD, Carreras F, Flotats A, Carrió I et al (2001) Spatial orientation of the ventricular muscle band: physiologic contribution and surgical implications. J Thorac Cardiovasc Surg 122:389–392
Zile MR, Brutsaert DL (2002) New concepts in diastolic dysfunction and diastolic heart failure: Part II: causal mechanisms and treatment. Circulation 105:1503–1508
Bolger AP, Sharma R, Li W, Leenarts M, Kalra PR, Kemp M (2002) Neurohormonal activation and the chronic heart failure syndrome in adults with congenital heart disease. Circulation 106:92–99
Jeewa A, Manickaraj AK, Mertens L, Manlhiot C, Kinnear C, Mondal T et al (2012) Genetic determinants of right-ventricular remodeling after tetralogy of Fallot repair. Pediatr Res 72:407–413
Uebing A, Gibson DG, Babu-Narayan SV, Diller GP, Dimopoulos K, Goktekin O et al (2007) Right ventricular mechanics and QRS duration in patients with repaired tetralogy of Fallot: implications of infundibular disease. Circulation 116:1532–1539
van der Hulst AE, Roest AA, Delgado V, Holman ER, de Roos A, Blom NA et al (2011) Relationship between temporal sequence of right ventricular deformation and right ventricular performance in patients with corrected tetralogy of Fallot. Heart 97:231–236
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised due to a retrospective Open Access order.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Miyake, M., Abe, R., Matsutani, H. et al. Relationship between right and left ventricular diastolic dysfunction assessed by 2-dimensional speckle-tracking echocardiography in adults with repaired tetralogy of Fallot. Int J Cardiovasc Imaging 37, 569–576 (2021). https://doi.org/10.1007/s10554-020-02045-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-020-02045-7