Abstract
Purpose
Androgen receptor (AR) expression is absent in 40–90% of estrogen receptor (ER)-negative breast cancers. The prognostic value of AR in ER-negative patients and therapeutic targets for patients absent in AR remains poorly explored.
Methods
We used an RNA-based multigene classifier to identify AR-low and AR-high ER-negative participants in the Carolina Breast Cancer Study (CBCS; N = 669) and The Cancer Genome Atlas (TCGA; N = 237). We compared AR-defined subgroups by demographics, tumor characteristics, and established molecular signatures [PAM50 risk of recurrence (ROR), homologous recombination deficiency (HRD), and immune response].
Results
AR-low tumors were more prevalent among younger (RFD = + 10%, 95% CI = 4% to 16%) participants in CBCS and were associated with HER2 negativity (RFD = − 35%, 95% CI = − 44% to − 26%), higher grade (RFD = + 17%, 95% CI = 8% to 26%), and higher risk of recurrence scores (RFD = + 22%, 95% CI = 16.1% to 28%), with similar results in TCGA. The AR-low subgroup was strongly associated with HRD in CBCS (RFD = + 33.3%, 95% CI = 23.8% to 43.2%) and TCGA (RFD = + 41.5%, 95% CI = 34.0% to 48.6%). In CBCS, AR-low tumors had high adaptive immune marker expression.
Conclusion
Multigene, RNA-based low AR expression is associated with aggressive disease characteristics as well as DNA repair defects and immune phenotypes, suggesting plausible precision therapies for AR-low, ER-negative patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Androgen receptor (AR), which is expressed in approximately 30–60% of estrogen receptor (ER)-/progesterone receptor(PR)-/human epidermal growth factor receptor 2(HER2)+ breast cancers and 10–53% of triple-negative breast cancers (ER-/PR-/HER2-; TNBCs) [1,2,3,4], has emerged as a candidate therapeutic target for breast cancer patients that lack ER, PR, or HER2 positivity by immunohistochemistry (IHC). Phase 2 clinical trials of AR-targeted therapies such as bicalutamide, abiraterone acetate, and enzalutamide resulted in some clinical benefit [5,6,7]. However, a large subset (40–90%) of ER-negative patients lack AR expression, and thus are exempt from benefiting from AR-targeted therapies. Furthermore, the prognostic value of AR in ER-negative breast cancer remains uncertain due to conflicting results from multiple studies. Some studies have reported that among TNBCs, lack of AR expression is associated with higher grade, stage, mitotic index, Ki-67, lymph node involvement, younger age at diagnosis, and shorter overall, disease-free, and recurrence-free survival, whereas other groups have reported opposing or no associations with these variables [3, 8,9,10,11,12,13,14,15]. This discordance may be due to differences in the populations studied and to technical factors, including sample procurement, AR antibodies, and cutoffs used for immunohistochemistry, staining protocols, AR production of constitutively active splice variants, and differences in cellular localization [16, 17].
Several research groups have reported that non-luminal androgen receptor TNBC molecular subtypes that express AR protein at low levels demonstrate AR dependence for tumor cell growth or viability [18,19,20,21]. Thus, AR IHC may not discern the full range of AR-low states. RNA-based methods allow assessment in large population-based studies with other pathway data. Given the high relative frequency of ER-negative and aggressive tumors in Black women, investigations of AR in diverse populations are needed.
The Carolina breast cancer study (CBCS; phase 1: 1993–1996, 2: 1996–2001; 3: 2008–2013) is a population-based study of breast cancer that is oversampled for Black and younger women. Using gene expression data for 1202 CBCS participants, we trained a pathway-based classifier to identify AR-low patients and to examine the relationship between AR status and tumor aggressiveness among ER-negative participants. Results in CBCS were validated in TCGA.
Methods
Study population
The Carolina breast cancer study (CBCS) is a population-based study [22, 23] of women between the ages of 20 and 74 residing in a 24 (CBCS Phases 1 and 2) or 44 (CBCS Phase 3) counties of North Carolina, all of whom were diagnosed with primary invasive breast cancer. Participants were identified through rapid case ascertainment. Black women and women under age 50 were oversampled to achieve a final sample population with approximately 50% Black women and 50% younger women. Race was self-reported. Given that fewer than 2% of the study’s non-Black participants self-identified as a race other than non-Hispanic white, we dichotomized race as Black and non-Black for this study; sensitivity analyses excluding participants self-identifying as something other than Black or white did not change results so we retained these participants to maximize power. Clinical tumor characteristics, including stage, grade, and hormone receptor status (ER and PR), were extracted from medical records and pathology reports. ER and PR status were dichotomized as positive (>1% IHC) or negative (≤1%) in accordance with clinical guidelines, though in sensitivity analyses, we also explored using a 10% cutpoint given that ER-borderline tumors have some features in common with ER negatives [24].
The Cancer Genome Atlas (TCGA) is a large, publicly available data source containing extensive genomic data on over 30 cancer types. Study details are described elsewhere [25]. For our analyses, we downloaded clinical, RNA sequencing, and reverse phase protein array (RPPA) data for 1095 primary breast cancer cases with available RNA data from the NCI Genomic Data Commons (GDC, https://gdc.cancer.gov/). In comparison to cases in CBCS, those in TCGA were older, with larger, higher stage tumors [26].
Dichotomizing of androgen receptor (AR) RNA expression
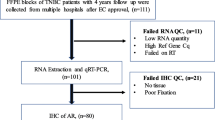
1649 CBCS tumor samples, chosen based on the availability of cores or slides for analysis, were included on a NanoString RNA panel that included AR, of which 1202 (72%) passed quality control. Samples which failed quality control were more likely to be from CBCS Phase I or II, while included samples were more likely to be from older participants (Mage = 50 vs 52 years), Black women, and ER- tumors (additional details shown in Supplemental Figure 1). Thus, AR RNA expression (alongside that of 416 other RNA-based targets) was profiled in 1202 CBCS samples (472 ER-negatives), using a custom NanoString protocol optimized for formalin-fixed paraffin-embedded (FFPE) samples [27,28,29]. To accommodate potential study-specific variability in RNA quantification and to address lack of a priori guidelines for categorizing AR expression, cut-points for AR were determined using a mixture model approach among ER-negative breast cancers. Specifically, we estimated 10 Gaussian mixture models, corresponding to the existence of one to nine distinct categories of AR expression, of AR expression values among the 472 ER-negative (absent in ER expression) samples with complete AR data, then selected the final classification model with the lowest BIC [30]. The optimal solution yielded two groups, corresponding to classes with low and high AR expression. We also explored classifications restricted to different indicators of tumor aggressiveness (i.e., age less than 50 years, pre-menopausal status) but found that categorizations restricted to ER-negative samples were a better fit to the data and thus proceeded with an ER/AR-based model (BIC ER = − 1740; BIC Age: − 2474; BIC menopause: − 2330).
To identify demographic and clinical features of AR-low tumors, we calculated relative frequency differences (RFDs) and 95% confidence intervals by fitting a generalized linear model with binomial distribution and identity link where AR status was the outcome and the variable of interest was the predictor. Because triple-negative status may confound associations between AR and clinical presentation, we also computed models adjusted for triple-negative status and in triple-negative tumors only. As further sensitivity analyses, we assessed demographic and clinical characteristics of ER+ tumors by AR status to further explore whether AR-low phenotype associations also held for ER+ tumors, and used multiple imputation to assess whether missing data biased results.
Building a classifier of AR status
To understand overlap between AR-phenotype and deficiencies in immune and DNA repair processes, we analyzed CBCS samples that had RNA expression data on both AR and immune or AR and DNA repair classes. However, because a relatively small number of samples profiled for AR also included information on RNA expression of DNA repair genes (N = 674; 271 ER-negative), we developed a predictor of AR phenotype to identify additional samples with low AR expression. To do this, we split the 472 ER-negative samples with measured AR expression into five groups (“folds”) using stratified random sampling, therefore, ensuring consistent distribution of AR-high and AR-low samples within each fold. In each of five iterations of testing, we retained four of the folds for training (training set, N = 375–379) and omitted the last for validation (test set, N = 93–97), then repeatedly fit Classification to Nearest Centroid (ClaNC) models that used between two and 150 genes (75 models, increasing the number of genes by two each time) to distinguish AR-low and AR-high tumors in the training set. For each of these 75 models, we estimated sensitivity, specificity, and the Youden’s index (sensitivity + specificity − 1) in the training and test sets. We selected the final number of genes to use in the classifier by finding the maximum Youden’s index, averaged across the five folds, among the training sets. From this final model, we predicted the AR status of all ER-negative samples, using predictions to calculate a final sensitivity and specificity. We also assessed model performance by conducting a principal component analysis of RNA expression for the selected genes, coloring samples by AR status to visually inspect how gene expression patterns correlated with AR. Finally, we applied the AR classifier to all ER-negative CBCS samples assayed for selected genes (N = 669) and proceeded to compare AR phenotypes to other molecular indicators, described below.
Associations with molecular signatures
Using custom panels of 50 immune-related and 51 DNA repair-related genes, we classified samples with respect to three immune classes (innate-enriched, adaptive-enriched, and immune-quiet) and two DNA repair classes (recombination/Fanconi anemia (HR/FA), and non-HR/FA) according to published methods [31]). As above, we cross-tabulated AR status with DNA repair status or immune class, and we estimated RFDs and 95% confidence intervals between AR-low and AR-high (referent) samples by fitting a generalized linear model with a binomial distribution and identity link. Positive RFDs indicate enrichment of a given characteristic among AR-low samples. Models were adjusted for TNBC status to determine whether AR status was associated with molecular features independent of TNBC status.
Validation in TCGA
We used TCGA to validate associations between AR status and molecular tumor characteristics. After applying our ClaNC classifier of AR to ER-negative samples from TCGA, we used the composite Homologous Recombination Deficiency (HRD) Scoring method from Kninjenburg et al. [32] to assess whether AR-low samples were more likely to carry DNA-level evidence of HRD defects than AR-high samples. To confirm associations identified in CBCS, we also compared distributions of DNA repair and immune classes, as defined by the same subsets of genes used in CBCS, across strata of AR status. We also compared AR RNA expression to normalized AR protein levels (N = 872 with protein and RNA data) to determine how well findings at the RNA level captured post-translation AR protein status.
Results
AR mRNA associations with demographics and clinical features in breast cancer
We detected associations between single-gene AR RNA expression classes (high vs. low) and aggressive clinical features. RNA levels of AR were strongly correlated with AR protein as measured by RPPA (r = 0.68, p < 0.001; Fig. 1) in TCGA. Figure 2 shows that lower AR RNA expression was observed in ER-negative tumors (Fig. 2a), and tumors from Black (Fig. 2b) and younger (<50 years old) women (Fig. 2c). Among ER-negative tumors we used model-based density estimation to identify subgroups based on ER expression, which confirmed the presence of two distinct AR expression phenotypes, which we refer to as AR-low/ER- and AR-high/ER-.
ER status assessed via central pathology review of immunohistochemistry. Dashed line is derived from finite mixture model of AR distribution in ER-negative samples and represents cutpoint defining empirical AR groups.
Clinical and molecular characteristics of ER- breast cancers stratified by AR status
Table 1 shows associations between AR status and selected clinical and molecular characteristics among ER-negative breast cancers. Compared to women with high AR expression (N = 168), women with low AR expression (N = 304) were more likely to be young, Black, HER2 negative, and high grade. Associations were strongest between AR status and high risk of recurrence genomic scores (ROR-P), with AR-low tumors having a 37% higher prevalence of high ROR-P scores than AR-high tumors (95% CI = 28%–45%). These associations, with the exception of Black race, were somewhat attenuated but remained significant after adjusting for triple-negative status and replicated within triple-negative tumors alone, suggesting that low AR is associated with tumor aggressiveness in ER-negative tumors independent of triple negativity. Similar patterns were observed in ER+ tumors, with AR-low/ER+ tumors being more likely to have high grade, stage, or ROR-P scores than AR-high/ ER+ tumors (Supplementary Table 1). Most ER-borderline tumors were also AR low. Results did not substantially differ after multiple imputation.
Given strong associations between low AR and aggressive tumor features, we developed a classifier to identify low AR status using other independent gene expression data when AR expression was missing (Fig. 3). Fivefold cross-validated Classification to Nearest Centroids (ClaNC) showed average sensitivities for identifying AR-low samples among ER-negative tumors in the training set ranging from 82.8% to 88.4%, while specificities ranged from 82.4% to 86.3% (Fig. 3a). The Youden’s index was maximized when using seven genes per AR phenotype (14 genes total), yielding a final sensitivity of 86.8% and specificity of 87.5% (Fig. 3c). Principal components analysis based on the selected genes shows separation of AR low and AR high (Fig. 3d).
Applying the classifier to the 669 ER-negative CBCS tumors with relevant RNA data, we detected 545 (82.8%) tumors with AR-low phenotypes. Again, AR-low tumors were more likely to be younger, Black, HER2-negative, grade III, stages II-IV, and have high ROR-PT scores (Supplementary Table 2) as also observed in the TCGA cohort (Supplementary Table 3). ER/AR-low tumors also showed strong evidence of aggressive molecular phenotypes, with 47.0% having enrichment for adaptive immune tumor subtypes, and 85.6% showing enrichment for homologous recombination-related genes (Table 2). Even after adjusting for triple-negative status, AR-low tumors showed a 33.3% higher frequency of homologous recombination deficient tumors than AR-high tumors, suggesting strong associations between these two tumor phenotypes and that expression of other receptors is not the sole driver of AR-related associations. The magnitude of associations with other (non-adaptive) immune subtypes was less pronounced, although AR-low tumors had 18.5% lower frequency of immune quiet subtype than AR-high tumors.
Applying the classifier to data from ER-negative tumors in TCGA, we identified 187 tumors (58.7% ER +) with AR-low phenotypes. AR-low tumors showed higher frequency of homologous recombination deficiency (HRD), with 81 (39.3%) having HRD scores above the clinical cutoff of 42 as compared to 11 (5.3%) of AR-high tumors. We did not find strong evidence of an association between AR and immune expression phenotypes in TCGA (RFD = − 5.5%, 95% CI = − 15.8%–4.9%), although the TCGA lacks evidence of the immune quiet phenotype due to different selection factors for inclusion in TCGA [26]. However, AR-low tumors again had higher proportions of expression-based HRD phenotypes than AR-high tumors (adjusted RFD: 41.5%, 95% CI = 34.0%–48.6%).
Fivefold cross-validated Youden’s index and sensitivity of Classification to Nearest Centroids classifier according to number of genes used to predict each AR phenotype. Confidence intervals represent mean plus or minus standard error. Blue lines represent predictions in the training set (N = 375–389), and red lines represent predictions in the test set (N = 93–97). C. Final performance of Youden’s Index-maximizing classifier using seven genes per group. Correct classifications are shown in green, while incorrect classifications are shown in red. Sensitivity was 86.8%, specificity was 87.5%, and overall accuracy was 87.1%. D. Principal component analysis based on RNA expression of classifier-selected genes in ER-negative CBCS samples. Red samples are those with low AR expression, and blue samples have high AR expression.
Discussion
Limitations and inconsistencies with protein-based AR assessment approaches have been previously cited as a barrier to AR expression interpretation and its consistency in the literature [3, 9,10,11,12, 14, 16, 17]. Using CBCS expression data, we designed and validated a multigene classifier that distinguishes AR-low versus AR-high ER-negative breast cancers. AR-low status in ER-negative breast cancer was significantly associated with younger age at diagnosis, Black race, HER2 negativity, high-grade, and higher ROR; these associations, with the exception of Black race, remained significant after adjusting for TNBC status. These findings suggest that in ER-negative breast cancers, low AR expression is associated with aggressive disease. Considering other biological phenotypes, AR-low cancers in the CBCS cohort exhibited adaptive immunity enrichment and both CBCS and TCGA datasets displayed significantly greater homologous recombination repair deficiency among AR-low cancers.
Our findings are consistent with what has been previously reported for demographic factors. Park et al. reported that women, under the age of 35 years, were diagnosed with AR-negative/ER-negative breast cancer more frequently than women over the age of 35 (11.7% and 7.0%, respectively) [33]. Several groups have reported that in TNBC, AR-negativity is significantly associated with younger age at diagnosis and that older age at diagnosis is more prevalent in the luminal androgen receptor subtype and among AR-positive TNBC patients [15, 18, 19, 34,35,36]. Prior evidence also showed that low AR expression in ER-negative cancer is associated with Black race and West African genetic ancestry [37,38,39].
Our findings also corroborate prior evidence suggesting that AR-negative TNBC is associated with aggressive disease features such as advanced stage and high histological grade [3, 8, 10, 11]. The multi-parametric gene expression-based signature ROR-PT is derived from the PAM50 intrinsic subtype signature and has been reported to predict distant recurrence in node-negative and node-positive ER-positive breast cancer patients [40,41,42,43,44]. Our ROR-PT analysis aligns with previous studies showing higher incidence of recurrence or relapse in women with AR-low versus AR-high ER-negative breast cancer [8, 12, 45,46,47,48]. Yang et al. reported that AR positivity is associated with longer relapse-free survival among HER2-negative patients [49]. Wang and colleagues showed that women with AR-low TNBC, a subset of AR-low ER-negative breast cancer, exhibited a greater incidence of distant metastases than women with AR-high TNBC [50].
Another distinction observed herein between AR-negative and AR-positive TNBC was with respect to immune profiles. Consistent with our findings, Davis and colleagues previously reported that AR-negative tumors are upregulated in T-cell marker (CD4 and CD8), immune checkpoint (PD1, PD-L1, and CTLA-4), and immune cell-signaling pathway marker (ILR2, CCR5, NFKBII2) RNA expression compared to AR-positive tumors in TNBC [51]. These findings suggest that AR-negative TNBC may display increased numbers of infiltrating lymphocytes, but exhibit greater immunosuppression compared to AR-positive TNBC. Our RNA-based classifier identified enrichment in adaptive immunity in AR-low compared to AR-high ER-negative tumors.
Previous studies have not evaluated associations between AR and specific DNA repair pathways. We observed a higher prevalence of homologous recombination deficiency in AR-low (versus AR-positive) ER-negative-breast cancer. This finding is consistent with previous studies showing increased genomic instability in AR-negative (versus AR-positive) TNBC. It has been shown that AR-negative TNBCs have 1) increased epidermal growth factor receptor, cyclin-dependent kinase 6, Ki-67, and topoisomerase 2a but 2) downregulated PTEN and HER4 [52,53,54,55]. It was also discovered that AR-negative tumors display a higher level of copy number alterations (CNAs), CIN25, centrosome amplification, and miRNAs/mRNAs pairings associated with genomic instability, cell cycle, and DNA damage [56]. Thus, AR negativity in TNBC may be linked to dysregulation in the cell cycle and impaired DNA damage response, specifically homologous recombination.
Due to study-specific differences in normalization methods and RNA sequencing protocols, the cutoffs used in our analysis may not translate directly to other analyses, though RNA-based classifiers and mixture models should yield similar results. Another limitation was the small sample size of ER-negative breast cancer patient participants, particularly and TNBC patients. QNBC is a subgroup of TNBC that lacks AR expression and has been reported to be more biologically aggressive and distinct from AR-positive TNBCs [51]. A larger pool of QNBC participants would improve our precision and enable analysis of associations with survival and recurrence among AR-low and AR-high subgroups of TNBC. We were also unable to compare directly to IHC, but previous studies have shown that AR RNA-based signatures expression do not always correlate with AR protein.
Our study’s findings suggest RNA-based methods may be valuable for identifying AR-low, ER-negative breast cancer patients. This gene expression-based approach may capture AR-dependent tumor biology that has been inconsistently observed based on IHC alone. Other research groups have designed multigene signatures that predict response to AR-therapy better than biomarker-based IHC alone [57, 58]. In a 2015 phase II study, Traina and colleagues showed that a novel binary multigene biomarker was a better predictor of enzalutamide response than AR expression by IHC [58]. In 2019, Nyquist et al. showed that a multigene signature indicated response to AR-targeted therapies better than monogenic biomarkers [57]. Thus, results from our group and others indicate that RNA-based signatures may have value for capturing AR levels in ER-negative patients. Our findings also suggest that AR-low ER-negative patients may be more likely to have biological features that have previously indicated response to DNA damage-based agents and immunotherapy. Future research may investigate AR as a prognostic biomarker for chemotherapy- or immunotherapy-treated breast cancer patients.”
Data availability
The datasets generated during the current study are available from the corresponding author upon reasonable request. The code in this study is available from the corresponding author upon reasonable request.
References
Collins LC et al (2011) Androgen receptor expression in breast cancer in relation to molecular phenotype: results from the Nurses’ Health Study. Mod Pathol 24(7):924–931. https://doi.org/10.1038/modpathol.2011.54
Safarpour D, Tavassoli FA (2015) A targetable androgen receptor-positive breast cancer subtype hidden among the triple-negative cancers. Arch Pathol Lab Med 139(5):612–617. https://doi.org/10.5858/arpa.2014-0122-RA
Park S et al (2010) Expression of androgen receptors in primary breast cancer. Ann Oncol 21(3):488–492. https://doi.org/10.1093/annonc/mdp510
Micello D et al (2010) Androgen receptor is frequently expressed in HER2-positive ER/PR-negative breast cancers. Virchows Arch 457(4):467–476. https://doi.org/10.1007/s00428-010-0964-y
Gucalp A et al (2013) Phase II trial of bicalutamide in patients with androgen receptor-positive, estrogen receptor-negative metastatic Breast Cancer. Clin Cancer Res 19(19):5505–5512. https://doi.org/10.1158/1078-0432.CCR-12-3327
Bonnefoi H et al (2016) A phase II trial of abiraterone acetate plus prednisone in patients with triple-negative androgen receptor positive locally advanced or metastatic breast cancer (UCBG 12–1). Ann Oncol 27(5):812–818. https://doi.org/10.1093/annonc/mdw067
Traina TA et al (2018) Enzalutamide for the treatment of androgen receptor-expressing triple-negative breast cancer. J Clin Oncol 36(9):884–890. https://doi.org/10.1200/JCO.2016.71.3495
Sutton LM et al (2012) Decreased androgen receptor expression is associated with distant metastases in patients with androgen receptor-expressing triple-negative breast carcinoma. Am J Clin Pathol 138(4):511–516. https://doi.org/10.1309/AJCP8AVF8FDPTZLH
Tang D et al (2012) The expression and clinical significance of the androgen receptor and E-cadherin in triple-negative breast cancer. Med Oncol 29(2):526–533. https://doi.org/10.1007/s12032-011-9948-2
Mrklic I et al (2013) Expression of androgen receptors in triple negative breast carcinomas. Acta Histochem 115(4):344–348. https://doi.org/10.1016/j.acthis.2012.09.006
Ogawa Y et al (2008) Androgen receptor expression in breast cancer: relationship with clinicopathological factors and biomarkers. Int J Clin Oncol 13(5):431–435. https://doi.org/10.1007/s10147-008-0770-6
Asano Y et al (2017) Expression and clinical significance of androgen receptor in triple-negative breast cancer. Cancers (Basel). https://doi.org/10.3390/cancers9010004
Cochrane DR et al (2014) Role of the androgen receptor in breast cancer and preclinical analysis of enzalutamide. Breast Cancer Res 16(1):R7. https://doi.org/10.1186/bcr3599
McGhan LJ et al (2014) Androgen receptor-positive triple negative breast cancer: a unique breast cancer subtype. Ann Surg Oncol 21(2):361–367. https://doi.org/10.1245/s10434-013-3260-7
Choi JE et al (2015) Androgen receptor expression predicts decreased survival in early stage triple-negative breast cancer. Ann Surg Oncol 22(1):82–89. https://doi.org/10.1245/s10434-014-3984-z
Hadgu E et al (2020) Distribution and characteristics of androgen receptor (AR) in breast cancer among women in Addis Ababa, Ethiopia: a cross sectional study. PLoS ONE 15(5):e0232519. https://doi.org/10.1371/journal.pone.0232519
Vera-Badillo FE et al (2014) Androgen receptor expression and outcomes in early breast cancer: a systematic review and meta-analysis. J Natl Cancer Inst 106(1):djt319. https://doi.org/10.1093/jnci/djt319
Lehmann BD et al (2011) Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest 121(7):2750–2767. https://doi.org/10.1172/JCI45014
Astvatsaturyan K et al (2018) Androgen receptor positive triple negative breast cancer: clinicopathologic, prognostic, and predictive features. PLoS ONE 13(6):e0197827. https://doi.org/10.1371/journal.pone.0197827
Barton VN et al (2015) Multiple molecular subtypes of triple-negative breast cancer critically rely on androgen receptor and respond to enzalutamide in vivo. Mol Cancer Ther 14(3):769–778. https://doi.org/10.1158/1535-7163.MCT-14-0926
Brumec M et al (2021) Clinical implications of androgen-positive triple-negative breast cancer. Cancers (Basel). https://doi.org/10.3390/cancers13071642
Harvey JM et al (1999) Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol 17(5):1474–1481. https://doi.org/10.1200/JCO.1999.17.5.1474
Newman B et al (1995) The Carolina breast cancer study: integrating population-based epidemiology and molecular biology. Breast Cancer Res Treat 35(1):51–60. https://doi.org/10.1007/BF00694745
Benefield HC et al (2020) Borderline estrogen receptor-positive breast cancers in black and white women. J Natl Cancer Inst 112(7):728–736. https://doi.org/10.1093/jnci/djz206
Cancer Genome Atlas Research et al (2013) The cancer genome atlas pan-cancer analysis project. Nat Genet 45(10):1113–1120. https://doi.org/10.1038/ng.2764
Hamilton AM et al (2022) The landscape of immune microenvironments in racially diverse breast cancer patients. Cancer Epidemiol Biomarkers Prev 31(7):1341–1350. https://doi.org/10.1158/1055-9965.EPI-21-1312
Bhattacharya A et al (2021) An approach for normalization and quality control for nanostring RNA expression data. Brief Bioinform. https://doi.org/10.1093/bib/bbaa163
Malkov VA et al (2009) Multiplexed measurements of gene signatures in different analytes using the Nanostring nCounter Assay System. BMC Res Notes 2:80. https://doi.org/10.1186/1756-0500-2-80
Troester MA et al (2018) Racial differences in PAM50 subtypes in the Carolina breast cancer study. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djx135
Scrucca L et al (2016) mclust 5: clustering, classification and density estimation using Gaussian finite mixture models. R J 8(1):289–317
Walens A et al (2022) RNA-based classification of homologous recombination deficiency in racially-diverse patients with breast cancer. Cancer Epidemiol Biomarkers Prev. https://doi.org/10.1158/1055-9965.EPI-22-0590
Knijnenburg TA et al (2018) Genomic and molecular landscape of DNA damage repair deficiency across the cancer genome atlas. Cell Rep 23(1):239-254 e6. https://doi.org/10.1016/j.celrep.2018.03.076
Park S et al (2011) Androgen receptor expression is significantly associated with better outcomes in estrogen receptor-positive breast cancers. Ann Oncol 22(8):1755–1762. https://doi.org/10.1093/annonc/mdq678
Angajala A et al (2019) Quadruple negative breast cancers (QNBC) demonstrate subtype consistency among primary and recurrent or metastatic breast cancer. Transl Oncol 12(3):493–501. https://doi.org/10.1016/j.tranon.2018.11.008
Kim EY et al (2017) Clinicopathologic significance of androgen receptor expression and discordant receptor status during progression in breast cancer. Int J Clin Exp Pathol 10(7):7929–7939
Dieci MV et al (2019) Androgen receptor expression and association with distant disease-free survival in triple negative breast cancer: analysis of 263 patients treated with standard therapy for stage i-iii disease. Front Oncol 9:452. https://doi.org/10.3389/fonc.2019.00452
Bhattarai S et al (2019) Prognostic role of androgen receptor in triple negative breast cancer: a multi-institutional study. Cancers (Basel). https://doi.org/10.3390/cancers11070995
Jiagge E et al (2018) Androgen receptor and ALDH1 expression among internationally diverse patient populations. J Glob Oncol 4:1–8. https://doi.org/10.1200/JGO.18.00056
Zakharia F et al (2009) Characterizing the admixed African ancestry of African Americans. Genome Biol 10(12):R141. https://doi.org/10.1186/gb-2009-10-12-r141
Ohnstad HO et al (2017) Prognostic value of PAM50 and risk of recurrence score in patients with early-stage breast cancer with long-term follow-up. Breast Cancer Res 19(1):120. https://doi.org/10.1186/s13058-017-0911-9
Paik S et al (2004) A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med 351(27):2817–2826. https://doi.org/10.1056/NEJMoa041588
Filipits M et al (2011) A new molecular predictor of distant recurrence in ER-positive, HER2-negative breast cancer adds independent information to conventional clinical risk factors. Clin Cancer Res 17(18):6012–6020. https://doi.org/10.1158/1078-0432.CCR-11-0926
Zhang Y et al (2013) Breast cancer index identifies early-stage estrogen receptor-positive breast cancer patients at risk for early- and late-distant recurrence. Clin Cancer Res 19(15):4196–4205. https://doi.org/10.1158/1078-0432.CCR-13-0804
Gnant M et al (2014) Predicting distant recurrence in receptor-positive breast cancer patients with limited clinicopathological risk: using the PAM50 Risk of Recurrence score in 1478 postmenopausal patients of the ABCSG-8 trial treated with adjuvant endocrine therapy alone. Ann Oncol 25(2):339–345. https://doi.org/10.1093/annonc/mdt494
Rakha EA et al (2007) Prognostic markers in triple-negative breast cancer. Cancer 109(1):25–32. https://doi.org/10.1002/cncr.22381
Qu Q et al (2013) The impact of androgen receptor expression on breast cancer survival: a retrospective study and meta-analysis. PLoS ONE 8(12):e82650. https://doi.org/10.1371/journal.pone.0082650
Wang C et al (2016) Prognostic value of androgen receptor in triple negative breast cancer: a meta-analysis. Oncotarget 7(29):46482–46491. https://doi.org/10.18632/oncotarget.10208
Luo X et al (2010) Expression and clinical significance of androgen receptor in triple negative breast cancer. Chin J Cancer 29(6):585–590. https://doi.org/10.5732/cjc.009.10673
Yang Y et al (2020) Prognostic role of androgen receptor expression in surgically resected early breast cancer patients. J Breast Cancer 23(2):182–193. https://doi.org/10.4048/jbc.2020.23.e28
Wang X et al (2020) The prognostic value of androgen receptor (AR) in HER2-enriched metastatic breast cancer. Endocr Relat Cancer 27(4):199–208. https://doi.org/10.1530/ERC-19-0315
Davis M et al (2018) AR negative triple negative or “quadruple negative” breast cancers in African American women have an enriched basal and immune signature. PLoS ONE 13(6):e0196909. https://doi.org/10.1371/journal.pone.0196909
Gasparini P et al (2014) Androgen receptor status is a prognostic marker in non-basal triple negative breast cancers and determines novel therapeutic options. PLoS ONE 9(2):e88525. https://doi.org/10.1371/journal.pone.0088525
Hon JD et al (2016) Breast cancer molecular subtypes: from TNBC to QNBC. Am J Cancer Res 6(9):1864–1872
Tsutsumi Y (2012) Apocrine carcinoma as triple-negative breast cancer: novel definition of apocrine-type carcinoma as estrogen/progesterone receptor-negative and androgen receptor-positive invasive ductal carcinoma. Jpn J Clin Oncol 42(5):375–386. https://doi.org/10.1093/jjco/hys034
Safarpour D, Pakneshan S, Tavassoli FA (2014) Androgen receptor (AR) expression in 400 breast carcinomas: is routine AR assessment justified? Am J Cancer Res 4(4):353–368
Bhattarai S et al (2021) QNBC is associated with high genomic instability characterized by copy number alterations and miRNA deregulation. Int J Mol Sci. https://doi.org/10.3390/ijms222111548
Nyquist MD et al (2019) Molecular determinants of response to high-dose androgen therapy in prostate cancer. JCI Insight. https://doi.org/10.1172/jci.insight.129715
Traina TA et al (2015) Results from a phase 2 study of enzalutamide (ENZA), an androgen receptor (AR) inhibitor, in advanced AR+ triple-negative breast cancer (TNBC). J Clin Oncol 33(15):1003–1003. https://doi.org/10.1200/jco.2015.33.15_suppl.1003
Acknowledgements
This research was supported by a grant from UNC Lineberger Comprehensive Cancer Center, which is funded by the University Cancer Research Fund of North Carolina, the Susan G Komen Foundation (OGUNC1202), the National Cancer Institute of the National Institutes of Health (P01CA151135), and the National Cancer Institute Specialized Program of Research Excellence (SPORE) in Breast Cancer (NIH/NCI P50-CA058223). This research recruited participants and/or obtained data with the assistance of Rapid Case Ascertainment, a collaboration between the North Carolina Central Cancer Registry and the Lineberger Comprehensive Cancer Center at the University of North Carolina at Chapel Hill. RCA is supported by a grant from the National Cancer Institute of the National Institutes of Health (P30CA016086) and by a grant from UNC Lineberger Comprehensive Cancer Center, which is funded by the University Cancer Research Fund of North Carolina. In addition, S. Van Alsten is supported by the UNC Cancer Control Education Program (T32CA057726). The authors would like to acknowledge the University of North Carolina BioSpecimen Processing Facility for sample processing, storage, and sample disbursements (http://bsp.web.unc.edu/). We are grateful to CBCS participants and study staff.
Funding
Open access funding provided by SCELC, Statewide California Electronic Library Consortium. This research was supported by a grant from UNC Lineberger Comprehensive Cancer Center, which is funded by the University Cancer Research Fund of North Carolina, the Susan G Komen Foundation (OGUNC1202), the National Cancer Institute of the National Institutes of Health (P01CA151135), and the National Cancer Institute Specialized Program of Research Excellence (SPORE) in Breast Cancer (NIH/NCI P50-CA058223). This research recruited participants and/or obtained data with the assistance of Rapid Case Ascertainment, a collaboration between the North Carolina Central Cancer Registry and UNC Lineberger. RCA is supported by a grant from the National Cancer Institute of the National Institutes of Health (P30CA016086).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by NJ, MT, and SVA. The first draft of the manuscript was written by NJ and SVA, and all authors commented on previous versions of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The University of North Carolina, Chapel Hill has a license of intellectual property interest in GeneCentric Diagnostics and BioClassifier, LLC, which may be used in this study. The University of North Carolina, Chapel Hill may benefit from this interest that is/are related to this research. The terms of this arrangement have been reviewed and approved by the University of North Carolina, Chapel Hill Conflict of Interest Program in accordance with its conflict of interest policies.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of University of North Carolina, Chapel Hill.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publication
The authors affirm that human research participants provided informed consent for publication of the images in Figs. 1, 2, and 3.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jinna, N.D., Van Alsten, S., Rida, P. et al. Molecular features of androgen-receptor low, estrogen receptor-negative breast cancers in the Carolina breast cancer study. Breast Cancer Res Treat 201, 171–181 (2023). https://doi.org/10.1007/s10549-023-07014-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-023-07014-x