Abstract
Purpose
There are limited data on trastuzumab–pertuzumab (HP)-based treatments beyond the first-line, HER2+ metastatic breast cancer (MBC) setting. We conducted a phase II study of eribulin mesylate, which extends survival in MBC, with HP in patients with previously treated HER2+ MBC to evaluate efficacy, toxicity, and genomic alterations driving therapeutic response.
Methods
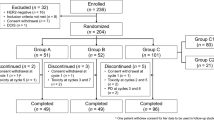
After a run-in phase for eribulin dosing, two cohorts were enrolled (Cohort A-no prior pertuzumab; Cohort B-prior pertuzumab). All patients received eribulin 1.4 mg/m2 on days 1, 8 with standard-dose HP on day 1 (21-day cycles). The primary endpoint was objective response rate (ORR). Genomic characterization via whole exome sequencing (WES) was completed on tumor DNA and matched germline DNA from 19 patients.
Results
The six-patient run-in established a dose of eribulin 1.4 mg/m2 with HP. Cohorts A and B enrolled 17 and 7 patients, respectively. Accrual stopped early due to an evolving treatment landscape and slow enrollment. The ORR was 26.3% (95% Confidence Interval [CI] 9.2–51.2%) in Cohort A and 0% in Cohort B (95% CI 0–41.0%). WES revealed more frequent alterations in TP53 (p < 0.05, q > 0.05) in patients without clinical benefit (disease control for < 24 weeks) which was not significant after multiple hypothesis correction.
Conclusion
Eribulin–HP had manageable toxicity and modest clinical activity in patients without prior pertuzumab exposure. This study provides a preliminary landscape of somatic alterations in this patient cohort. Our data add to the literature on how genomic alterations may predict for therapy response/resistance, as we work to individualize choices in a quickly evolving HER2+ MBC treatment landscape.
Trial registration
www.clinicaltrials.gov, NCT01912963. Registered 24 July 2013.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Swain SM, Baselga J, Kim S-B, Ro J, Semiglazov V, Campone M, Ciruelos E, Ferrero J-M, Schneeweiss A, Heeson S, Clark E, Ross G, Benyunes MC, Cortés J (2015) Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N Engl J Med 372(8):724–734
Freedman RA, Gelman RS, Wefel JS, Melisko ME, Hess KR, Connolly RM, Poznak CHV, Niravath PA, Puhalla SL, Ibrahim N, Blackwell KL, Moy B, Herold C, Liu MC, Lowe A, Agar NYR, Ryabin N, Farooq S, Lawler E, Rimawi MF, Krop IE, Wolff AC, Winer EP, Lin NU (2016) Translational Breast Cancer Research Consortium (TBCRC) 022: a phase II trial of neratinib for patients with human epidermal growth factor receptor 2-positive breast cancer and brain metastases. J Clin Oncol 34(9):945–952
Verma S, Miles D, Gianni L, Krop IE, Welslau M, Baselga J, Pegram M, Oh D-Y, Diéras V, Guardino E, Fang L, Lu MW, Olsen S, Blackwell K (2012) Trastuzumab emtansine for HER2-positive advanced breast cancer. N Engl J Med 367(19):1783–1791
Geyer CE, Forster J, Lindquist D, Chan S, Romieu CG, Pienkowski T, Jagiello-Gruszfeld A, Crown J, Chan A, Kaufman B, Skarlos D, Campone M, Davidson N, Berger M, Oliva C, Rubin SD, Stein S, Cameron D (2006) Lapatinib plus capecitabine for HER2-positive advanced breast cancer. N Engl J Med 355(26):2733–2743
Freedman RA, Gelman RS, Anders CK, Melisko ME, Parsons HA, Cropp AM, Silvestri K, Cotter CM, Componeschi KP, Marte JM, Connolly RM, Moy B, Poznak CHV, Blackwell KL, Puhalla SL, Jankowitz RC, Smith KL, Ibrahim N, Moynihan TJ, O’Sullivan CC, Nangia J, Niravath P, Tung N, Pohlmann PR, Burns R, Rimawi MF, Krop IE, Wolff AC, Winer EP, Lin NU (2019) TBCRC 022: a phase II trial of neratinib and capecitabine for patients with human epidermal growth factor receptor 2-positive breast cancer and brain metastases. J Clin Oncol 37(13):1081–1089
Murthy RK, Loi S, Okines A, Paplomata E, Hamilton E, Hurvitz SA, Lin NU, Borges V, Abramson V, Anders C, Bedard PL, Oliveira M, Jakobsen E, Bachelot T, Shachar SS, Müller V, Braga S, Duhoux FP, Greil R, Cameron D, Carey LA, Curigliano G, Gelmon K, Hortobagyi G, Krop I, Loibl S, Pegram M, Slamon D, Palanca-Wessels MC, Walker L, Feng W, Winer EP (2020) Tucatinib, trastuzumab, and capecitabine for HER2-positive metastatic breast cancer. N Engl J Med 382(7):597–609
Modi S, Saura C, Yamashita T, Park YH, Kim S-B, Tamura K, Andre F, Iwata H, Ito Y, Tsurutani J, Sohn J, Denduluri N, Perrin C, Aogi K, Tokunaga E, Im S-A, Lee KS, Hurvitz SA, Cortes J, Lee C, Chen S, Zhang L, Shahidi J, Yver A, Krop I (2020) Trastuzumab deruxtecan in previously treated HER2-positive breast cancer. N Engl J Med 382(7):610–621
Cortés J, Fumoleau P, Bianchi GV, Petrella TM, Gelmon K, Pivot X, Verma S, Albanell J, Conte P, Lluch A, Salvagni S, Servent V, Gianni L, Scaltriti M, Ross GA, Dixon J, Szado T, Baselga J (2012) Pertuzumab monotherapy after trastuzumab-based treatment and subsequent reintroduction of trastuzumab: activity and tolerability in patients with advanced human epidermal growth factor receptor 2-positive breast cancer. J Clin Oncol 30(14):1594–1600
Arteaga CL, Sliwkowski MX, Osborne CK, Perez EA, Puglisi F, Gianni L (2011) Treatment of HER2-positive breast cancer: current status and future perspectives. Nat Rev Clin Oncol 9(1):16–32
Harbeck N, Beckmann MW, Rody A, Schneeweiss A, Müller V, Fehm T, Marschner N, Gluz O, Schrader I, Heinrich G, Untch M, Jackisch C (2013) HER2 dimerization inhibitor pertuzumab—mode of action and clinical data in breast cancer. Breast Care (Basel) 8(1):49–55
Baselga J, Cortés J, Kim S-B, Im S-A, Hegg R, Im Y-H, Roman L, Pedrini JL, Pienkowski T, Knott A, Clark E, Benyunes MC, Ross G, Swain SM (2012) Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N Engl J Med 366(2):109–119
Dybdal-Hargreaves NF, Risinger AL, Mooberry SL (2015) Eribulin mesylate: mechanism of action of a unique microtubule-targeting agent. Clin Cancer Res 21(11):2445–2452
Vahdat LT, Pruitt B, Fabian CJ, Rivera RR, Smith DA, Tan-Chiu E, Wright J, Tan AR, DaCosta NA, Chuang E, Smith J, O’Shaughnessy J, Shuster DE, Meneses NL, Chandrawansa K, Fang F, Cole PE, Ashworth S, Blum JL (2009) Phase II study of eribulin mesylate, a halichondrin b analog, in patients with metastatic breast cancer previously treated with an anthracycline and a taxane. J Clin Oncol 27(18):2954–2961
Cortes J, Vahdat L, Blum JL, Twelves C, Campone M, Roché H, Bachelot T, Awada A, Paridaens R, Goncalves A, Shuster DE, Wanders J, Fang F, Gurnani R, Richmond E, Cole PE, Ashworth S, Allison MA (2010) Phase II study of the halichondrin B analog eribulin mesylate in patients with locally advanced or metastatic breast cancer previously treated with an anthracycline, a taxane, and capecitabine. J Clin Oncol 28(25):3922–3928
Cortes J, O’Shaughnessy J, Loesch D, Blum JL, Vahdat LT, Petrakova K, Chollet P, Manikas A, Diéras V, Delozier T, Vladimirov V, Cardoso F, Koh H, Bougnoux P, Dutcus CE, Seegobin S, Mir D, Meneses N, Wanders J, Twelves C (2011) Eribulin monotherapy versus treatment of physician’s choice in patients with metastatic breast cancer (EMBRACE): a phase 3 open-label randomised study. Lancet 377(9769):914–923
Twelves C, Cortes J, Vahdat LT, Wanders J, Akerele C, Kaufman PA (2010) Phase III trials of eribulin mesylate (E7389) in extensively pretreated patients with locally recurrent or metastatic breast cancer. Clin Breast Cancer 10(2):160–163
Gale M, Li Y, Cao J, Liu ZZ, Holmbeck MA, Zhang M, Lang SM, Wu L, Do Carmo M, Gupta S, Aoshima KA-O, DiGiovanna MP, Stern DA-O, Rimm DL, Shadel GS, Chen X, Yan QA-OX (2020) Acquired resistance to HER2-targeted therapies creates vulnerability to ATP synthase inhibition. Cancer Res 80(3):524–535
Pohlmann PR, Mayer IA, Mernaugh R (2009) Resistance to trastuzumab in breast cancer. Clin Cancer Res 15(24):7479–7491
O’Shaughnessy J, Huggins-Puhalla SL, Wilks S, Brufsky A, Schwartzberg LS, Berrak E, Song JX, Cox D, Vahdat LT (2014) Clinical effects of prior trastuzumab on combination eribulin mesylate plus trastuzumab as first-line treatment for HER2+ locally recurrent or metastatic breast cancer (MBC): results from a phase 2, single-arm, multicenter study. J Clin Oncol 32(26 Suppl.):139–139
Wilks S, Puhalla S, O’Shaughnessy J, Schwartzberg L, Berrak E, Song J, Cox D, Vahdat L (2014) Phase 2, Multicenter, single-arm study of eribulin mesylate with trastuzumab as first-line therapy for locally recurrent or metastatic HER2-positive breast cancer. Clin Breast Cancer 14(6):405–412
Wolff AC, Hammond MEH, Hicks DG, Dowsett M, McShane LM, Allison KH, Allred DC, Bartlett JMS, Bilous M, Fitzgibbons P, Hanna W, Jenkins RB, Mangu PB, Paik S, Perez EA, Press MF, Spears PA, Vance GH, Viale G, Hayes DF (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Update. J Clin Oncol 31(31):3997–4013
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J (2009) New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247
Adalsteinsson VA, Ha G, Freeman SS, Choudhury AD, Stover DG, Parsons HA, Gydush G, Reed SC, Rotem D, Rhoades J, Loginov D, Livitz D, Rosebrock D, Leshchiner I, Kim J, Stewart C, Rosenberg M, Francis JM, Zhang C-Z, Cohen O, Oh C, Ding H, Polak P, Lloyd M, Mahmud S, Helvie K, Merrill MS, Santiago RA, O’Connor EP, Jeong SH, Leeson R, Barry RM, Kramkowski JF, Zhang Z, Polacek L, Lohr JG, Schleicher M, Lipscomb E, Saltzman A, Oliver NM, Marini L, Waks AG, Harshman LC, Tolaney SM, Allen EMV, Winer EP, Lin NU, Nakabayashi M, Taplin M-E, Johannessen CM, Garraway LA, Golub TR, Boehm JS, Wagle N, Getz G, Love JC, Meyerson M (2017) Scalable whole-exome sequencing of cell-free DNA reveals high concordance with metastatic tumors. Nat Commun 8(1):1324
Sondka Z, Bamford S, Cole CG, Ward SA, Dunham I, Forbes SA (2018) The COSMIC Cancer Gene Census: describing genetic dysfunction across all human cancers. Nat Rev Cancer 18(11):696–705
Mao P, Cohen O, Kowalski KJ, Kusiel JG, Buendia-Buendia JE, Cuoco MS, Exman P, Wander SA, Waks AG, Nayar U, Chung J, Freeman S, Rozenblatt-Rosen O, Miller VA, Piccioni F, Root DE, Regev A, Winer EP, Lin NU, Wagle N (2020) Acquired FGFR and FGF alterations confer resistance to estrogen receptor (ER) targeted therapy in ER+ metastatic breast cancer. Clin Cancer Res 26(22):5974–5989
Wander SA, Cohen O, Gong X, Johnson GN, Buendia-Buendia JE, Lloyd MR, Kim D, Luo F, Mao P, Helvie K, Kowalski KJ, Nayar U, Waks AG, Parsons SH, Martinez R, Litchfield LM, Ye XS, Yu C, Jansen VM, Stille JR, Smith PS, Oakley GJ, Chu QS, Batist G, Hughes ME, Kremer JD, Garraway LA, Winer EP, Tolaney SM, Lin NU, Buchanan SG, Wagle N (2020) The genomic landscape of intrinsic and acquired resistance to cyclin-dependent kinase 4/6 inhibitors in patients with hormone receptor-positive metastatic breast cancer. Cancer Discov 10(8):1174–1193
Belinky F, Nativ N, Stelzer G, Zimmerman S, Stein TI, Safran M, Lancet D (2015) PathCards: multi-source consolidation of human biological pathways. Database 2015:bav006.
Waks AG, Cohen O, Kochupurakkal B, Kim D, Wander SA, Buendia-Buendia J, Helvie K, Matulonis UA, Krop IE, Tolaney SM, Winer EP, D’Andrea AD, Shapiro G, Lin NU, Wagle N (2019) Reversion and non-reversion mechanisms of resistance (MoR) to PARP inhibitor (PARPi) or platinum chemotherapy (chemotx) in patients (pts) with BRCA1/2-mutant metastatic breast cancer (MBC). J Clin Oncol 37(15 Suppl):1085–1085
Alexandrov LB, Nik-Zainal S, Wedge DC, Aparicio SAJR, Behjati S, Biankin AV, Bignell GR, Bolli N, Borg A, Børresen-Dale A-L, Boyault S, Burkhardt B, Butler AP, Caldas C, Davies HR, Desmedt C, Eils R, Eyfjörd JE, Foekens JA, Greaves M, Hosoda F, Hutter B, Ilicic T, Imbeaud S, Imielinski M, Jäger N, Jones DTW, Jones D, Knappskog S, Kool M, Lakhani SR, López-Otín C, Martin S, Munshi NC, Nakamura H, Northcott PA, Pajic M, Papaemmanuil E, Paradiso A, Pearson JV, Puente XS, Raine K, Ramakrishna M, Richardson AL, Richter J, Rosenstiel P, Schlesner M, Schumacher TN, Span PN, Teague JW, Totoki Y, Tutt ANJ, Valdés-Mas R, van Buuren MM, van’t Veer L, Vincent-Salomon A, Waddell N, Yates LR, Zucman-Rossi J, Futreal PA, McDermott U, Lichter P, Meyerson M, Grimmond SM, Siebert R, Campo EA, Shibata T, Pfister SM, Campbell PJ, Stratton MR (2013) Signatures of mutational processes in human cancer. Nature 500(7463):415–421
Kim J, Mouw KW, Polak P, Braunstein LZ, Kamburov A, Tiao G, Kwiatkowski DJ, Rosenberg JE, Allen EMV, D’Andrea AD, Getz G (2016) Somatic ERCC2 mutations are associated with a distinct genomic signature in urothelial tumors. Nat Genet 48(6):600–606
Kasar S, Kim J, Improgo R, Tiao G, Polak P, Haradhvala N, Lawrence MS, Kiezun A, Fernandes SM, Bahl S, Sougnez C, Gabriel S, Lander ES, Kim HT, Getz G, Brown JR (2015) Whole-genome sequencing reveals activation-induced cytidine deaminase signatures during indolent chronic lymphocytic leukaemia evolution. Nat Commun 6(1):8866
Tan VYF, Fevotte C (2013) Automatic relevance determination in nonnegative matrix factorization with the /spl beta/-Divergence. IEEE Trans Pattern Anal Mach Intell 35(7):1592–1605
Painter CA, Jain E, Tomson BN, Dunphy M, Stoddard RE, Thomas BS, Damon AL, Shah S, Kim D, Zañudo JGT, Hornick JL, Chen Y-L, Merriam P, Raut CP, Demetri GD, Tine BAV, Lander ES, Golub TR, Wagle N (2020) The Angiosarcoma Project: enabling genomic and clinical discoveries in a rare cancer through patient-partnered research. Nat Med 26(2):181–187
Reardon B (2018) Calculate Mutation Burden. https://github.com/brendanreardon/calculate_mutational_burden (accessed 19 Mar 2021)
Sanchez-Vega F, Mina M, Armenia J, Chatila WK, Luna A, La KC, Dimitriadoy S, Liu DL, Kantheti HS, Saghafinia S, Chakravarty D, Daian F, Gao Q, Bailey MH, Liang W-W, Foltz SM, Shmulevich I, Ding L, Heins Z, Ochoa A, Gross B, Gao J, Zhang H, Kundra R, Kandoth C, Bahceci I, Dervishi L, Dogrusoz U, Zhou W, Shen H, Laird PW, Way GP, Greene CS, Liang H, Xiao Y, Wang C, Iavarone A, Berger AH, Bivona TG, Lazar AJ, Hammer GD, Giordano T, Kwong LN, McArthur G, Huang C, Tward AD, Frederick MJ, McCormick F, Meyerson M, Allen EMV, Cherniack AD, Ciriello G, Sander C, Schultz N (2018) Oncogenic signaling pathways in the cancer genome atlas. Cell 173(2):321-337.e310
Chang MT, Bhattarai TS, Schram AM, Bielski CM, Donoghue MTA, Jonsson P, Chakravarty D, Phillips S, Kandoth C, Penson A, Gorelick A, Shamu T, Patel S, Harris C, Gao J, Sumer SO, Kundra R, Razavi P, Li BT, Reales DN, Socci ND, Jayakumaran G, Zehir A, Benayed R, Arcila ME, Chandarlapaty S, Ladanyi M, Schultz N, Baselga J, Berger MF, Rosen N, Solit DB, Hyman DM, Taylor BS (2017) Accelerating discovery of functional mutant alleles in cancer. Cancer Discov 8(2):174–183
Chakravarty D, Gao J, Phillips SM, Kundra R, Zhang H, Wang J, Rudolph JE, Yaeger R, Soumerai T, Nissan MH, Chang MT, Chandarlapaty S, Traina TA, Paik PK, Ho AL, Hantash FM, Grupe A, Baxi SS, Callahan MK, Snyder A, Chi P, Danila D, Gounder M, Harding JJ, Hellmann MD, Iyer G, Janjigian Y, Kaley T, Levine DA, Lowery M, Omuro A, Postow MA, Rathkopf D, Shoushtari AN, Shukla N, Voss M, Paraiso E, Zehir A, Berger MF, Taylor BS, Saltz LB, Riely GJ, Ladanyi M, Hyman DM, Baselga J, Sabbatini P, Solit DB, Schultz N (2017) OncoKB: a precision oncology knowledge base. JCO Precis Oncol 2017:PO.17.00011.
Crowdis J, He MX, Reardon B, Allen EMV (2020) CoMut: visualizing integrated molecular information with comutation plots. Bioinformatics 36(15):4348–4349
Lawrence MS, Stojanov P, Polak P, Kryukov GV, Cibulskis K, Sivachenko A, Carter SL, Stewart C, Mermel CH, Roberts SA, Kiezun A, Hammerman PS, McKenna A, Drier Y, Zou L, Ramos AH, Pugh TJ, Stransky N, Helman E, Kim J, Sougnez C, Ambrogio L, Nickerson E, Shefler E, Cortés ML, Auclair D, Saksena G, Voet D, Noble M, DiCara D, Lin P, Lichtenstein L, Heiman DI, Fennell T, Imielinski M, Hernandez B, Hodis E, Baca S, Dulak AM, Lohr J, Landau D-A, Wu CJ, Melendez-Zajgla J, Hidalgo-Miranda A, Koren A, McCarroll SA, Mora J, Lee RS, Crompton B, Onofrio R, Parkin M, Winckler W, Ardlie K, Gabriel SB, Roberts CWM, Biegel JA, Stegmaier K, Bass AJ, Garraway LA, Meyerson M, Golub TR, Gordenin DA, Sunyaev S, Lander ES, Getz G (2013) Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature 499:214–218
Araki K, Fukada I, Yanagi H, Kobayashi K, Shibayama T, Horii R, Takahashi S, Akiyama F, Ohno S, Ito Y (2017) First report of eribulin in combination with pertuzumab and trastuzumab for advanced HER2-positive breast cancer. Breast 35:78–84
Tono Y, Ishihara M, Miyahara Y, Tamaru S, Oda H, Yamashita Y, Tawara I, Ikeda H, Shiku H, Mizuno T, Katayama N (2018) Pertuzumab, trastuzumab and eribulin mesylate therapy for previously treated advanced HER2-positive breast cancer: a feasibility study with analysis of biomarkers. Oncotarget 9(19):14909–14921
Krop IE, Kim S-B, González-Martı́n A, LoRusso PM, Ferrero J-M, Smitt M, Yu R, Leung ACF, Wildiers H (2014) Trastuzumab emtansine versus treatment of physician’s choice for pretreated HER2-positive advanced breast cancer (TH3RESA): a randomised, open-label, phase 3 trial. Lancet Oncol 15(7):689–699
Krop IE, Kim S-B, Martin AG, LoRusso PM, Ferrero J-M, Badovinac-Crnjevic T, Hoersch S, Smitt M, Wildiers H (2017) Trastuzumab emtansine versus treatment of physician’s choice in patients with previously treated HER2-positive metastatic breast cancer (TH3RESA): final overall survival results from a randomised open-label phase 3 trial. Lancet Oncol 18(6):743–754
von Minckwitz G, Schwedler K, Schmidt M, Barinoff J, Mundhenke C, Cufer T, Maartense E, de Jongh FE, Baumann KH, Bischoff J, Harbeck N, Lück H-J, Maass N, Zielinski C, Andersson M, Stein RC, Nekljudova V, Loibl S (2011) Trastuzumab beyond progression: overall survival analysis of the GBG 26/BIG 3–05 phase III study in HER2-positive breast cancer. Eur J Cancer 47(15):2273–2281
Cameron D, Casey M, Press M, Lindquist D, Pienkowski T, Romieu CG, Chan S, Jagiello-Gruszfeld A, Kaufman B, Crown J, Chan A, Campone M, Viens P, Davidson N, Gorbounova V, Raats JI, Skarlos D, Newstat B, Roychowdhury D, Paoletti P, Oliva C, Rubin S, Stein S, Geyer CE (2008) A phase III randomized comparison of lapatinib plus capecitabine versus capecitabine alone in women with advanced breast cancer that has progressed on trastuzumab: updated efficacy and biomarker analyses. Breast Cancer Res Treat 112(3):533–543
Cameron D, Casey M, Oliva C, Newstat B, Imwalle B, Geyer CE (2010) Lapatinib plus capecitabine in women with HER-2-positive advanced breast cancer: final survival analysis of a phase III randomized trial. Oncologist 15(9):924–934
Blackwell KL, Burstein HJ, Storniolo AM, Rugo HS, Sledge G, Aktan G, Ellis C, Florance A, Vukelja S, Bischoff J, Baselga J, O’Shaughnessy J (2012) Overall survival benefit with lapatinib in combination with trastuzumab for patients with human epidermal growth factor receptor 2-positive metastatic breast cancer: final results from the EGF104900 study. J Clin Oncol 30(21):2585–2592
Inoue K, Ninomiya J, Okubo K, Nakakuma T, Yamada H, Kimizuka K, Higuchi T, Saito T (2019) Abstract P6–17-14: eribulin, trastuzumab, and pertuzumab as first-line therapy for patients with HER2-positive metastatic breast cancer: a multicenter, collaborative, open-label, phase II clinical trial for the SBCCSG-36 investigators. Cancer Res. https://doi.org/10.1158/1538-7445.SABCS18-P6-17-14
Urruticoechea A, Rizwanullah M, Im S-A, Sánchez-Ruiz AC, Lang I, Tomasello G, Douthwaite H, Crnjevic TB, Heeson S, Eng-Wong J, Munoz M (2016) PHEREXA: A phase III study of trastuzumab (H) + capecitabine (X) ± pertuzumab (P) for patients (pts) who progressed during/after one line of H-based therapy in the HER2-positive metastatic breast cancer (MBC) setting. J Clin Oncol 34(15 Suppl):504
Argolo DFSDTE, Smyth LM, Iyengar NM, Patil S, Norton L, Baselga J, Hudis CA, Dang CT (2015) Trastuzumab and pertuzumab-based versus other therapy as second-line therapy in HER2-positive metastatic breast cancer: a retrospective study from single center. J Clin Oncol 33(15 Suppl):616
Bergin ART, Luen SJ, Savas P, Boolell V, Cho D, Lynch J, Nott L, Stuart-Harris R, Teo LN, Yap SY, Loi S (2019) Efficacy of late line pertuzumab with trastuzumab and chemotherapy in HER2-positive metastatic breast cancer: an Australian case series. Asia Pac J Clin Oncol 15(6):377–382
Iyengar NM, Smyth LM, Lake D, Gucalp A, Singh JC, Traina TA, DeFusco P, Fornier MN, Goldfarb S, Jhaveri K, Modi S, Troso-Sandoval T, Patil S, Ulaner GA, Jochelson M, Norton L, Hudis CA, Dang CT (2019) Efficacy and safety of gemcitabine with trastuzumab and pertuzumab after prior pertuzumab-based therapy among patients with human epidermal growth factor receptor 2–positive metastatic breast cancer. JAMA Netw Open 2(11):e1916211
Fedorova O, Daks A, Shuvalov O, Kizenko A, Petukhov A, Gnennaya Y, Barlev N (2020) Attenuation of p53 mutant as an approach for treatment Her2-positive cancer. Cell Death Discov 6(1):100
Thol F, Kade S, Schlarmann C, Löffeld P, Morgan M, Krauter J, Wlodarski MW, Kölking B, Wichmann M, Görlich K, Göhring G, Bug G, Ottmann O, Niemeyer CM, Hofmann W-K, Schlegelberger B, Ganser A, Heuser M (2012) Frequency and prognostic impact of mutations in SRSF2, U2AF1, and ZRSR2 in patients with myelodysplastic syndromes. Blood 119(15):3578–3584
Esfahani MS, Lee LJ, Jeon YJ, Flynn RA-O, Stehr H, Hui AB, Ishisoko N, Kildebeck E, Newman AA-O, Bratman SA-O, Porteus MA-O, Chang HA-O, Alizadeh AA-O, Diehn MA-O (2019) Functional significance of U2AF1 S34F mutations in lung adenocarcinomas. Nat Commun 10(1):5712
Razavi P, Chang MT, Xu G, Bandlamudi C, Ross DS, Vasan N, Cai Y, Bielski CM, Donoghue MTA, Jonsson P, Penson A, Shen R, Pareja F, Kundra R, Middha S, Cheng ML, Zehir A, Kandoth C, Patel R, Huberman K, Smyth LM, Jhaveri K, Modi S, Traina TA, Dang C, Zhang W, Weigelt B, Li BT, Ladanyi M, Hyman DM, Schultz N, Robson ME, Hudis C, Brogi E, Viale A, Norton L, Dickler MN, Berger MF, Iacobuzio-Donahue CA, Chandarlapaty S, Scaltriti M, Reis-Filho JS, Solit DB, Taylor BS, Baselga J (2018) The genomic landscape of endocrine-resistant advanced breast cancers. Cancer Cell 34(3):427-438.e6
Cancer Genome Atlas Network (2012) Comprehensive molecular portraits of human breast tumours. Nature 490(7418):61–70
Seiler M, Peng S, Agrawal AA, Palacino J, Teng T, Zhu P, Smith PG, Buonamici S, Yu L (2018) Somatic mutational landscape of splicing factor genes and their functional consequences across 33 cancer types. Cell Rep 23(1):282-296.e4
Zhou Q, Derti A, Ruddy D, Rakiec D, Kao I, Lira M, Gibaja V, Chan H, Yang Y, Min J, Schlabach MR, Stegmeier F (2015) A chemical genetics approach for the functional assessment of novel cancer genes. Cancer Res 75(10):1949–16958
Acknowledgements
We thank the patients who participated in this study and who graciously provided blood and tissue samples for genomic analyses. We also thank Kaitlyn Bifolck, BA, for her assistance with manuscript formatting and journal submission.
Funding
Eisai Co. Ltd. In addition, Susan G. Komen (CCR14298143 and CCRCR18552788 to RAF) and American Cancer Society (MRSG-14-240-01-CPPB to RAF).
Author information
Authors and Affiliations
Contributions
Conceptualization—RAF, IVL, NUL, NW; data curation—RAF, IVL, SMB, EJ, KH, J B-B, NW; formal analysis—RAF, IVL, TL, NT, SMB, EJ, JB-B, NW; funding acquisition—RAF and NUL; investigation—all authors; methodology—RAF, IVL, SMB, EJ, NW, NUL; supervision—RAF and NW; writing: original draft—IVL, RAF, SMB; writing: review & editing—all authors.
Corresponding author
Ethics declarations
Conflict of interest
IVL reports institutional honoraria from Pfizer, Astra-Zeneca, and Amgen. IEK reports research support (paid to his institution) from Genentech/Roche and Pfizer, has received fees from Novartis and Merck for Data Monitoring Board participation, received honoraria from Celltrion, and has received consulting fees from Bristol Meyers Squibb, Daiichi/Sankyo, Macrogenics, Context Therapeutics, Taiho Oncology, Genentech/Roche, Seattle Genetics, and AstraZeneca. NU reports research support (to institution) from Genentech, Merck, Pfizer, Seattle Genetics; Consultant/ad board: Puma, Seattle Genetics, Daiichi Sankyo, AstraZeneca, Denali Therapeutics, California Institute for Regenerative Medicine, Prelude Therapeutics. RAF reports research support (to institution, no salary support): Eisai and Puma. SJI has received institutional research funding from Abbive, Astrazeneca, Genetech, Merck and OcnoPep, and fees from Seattle genetics, Novartis, Puma. N.W. reports advisory relationships and consulting with Eli Lilly and Co.; advising and stockholding interest in Relay Therapeutics; and grant support from Puma Biotechnology. JB-B is now employed by Cellarity, Inc. Cambridge, MA, USA.
Ethical approval
The study was conducted according to the current guidelines for good and sound research practice after approval from the Dana-Farber Cancer Institute Office for Human Research Studies.
Informed consent
Informed consent was obtained from all individual participants included in the study. Prior to any study procedures, all patients provided written informed consent for study participation.
Consent for publication
Included in patient consent for study.
Research involving human and animal participants
The study was conducted according to the current guidelines for good and sound research practice after approval from the Dana-Farber Cancer Institute Office for Human Research Studies.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Nikhil Wagle and Rachel A. Freedman share co-senior authorship.
Prior Presentations: American Society of Clinical Oncology (ASCO) Annual Meeting, June 2–6, 2017, Chicago, IL.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Balch, S.M., Vaz-Luis, I., Li, T. et al. A phase II study of efficacy, toxicity, and the potential impact of genomic alterations on response to eribulin mesylate in combination with trastuzumab and pertuzumab in women with human epidermal growth factor receptor 2 (HER2)+ metastatic breast cancer. Breast Cancer Res Treat 189, 411–423 (2021). https://doi.org/10.1007/s10549-021-06329-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-021-06329-x