Abstract
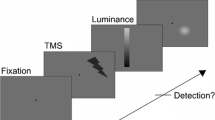
Existing literature on sensory deprivation suggests that short-lasting periods of dark adaptation (DA) can cause changes in visual cortex excitability. DA cortical effects have previously been assessed through phosphene perception, i.e., the ability to report visual sensations when a transcranial magnetic stimulation (TMS) pulse is delivered over the visual cortex. However, phosphenes represent an indirect measure of visual cortical excitability which relies on a subjective report. Here, we aimed at overcoming this limitation by assessing visual cortical excitability by combining subjective (i.e., TMS-induced phosphenes) and objective (i.e., TMS-evoked potentials - TEPs) measurements in a TMS-EEG protocol after 30 min of DA. DA effects were compared to a control condition, entailing 30 min of controlled light exposure. TMS was applied at 11 intensities in order to estimate the psychometric function of phosphene report and explore the relationship between TEPs and TMS intensity. Compared to light adaptation, after DA the slope of the psychometric function was significantly steeper, and the amplitude of a TEP component (P60) was lower, only for high TMS intensities. The perceptual threshold was not affected by DA. These results support the idea that DA leads to a change in the excitability of the visual cortex, accompanied by a behavioral modification of visual perception. Furthermore, this study provides a first valuable description of the relationship between TMS intensity and visual TEPs.






Similar content being viewed by others
References
Abrahamyan A, Clifford CWG, Ruzzoli M, Phillips D, Arabzadeh E, Harris JA (2011) Accurate and rapid estimation of phosphene thresholds (REPT). PLoS ONE 6(7):e22342. https://doi.org/10.1371/journal.pone.0022342
Bagattini C, Mazzi C, Savazzi S (2015) Waves of awareness for occipital and parietal phosphenes perception. Neuropsychologia 70:114–125. https://doi.org/10.1016/j.neuropsychologia.2015.02.021
Boroojerdi B, Bushara KO, Corwell B, Immisch I, Battaglia F, Muellbacher W, Cohen LG (2000) Enhanced excitability of the human visual cortex induced by short-term light deprivation. Cereb Cortex (New York, N.Y.: 1991) 10(5):529–534. https://doi.org/10.1093/cercor/10.5.529
Boroojerdi B, Battaglia F, Muellbacher W, Cohen LG (2001) Mechanisms underlying rapid experience-dependent plasticity in the human visual cortex. Proc Natl Acad Sci USA 98:14698–14701. https://doi.org/10.1073/pnas.251357198
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach (2nd ed). Ecol Model 172:109–139. https://doi.org/10.1016/j.ecolmodel.2003.11.004
Burnham KP, Anderson DR (2004) Multimodel inference: understanding AIC and BIC in model selection. Sociol Methods Res 33(2):261–304. https://doi.org/10.1177/0049124104268644
Casarotto S, Romero Lauro LJ, Bellina V, Casali AG, Rosanova M, Pigorini A, Defendi S, Mariotti M, Massimini M (2010) EEG responses to TMS are sensitive to changes in the perturbation parameters and repeatable over time. PLoS ONE 5(4):e10281
Casula EP, Tarantino V, Basso D, Arcara G, Marino G, Toffolo GM, Rothwell JC, Bisiacchi PS (2014) Low-frequency rTMS inhibitory effects in the primary motor cortex: Insights from TMS-evoked potentials. NeuroImage 98:225–232. https://doi.org/10.1016/j.neuroimage.2014.04.065
Cloninger CR (1994) The temperament and character inventory (TCI): a guide to its development and use. Center for Psychobiology of Personality, Washington University, St. Louis
Conde V, Tomasevic L, Akopian I, Stanek K, Saturnino GB, Thielscher A, Bergmann TO, Siebner HR (2019) The non-transcranial TMS-evoked potential is an inherent source of ambiguity in TMS-EEG studies. NeuroImage 185:300–312. https://doi.org/10.1016/j.neuroimage.2018.10.052
de Graaf TA, Duecker F, Stankevich Y, Oever S, Sack AT (2017) Brain stimulation seeing in the dark: phosphene thresholds with eyes open versus closed in the absence of visual inputs. Brain Stimul 10(4):828–835. https://doi.org/10.1016/j.brs.2017.04.127
Fierro B, Brighina F, Vitello G, Piazza A, Scalia S, Giglia G, Daniele O, Pascual-Leone A (2005) Modulatory effects of low- and high-frequency repetitive transcranial magnetic stimulation on visual cortex of healthy subjects undergoing light deprivation. J Physiol 565(Pt 2):659–665. https://doi.org/10.1113/jphysiol.2004.080184
Fossati A, Cloninger CR, Villa D, Borroni S, Grazioli F, Giarolli L, Battaglia M, Maffei C (2007) Reliability and validity of the Italian version of the temperament and character inventory-revised in an outpatient sample. Compr Psychiatry 48(4):380–387. https://doi.org/10.1016/j.comppsych.2007.02.003
Gilbert CD, Wiesel TN (1992) Receptive field dynimics in adult primary visual cortex. Nature 365:150–152
Glass JAYD, Crowder JV, Kennerdell JS, Merikangas JR (1977) Visually evoked potentials from occipital and precentral cortex in vsually deprived humans. Electroencephalogr Clin Neurophysiol 43:207–217
Gordon PC, Desideri D, Belardinelli P, Zrenner C, Ziemann U (2018) Brain stimulation comparison of cortical EEG responses to realistic sham versus real TMS of human motor cortex. Brain Stimul 11(6):1322–1330. https://doi.org/10.1016/j.brs.2018.08.003
Gothe J, Brandt SA, Irlbacher K, Röricht S, Sabel BA, Meyer BU (2002) Changes in visual cortex excitability in blind subjects as demonstrated by transcranial magnetic stimulation. Brain 125(3):479–490. https://doi.org/10.1093/brain/awf045
Herring JD, Thut G, Jensen O, Bergmann TO (2015) Attention modulates TMS-locked alpha oscillations in the visual cortex. J Neurosci 35(43):14435–14447. https://doi.org/10.1523/JNEUROSCI.1833-15.2015
Ilić TV, Meintzschel F, Cleff U, Ruge D, Kessler KR, Ziemann U (2002) Short-interval paired-pulse inhibition and facilitation of human motor cortex: the dimension of stimulus intensity. J Physiol 545(1):153–167. https://doi.org/10.1113/jphysiol.2002.030122
Kähkönen S, Komssi S, Wilenius J, Ilmoniemi RJ (2005) Prefrontal transcranial magnetic stimulation produces intensity-dependent EEG responses in humans. NeuroImage 24(4):955–960. https://doi.org/10.1016/j.neuroimage.2004.09.048
Kammer T, Beck S, Erb M, Grodd W (2001) The influence of current direction on phosphene thresholds evoked by transcranial magnetic stimulation. Clin Neurophysiol 112(11):2015–2021. https://doi.org/10.1016/S1388-2457(01)00673-3
Komssi S, Kähkönen S (2006) The novelty value of the combined use of electroencephalography and transcranial magnetic stimulation for neuroscience research. Brain Res Rev 52:9. https://doi.org/10.1016/j.brainresrev.2006.01.008
Komssi S, Kähkönen S, Ilmoniemi RJ (2004) The effect of stimulus intensity on brain responses evoked by transcranial magnetic stimulation. Hum Brain Mapp 21(3):154–164. https://doi.org/10.1002/hbm.10159
Kremlácek J, Radovan Š, Miroslav K, Jana S, Zuzana K, Jana L (2013) Spared cognitive processing of visual oddballs despite delayed visual evoked potentials in patient with partial recovery of vision after 53 years of blindness. Vis Res 81:1–5. https://doi.org/10.1016/j.visres.2012.12.013
Lioumis P, Kičić D, Savolainen P, Mäkelä JP, Kähkönen S (2009) Reproducibility of TMS—Evoked EEG responses. Hum Brain Mapp 30(4):1387–1396. https://doi.org/10.1002/hbm.20608
Marjerrison G, Keogh RP (1967) Electroencephalographic changes during brief periods of perceptual deprivation. Percept Mot Skills 24:611–615
Mazzi C, Savazzi S, Abrahamyan A, Ruzzoli M (2017) Reliability of TMS phosphene threshold estimation: toward a standardized protocol. Brain Stimul 10(3):609–617. https://doi.org/10.1016/j.brs.2017.01.582
Merabet L, Theoret H, Pascual-Leone A (2003) Transcranial magnetic stimulation as an investigative tool in the study of visual function. Optom Vis Sci 80(5):356–368
Miniussi C, Thut G (2010) Combining TMS and EEG offers new prospects in cognitive neuroscience. Brain Topogr 22(4):249–256. https://doi.org/10.1007/s10548-009-0083-8
Miniussi C, Brignani D, Pellicciari MC (2012) Combining transcranial electrical stimulation with electroencephalography: a multimodal approach. Clin EEG Neurosci 43(3):184–191. https://doi.org/10.1177/1550059412444976
Nikouline V, Ruohonen J, Ilmoniemi RJ (1999) The role of the coil click in TMS assessed with simultaneous EEG. Clin Neurophysiol 110(8):1325–1328. https://doi.org/10.1016/S1388-2457(99)00070-X
Parker AJ, Newsome WT (1998) Sense and the single neuron: probing the physiology of perception. Annu Rev Neurosci 21(1):227–277. https://doi.org/10.1146/annurev.neuro.21.1.227
Paulus W, Classen J, Cohen LG, Large CH, Lazzaro V Di, Nitsche M, Pascual-Leone A (2008) State of the art: pharmacologic effects on cortical excitability measures tested by transcranial magnetic stimulation. Brain Stimul 1(3):151–163. https://doi.org/10.1016/j.brs.2008.06.002
Pellicciari MC, Miniussi C, Ferrari C, Koch G, Bortoletto M (2016) Ongoing cumulative effects of single TMS pulses on corticospinal excitability: an intra- and inter-block investigation. Clin Neurophysiol 127(1):621–628. https://doi.org/10.1016/j.clinph.2015.03.002
Pitskel NB, Merabet LB, Ramos-Estebanez C, Kauffman T, Pascual-Leone A (2007) Time-dependent changes in cortical excitability after prolonged visual deprivation. NeuroReport 18(16):1703–1707. https://doi.org/10.1097/WNR.0b013e3282f0d2c1
Premoli I, Costantini A, Rivolta D, Biondi A, Richardson MP (2017) The effect of lamotrigine and levetiracetam on TMS-evoked EEG responses depends on stimulation intensity. Front Neurosci 11(585):1–7. https://doi.org/10.3389/fnins.2017.00585
Rogasch NC, Daskalakis ZJ, Fitzgerald PB (2013) Mechanisms underlying long-interval cortical inhibition in the human motor cortex: a TMS-EEG study. J Neurophysiol 109:89–98. https://doi.org/10.1152/jn.00762.2012
Romei V, Brodbeck V, Michel C, Amedi A, Pascual-Leone A, Thut G (2008) Spontaneous fluctuations in posterior alpha-band EEG activity reflect variability in excitability of human visual areas. Cereb Cortex 18(9):2010–2018. https://doi.org/10.1093/cercor/bhm229
Rosanova M, Casali AG, Bellina V, Resta F, Mariotti M, Massimini M (2009) Natural frequencies of human corticothalamic circuits. J Neurosci 29(24):7679–7685. https://doi.org/10.1523/JNEUROSCI.0445-09.2009
Rossi S, Hallett M, Rossini PM, Pascual-Leone A, The Safety of TMS Consensus Group (2009) Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol 120(12):2008–2039. https://doi.org/10.1016/j.clinph.2009.08.016
Schaworonkow N, Triesch J, Ziemann U, Zrenner C (2019) Brain Stimulation EEG-triggered TMS reveals stronger brain state-dependent modulation of motor evoked potentials at weaker stimulation intensities. Brain Stimul 12(1):110–118. https://doi.org/10.1016/j.brs.2018.09.009
Taylor PCJ, Walsh V, Eimer M (2010) The neural signature of phosphene perception. Hum Brain Mapp 31(9):1408–1417. https://doi.org/10.1002/hbm.20941
Théoret H, Merabet L, Pascual-Leone A (2004) Behavioral and neuroplastic changes in the blind: evidence for functionally relevant cross-modal interactions. J Physiol Paris 98(1–3):221–233. https://doi.org/10.1016/j.jphysparis.2004.03.009
Thut G, Miniussi C (2009) New insights into rhythmic brain activity from TMS-EEG studies. Trends Cognit Sci 13(4):182–189. https://doi.org/10.1016/j.tics.2009.01.004
Voineskos AN, Farzan F, Barr MS, Lobaugh NJ, Mulsant BH, Chen R, Fitzgerald, PB, Daskalakis ZJ (2010) The role of the corpus callosum in transcranial magnetic stimulation induced interhemispheric signal propagation. Biol Psychiatry 68(9):825–831. https://doi.org/10.1016/j.biopsych.2010.06.021
Acknowledgements
We would like to thank Clarissa Ferrari for her valuable help and suggestions about statistics.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Handling Editor: Gregor Thut.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This is one of several papers published together in Brain Topography on the “Special Topic: Cortical Network Analysis with EEG/MEG”.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10548_2019_715_MOESM1_ESM.tif
Supplementary material 1 (TIFF 6095 kb). Fig. S1 (Supplementary) Effect of TMS intensity on late-latency TEPs (N100, P150, P280) after DA and LA. Top: topographical maps of each component (amplitude range as shown in colorbar); squares indicate the channel where the maximum amplitude was recorded for each component selected to plot the average waveform below. Bottom: TEP grand-average as function of TMS intensity (low: green; medium: magenta; high: blue) and adaptation conditions (DA: first row; LA: second row); vertical bars indicate the latency of each component
Rights and permissions
About this article
Cite this article
Zazio, A., Bortoletto, M., Ruzzoli, M. et al. Perceptual and Physiological Consequences of Dark Adaptation: A TMS-EEG Study. Brain Topogr 32, 773–782 (2019). https://doi.org/10.1007/s10548-019-00715-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-019-00715-x