Abstract
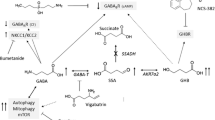
Recent studies have identified a role for supraphysiological gamma-aminobutyric acid (GABA) in the regulation of mechanistic target of rapamycin (mTOR), a protein kinase with pleiotropic roles in cellular development and homeostasis, including integration of growth factors and nutrient sensing and synaptic input in neurons (Lakhani et al. 2014; Vogel et al. 2015). Aldehyde dehydrogenase 5a1-deficient (aldh5a1 -/-) mice, the murine orthologue of human succinic semialdehyde dehydrogenase deficiency (SSADHD), manifest increased GABA that disrupts mitophagy and increases mitochondria number with enhanced oxidant stress. Treatment with the mTOR inhibitor, rapamycin, significantly attenuates these GABA-related anomalies. We extend those studies through characterization of additional rapamycin analog (rapalog) agents including temsirolimus, dual mTOR inhibitors [Torin 1 and 2 (Tor 1/ Tor 2), Ku-0063794, and XL-765], as well as mTOR-independent autophagy inducers [trehalose, tat-Beclin 1, tacrolimus (FK-506), and NF-449) in aldh5a1 -/- mice. Rapamycin, Tor 1, and Tor 2 rescued these mice from premature lethality associated with status epilepticus. XL-765 extended lifespan significantly and induced weight gain in aldh5a1 -/- mice; untreated aldh5a1 -/- mice failed to increase body mass. Expression profiling of animals rescued with Tor 1/Tor 2 and XL-765 revealed multiple instances of pharmacological compensation and/or correction of GABAergic and glutamatergic receptors, GABA/glutamate transporters, and GABA/glutamate-associated proteins, with Tor 2 and XL-765 showing optimal outcomes. Our studies lay the groundwork for further evaluation of mTOR inhibitors in aldh5a1 -/- mice, with therapeutic ramifications for heritable disorders of GABA and glutamate neurotransmission.







Similar content being viewed by others
Abbreviations
- GABA:
-
Gamma-aminobutyric acid
- Glu:
-
Glutamate
- Tor 1:
-
Torin 1
- Tor 2:
-
Torin 2
- SSADH:
-
Succinic semialdehyde dehydrogenase
- SSADHD:
-
Succinic semialdehyde dehydrogenase deficiency
- aldh5a1 :
-
aldehyde dehydrogenase 5a1 (identical to succinic semialdehyde dehydrogenase)
References
Bustin SA, Benes V, Garson JA et al (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55:611–622. doi:10.1373/clinchem.2008.112797
Buzzi A, Wu Y, Frantseva MV et al (2006) Succinic semialdehyde dehydrogenase deficiency: GABAB receptor-mediated function. Brain Res 1090:15–22. doi:10.1016/j.brainres.2006.02.131
Chen W-Y, Cheng Y-H, Hsieh N-H et al (2015) Physiologically based pharmacokinetic modeling of zinc oxide nanoparticles and zinc nitrate in mice. Int J Nanomedicine 10:6277–6292. doi:10.2147/IJN.S86785
Chowdhury GMI, Gupta M, Gibson KM et al (2007) Altered cerebral glucose and acetate metabolism in succinic semialdehyde dehydrogenase-deficient mice: evidence for glial dysfunction and reduced glutamate/glutamine cycling. J Neurochem 103:2077–2091. doi:10.1111/j.1471-4159.2007.04887.x
Cortez MA, Wu Y, Gibson KM, Snead OC (2004) Absence seizures in succinic semialdehyde dehydrogenase deficient mice: a model of juvenile absence epilepsy. Pharmacol Biochem Behav 79:547–553. doi:10.1016/j.pbb.2004.09.008
Donarum EA, Stephan DA, Larkin K et al (2006) Expression profiling reveals multiple myelin alterations in murine succinate semialdehyde dehydrogenase deficiency. J Inherit Metab Dis 29:143–156. doi:10.1007/s10545-006-0247-6
Dósa Z, Nieto-Gonzalez JL, Korshoej AR et al (2010) Effect of gene dosage on single-cell hippocampal electrophysiology in a murine model of SSADH deficiency (gamma-hydroxybutyric aciduria). Epilepsy Res 90:39–46. doi:10.1016/j.eplepsyres.2010.03.005
Drasbek KR, Vardya I, Delenclos M et al (2008) SSADH deficiency leads to elevated extracellular GABA levels and increased GABAergic neurotransmission in the mouse cerebral cortex. J Inherit Metab Dis 31:662–668. doi:10.1007/s10545-008-0941-7
Errington AC, Gibson KM, Crunelli V, Cope DW (2011) Aberrant GABA(A) receptor-mediated inhibition in cortico-thalamic networks of succinic semialdehyde dehydrogenase deficient mice. PLoS One 6:e19021. doi:10.1371/journal.pone.0019021
Ethofer T, Seeger U, Klose U et al (2004) Proton MR spectroscopy in succinic semialdehyde dehydrogenase deficiency. Neurology 62:1016–1018
Gupta M, Greven R, Jansen EEW et al (2002) Therapeutic intervention in mice deficient for succinate semialdehyde dehydrogenase (gamma-hydroxybutyric aciduria). J Pharmacol Exp Ther 302:180–187
Gupta M, Polinsky M, Senephansiri H et al (2004) Seizure evolution and amino acid imbalances in murine succinate semialdehyde dehydrogenase (SSADH) deficiency. Neurobiol Dis 16:556–562. doi:10.1016/j.nbd.2004.04.008
Han C, Wei S, Song Q et al (2016) Insulin stimulates goose liver cell growth by activating PI3K-AKT-mTOR signal pathway. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharmacol 38:558–570. doi:10.1159/000438650
Hogema BM, Gupta M, Senephansiri H et al (2001) Pharmacologic rescue of lethal seizures in mice deficient in succinate semialdehyde dehydrogenase. Nat Genet 29:212–216. doi:10.1038/ng727
Jansen EEW, Verhoeven NM, Jakobs C et al (2006) Increased guanidino species in murine and human succinate semialdehyde dehydrogenase (SSADH) deficiency. Biochim Biophys Acta 1762:494–498. doi:10.1016/j.bbadis.2006.01.006
Jansen EEW, Struys E, Jakobs C et al (2008) Neurotransmitter alterations in embryonic succinate semialdehyde dehydrogenase (SSADH) deficiency suggest a heightened excitatory state during development. BMC Dev Biol 8:112. doi:10.1186/1471-213X-8-112
Kang R, Zeh HJ, Lotze MT, Tang D (2011) The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 18:571–580. doi:10.1038/cdd.2010.191
Lafourcade CA, Lin TV, Feliciano DM et al (2013) Rheb activation in subventricular zone progenitors leads to heterotopia, ectopic neuronal differentiation, and rapamycin-sensitive olfactory micronodules and dendrite hypertrophy of newborn neurons. J Neurosci Off J Soc Neurosci 33:2419–2431. doi:10.1523/JNEUROSCI.1840-12.2013
Lakhani R, Vogel KR, Till A et al (2014) Defects in GABA metabolism affect selective autophagy pathways and are alleviated by mTOR inhibition. EMBO Mol Med 6:551–566. doi:10.1002/emmm.201303356
Lapalme-Remis S, Lewis EC, De Meulemeester C et al (2015) Natural history of succinic semialdehyde dehydrogenase deficiency through adulthood. Neurology. doi:10.1212/WNL.0000000000001906
Maitre M, Klein C, Mensah-Nyagan AG (2016) Mechanisms for the specific properties of γ-hydroxybutyrate in brain. Med Res Rev. doi:10.1002/med.21382
Morice E, Farley S, Poirier R et al (2013) Defective synaptic transmission and structure in the dentate gyrus and selective fear memory impairment in the Rsk2 mutant mouse model of Coffin-Lowry syndrome. Neurobiol Dis 58:156–168. doi:10.1016/j.nbd.2013.05.016
Nylen K, Velazquez JLP, Likhodii SS et al (2008) A ketogenic diet rescues the murine succinic semialdehyde dehydrogenase deficient phenotype. Exp Neurol 210:449–457. doi:10.1016/j.expneurol.2007.11.015
Parviz M, Vogel K, Gibson KM, Pearl PL (2014) Disorders of GABA metabolism: SSADH and GABA-transaminase deficiencies. J Pediatr Epilepsy 3:217–227. doi:10.3233/PEP-14097
Pearl PL, Gibson KM, Quezado Z et al (2009) Decreased GABA-A binding on FMZ-PET in succinic semialdehyde dehydrogenase deficiency. Neurology 73:423–429. doi:10.1212/WNL.0b013e3181b163a5
Pearl PL, Parviz M, Vogel K et al (2014) Inherited disorders of gamma-aminobutyric acid metabolism and advances in ALDH5A1 mutation identification. Dev Med Child Neurol. doi:10.1111/dmcn.12668
Pearl PL, Koenig M, Riviello J et al (2015) Novel intervention in GABA-transaminase deficiency
Reis J, Cohen LG, Pearl PL et al (2012) GABAB-ergic motor cortex dysfunction in SSADH deficiency. Neurology 79:47–54. doi:10.1212/WNL.0b013e31825dcf71
Rodan LH, Gibson KM, Pearl PL (2015) Clinical use of CSF neurotransmitters. Pediatr Neurol 53:277–286. doi:10.1016/j.pediatrneurol.2015.04.016
Rosenberg SA, Niglio SA, Salehomoum N et al (2015) Targeting glutamatergic signaling and the PI3 kinase pathway to halt melanoma progression. Transl Oncol 8:1–9. doi:10.1016/j.tranon.2014.11.001
Santinon G, Pocaterra A, Dupont S (2015) Control of YAP/TAZ activity by metabolic and nutrient-sensing pathways. Trends Cell Biol. doi:10.1016/j.tcb.2015.11.004
Shoji-Kawata S, Sumpter R, Leveno M et al (2013) Identification of a candidate therapeutic autophagy-inducing peptide. Nature 494:201–206. doi:10.1038/nature11866
Snead OC, Gibson KM (2005) Gamma-hydroxybutyric acid. N Engl J Med 352:2721–2732. doi:10.1056/NEJMra044047
Soto D, Altafaj X, Sindreu C, Bayés A (2014) Glutamate receptor mutations in psychiatric and neurodevelopmental disorders. Commun Integr Biol 7:e27887. doi:10.4161/cib.27887
Theilhaber J, Rakhade SN, Sudhalter J et al (2013) Gene expression profiling of a hypoxic seizure model of epilepsy suggests a role for mTOR and Wnt signaling in epileptogenesis. PLoS One 8:e74428. doi:10.1371/journal.pone.0074428
Vardya I, Drasbek KR, Gibson KM, Jensen K (2010) Plasticity of postsynaptic, but not presynaptic, GABAB receptors in SSADH deficient mice. Exp Neurol 225:114–122. doi:10.1016/j.expneurol.2010.05.022
Vaz SH, Lérias SR, Parreira S et al (2015) Adenosine A2A receptor activation is determinant for BDNF actions upon GABA and glutamate release from rat hippocampal synaptosomes. Purinergic Signal 11:607–612. doi:10.1007/s11302-015-9476-1
Vogel KR, Ainslie GR, Jansen EEW et al (2015) Torin 1 partially corrects vigabatrin-induced mitochondrial increase in mouse. Ann Clin Transl Neurol 2:699–706. doi:10.1002/acn3.200
Wang KY, Barker PB, Lin DDM (2015) A case of acute onset succinic semialdehyde dehydrogenase deficiency: neuroimaging findings and literature review. Childs Nerv Syst ChNS Off J Int Soc Pediatr Neurosurg. doi:10.1007/s00381-015-2942-9
Weston MC, Chen H, Swann JW (2012) Multiple roles for mammalian target of rapamycin signaling in both glutamatergic and GABAergic synaptic transmission. J Neurosci Off J Soc Neurosci 32:11441–11452. doi:10.1523/JNEUROSCI.1283-12.2012
Workman ER, Niere F, Raab-Graham KF (2013) mTORC1-dependent protein synthesis underlying rapid antidepressant effect requires GABABR signaling. Neuropharmacology 73:192–203. doi:10.1016/j.neuropharm.2013.05.037
Wu Y, Buzzi A, Frantseva M et al (2006) Status epilepticus in mice deficient for succinate semialdehyde dehydrogenase: GABAA receptor-mediated mechanisms. Ann Neurol 59:42–52. doi:10.1002/ana.20686
Acknowledgments
This work was supported in part by R21 NS 85369
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Animal rights
All institutional and national guidelines for the care and use of laboratory animals were followed.
Additional information
Communicated by: Niels Gregersen
Rights and permissions
About this article
Cite this article
Vogel, K.R., Ainslie, G.R. & Gibson, K.M. mTOR inhibitors rescue premature lethality and attenuate dysregulation of GABAergic/glutamatergic transcription in murine succinate semialdehyde dehydrogenase deficiency (SSADHD), a disorder of GABA metabolism. J Inherit Metab Dis 39, 877–886 (2016). https://doi.org/10.1007/s10545-016-9959-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-016-9959-4