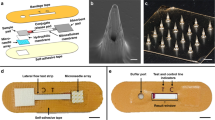
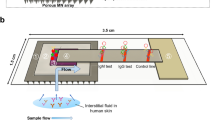
Abstract
There is a clinical need for differential diagnosis of the latent versus active stages of tuberculosis (TB) disease by a simple-to-administer test. Alpha-crystallin (Acr) and early secretory antigenic target-6 (ESAT-6) are protein biomarkers associated with the latent and active stages of TB, respectively, and could be used for differential diagnosis. We therefore developed a microneedle patch (MNP) designed for application to the skin to quantify Acr and ESAT-6 in dermal interstitial fluid by enzyme-linked immunosorbent assay (ELISA). We fabricated mechanically strong microneedles made of polystyrene and coated them with capture antibodies against Acr and ESAT-6. We then optimized assay sensitivity to achieve a limit of detection of 750 pg/ml and 3,020 pg/ml for Acr and ESAT-6, respectively. This study demonstrates the feasibility of an MNP-based ELISA for differential diagnosis of latent TB disease.
Graphical abstract






Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the article.
References
B. Acharya, A. Acharya, S. Gautam, S.P. Ghimire, G. Mishra, N. Parajuli, B. Sapkota, Advances in diagnosis of tuberculosis: an update into molecular diagnosis of Mycobacterium tuberculosis. Mol. Biol. Rep. 47, 4065 (2020). https://doi.org/10.1007/s11033-020-05413-7
C.E. Barry, H.I. Boshoff, V. Dartois, T. Dick, S. Ehrt, J. Flynn, D. Schnappinger, R.J. Wilkinson, D. Young, The spectrum of latent tuberculosis: rethinking the biology and intervention strategies. Nat. Rev. Microbiol. 7, 845 (2009). https://doi.org/10.1038/nrmicro2236
N.H. Bhuiyan, M.J. Uddin, J. Lee, J.H. Hong, J.S. Shim, An internet-of-disease system for COVID-19 testing using saliva by an AI-controlled microfluidic ELISA Device. Adv. Mater. Technol. B (2022). https://doi.org/10.1002/admt.202101690
T. Broger, M. Tsionksy, A. Mathew, T.L. Lowary, A. Pinter, T. Plisova, D. Bartlett, S. Barbero, C.M. Denkinger, E. Moreau, Sensitive electrochemiluminescence (ECL) immunoassays for detecting Lipoarabinomannan (LAM) and ESAT-6 in urine and serum from Tuberculosis patients. PLoS ONE 14, e0215443 (2019). https://doi.org/10.1371/journal.pone.0215443
J. Castro-Garza, P. Garcia-Jacobo, L.G. Rivera-Morales, F.D. Quinn, J. Barber, R. Karls, D. Haas, S. Helms, T. Gupta, H. Blumberg, J. Tapia, I. Luna-Cruz, A. Rendon, J. Vargas-Villarreal, L. Vera-Cabrera, Rodriguez-Padilla, Detection of anti-HspX antibodies and HspX protein in patient sera for the identification of recent latent infection by Mycobacterium tuberculosis. Plos ONE 12 (2017). https://doi.org/10.1371/journal.pone.0181714
CDC, Latent TB infection and TB disease. (2020). https://www.cdc.gov/tb/topic/basics/tbinfectiondisease.htm
J.W. Coffey, S.R. Corrie, M.A. Kendall, Rapid and selective sampling of IgG from skin in less than 1 min using a high surface area wearable immunoassay patch. Biomaterials 170, 49 (2018). https://doi.org/10.1016/j.biomaterials.2018.03.039
S.P. Davis, B.J. Landis, Z.H. Adams, M.G. Allen, M.R. Prausnitz, Insertion of microneedles into skin: measurement and prediction of insertion force and needle fracture force. J. Biomech. 37, 1155 (2004). https://doi.org/10.1016/j.jbiomech.2003.12.010
P. Esser, Principles in adsorption to polystyrene. Thermo. Sci. Nunc. Bull. 1 (1988)
N. Fogh-Andersen, B.M. Altura, B.T. Altura, O. Siggaard-Andersen, Composition of interstitial fluid. Clin. Chem. 41, 1522 (1995). https://doi.org/10.1093/clinchem/41.10.1522
M. Friedel, I.A. Thompson, G. Kasting, R. Polsky, D. Cunningham, H.T. Soh, J. Heikenfeld, Opportunities and challenges in the diagnostic utility of dermal interstitial fluid. Nat. Biomed. Eng. 1 (2023). https://doi.org/10.1038/s41551-022-00998-9
N.R. Gandhi, P. Nunn, K. Dheda, H.S. Schaaf, M. Zignol, D. van Soolingen, P. Jensen, J. Bayona, Multidrug-resistant and extensively drug-resistant tuberculosis: a threat to global control of tuberculosis. Lancet 375, 1830 (2010). https://doi.org/10.1016/S0140-6736(10)60410-2
E.J. Goldstein, E. Lee, R.S. Holzman, Evolution and current use of the tuberculin test. Clin. Infect. Dis. 34, 365 (2002). https://doi.org/10.1086/338149
M. Harboe, T. Oettinger, H.G. Wiker, I. Rosenkrands, P. Andersen, Evidence for occurrence of the ESAT-6 protein in Mycobacterium tuberculosis and virulent Mycobacterium bovis and for its absence in Mycobacterium bovis BCG. Infect. Immun. 64, 16 (1996). https://doi.org/10.1128/iai.64.1.16-22.1996
B.T. Ho, T.K. Roberts, S. Lucas, An overview on biodegradation of polystyrene and modified polystyrene: the microbial approach. Crit. Rev. Biotechnol. 38, 308 (2018). https://doi.org/10.1080/07388551.2017.1355293
E. Iona, M. Pardini, A. Mustazzolu, G. Piccaro, R. Nisini, L. Fattorini, F. Giannoni, Mycobacterium tuberculosis gene expression at different stages of hypoxia-induced dormancy and upon resuscitation. J. Microbiol. 54, 565 (2016). https://doi.org/10.1007/s12275-016-6150-4
S. Kiazyk, T.B. Ball, Latent tuberculosis infection: an overview. Can. Commun. Dis. Rep. 43, 62 (2017). https://doi.org/10.14745/ccdr.v43i34a01
Y. Kim, M.R. Prausnitz, Sensitive sensing of biomarkers in interstitial fluid. Nat. Biomed. Eng. 5, 3 (2021). https://doi.org/10.1038/s41551-020-00679-5
Li-cor, IRDye 800CW Streptavidin. (2023). https://www.licor.com/bio/reagents/irdye-800cw-streptavidin
J. Luan, A. Seth, R. Gupta, Z. Wang, P. Rathi, S. Cao, H. Gholami Derami, R. Tang, B. Xu, S. Achilefu, J.J. Morrissey, S. Singamaneni, Ultrabright fluorescent nanoscale labels for the femtomolar detection of analytes with standard bioassays. Nat. Biomed. Eng. 4, 518 (2020). https://doi.org/10.1038/s41551-020-0547-4
U. Mack, G. Migliori, M. Sester, H. Rieder, S. Ehlers, D. Goletti, A. Bossink, K. Magdorf, C. Hölscher, B. Kampmann, LTBI: latent tuberculosis infection or lasting immune responses to M. Tuberculosis? A TBNET consensus statement. Eur. Respir. J. 33, 956 (2009). https://doi.org/10.1183/09031936.00120908
E. MacLean, M. Kohli, S.F. Weber, A. Suresh, S.G. Schumacher, C.M. Denkinger, M. Pai, Advances in molecular diagnosis of tuberculosis. J. Clin. Microbiol. 58 (2020). https://doi.org/10.1128/JCM.01582-19
P.R. Miller, R.M. Taylor, B.Q. Tran, G. Boyd, T. Glaros, V.H. Chavez, R. Krishnakumar, A. Sinha, K. Poorey, K.P. Williams, S.S. Branda, J.T. Baca, R. Polsky, Extraction and biomolecular analysis of dermal interstitial fluid collected with hollow microneedlesis. Commun. Biol. 1 (2018). https://doi.org/10.1038/s42003-018-0170-z
D.A.J. Moore, C.A.W. Evans, R.H. Gilman, L. Caviedes, J. Coronel, A. Vivar, E. Sanchez, Y. Piñedo, J.C. Saravia, C. Salazar, R. Oberhelman, M.G. Hollm-Delgado, D. LaChira, A.R. Escombe, J.S. Friedland, Microscopic-observation drug-susceptibility assay for the diagnosis of TB. New. Engl. J. Med. 355, 1539 (2006). https://doi.org/10.1056/NEJMoa055524
L. Mou, R.H. Dong, B.F. Hu, Z.L. Li, J.J. Zhang, X.Y. Jian, Hierarchically structured microchip for point-of-care immunoassays with dynamic detection ranges. Lab Chip 19, 2750 (2019). https://doi.org/10.1039/c9lc00517j
D.A. Muller, S.R. Corrie, J. Coffey, P.R. Young, M.A. Kendall, Surface modified microprojection arrays for the selective extraction of the Dengue Virus NS1 protein as a marker for Disease. Anal. Chem. 84, 3262 (2012). https://doi.org/10.1021/ac2034387
E.A. Nardell, Tuberculosis (TB). (2022). https://www.merckmanuals.com/professional/infectious-diseases/mycobacteria/tuberculosis-tb
M. Pai, L.W. Riley, J.M. Colford, Interferon-γ assays in the immunodiagnosis of tuberculosis: a systematic review. Lancet Infect. Dis. 4, 761 (2004). https://doi.org/10.1016/S1473-3099(04)01206-X
J.E. Qualls, P.J. Murray, Immunometabolism within the tuberculosis granuloma: amino acids, hypoxia, and cellular respiration. Semin. Immunopathol. 38, 139 (2016). https://doi.org/10.1007/s00281-015-0534-0
E. Renard, Implantable continuous glucose sensors. Curr. Diabetes Rev. 4, 169 (2008). https://doi.org/10.2174/157339908785294406
B.J. Rogerson, Y.J. Jung, R. LaCourse, L. Ryan, N. Enright, R.J. North, Expression levels of antigen-encoding genes versus production levels of antigen-specific T cells during stationary level lung infection in mice. Immunology 118, 195 (2006). https://doi.org/10.1111/j.1365-2567.2006.02355.x
P.P. Samant, M.M. Niedzwiecki, N. Raviele, V. Tran, J. Mena-Lapaix, D.I. Walker, E.I. Felner, D.P. Jones, G.W. Miller, M.R. Prausnitz, Sampling interstitial fluid from human skin using a microneedle patch. Sci. Transl. Med. 12, eaaw0285 (2020). https://doi.org/10.1126/scitranslmed.aaw0285
A.R. Sholeye, A.A. Williams, D.T. Loots, A.M. Tutu van Furth, M. van der Kuip, S. Mason, Tuberculous granuloma: emerging insights from proteomics and metabolomics. Front. Neurol. 13, 804838 (2022). https://doi.org/10.3389/fneur.2022.804838
J.N. Stewart, H.N. Rivera, R. Karls, F.D. Quinn, J. Roman, C.A. Rivera-Marrero, Increased pathology in lungs of mice after infection with an alpha-crystallin mutant of Mycobacterium tuberculosis: changes in cathepsin proteases and certain cytokines. Microbiol. SGM 152, 233 (2006). https://doi.org/10.1099/mic.0.28275-0
S. Sukriti, M. Tauseef, P. Yazbeck, D. Mehta, Mechanisms regulating endothelial permeability. Pulm. Circ. 4, 535 (2014). https://doi.org/10.1086/677356
J.A. Tamada, S. Garg, L. Jovanovic, K.R. Pitzer, S. Fermi, R.O. Potts, C.R. Team, Noninvasive glucose monitoring - comprehensive clinical results. J. Am. Med. Assoc. 282, 1839 (1999). https://doi.org/10.1001/jama.282.19.1839
B.Q. Tran, P.R. Miller, R.M. Taylor, G. Boyd, P.M. Mach, C.N. Rosenzweig, J.T. Baca, R. Polsky, T. Glaros, Proteomic characterization of dermal interstitial fluid extracted using a Novel Microneedle-assisted technique. J. Proteome. Res. 17, 479 (2018). https://doi.org/10.1021/acs.jproteome.7b00642
Z.Y. Wang, J.Y. Luan, A. Seth, L. Liu, M.L. You, P. Gupta, P. Rathi, Y.X. Wang, S.S. Cao, Q.S. Jiang, X. Zhang, R. Gupta, Q.J. Zhou, J.J. Morrissey, E.L. Scheller, J.S. Rudra, S. Singamaneni, Microneedle patch for the ultrasensitive quantification of protein biomarkers in interstitial fluid. Nat. Biomed. Eng. 5, 64 (2021). https://doi.org/10.1038/s41551-020-00672-y
WHO, Tuberculosis. (2022). https://www.who.int/news-room/fact-sheets/detail/tuberculosis
B.F. Yin, X.H. Wan, W.K. Yue, T. Zhou, L.Y. Shi, S.B. Wang, X.D. Lin, A portable automated chip for simultaneous rapid point-of-care testing of multiple β-agonists. Biosens. Bioelectron. 239 (2023). https://doi.org/10.1016/j.bios.2023.115586
Y. Yuan, D.D. Crane, C.E. Barry 3rd., Stationary phase-associated protein expression in Mycobacterium tuberculosis: function of the mycobacterial alpha-crystallin homolog. J. Bacteriol. 178, 4484 (1996). https://doi.org/10.1128/jb.178.15.4484-4492.1996
Acknowledgements
We thank Donna Bondy for administrative support. The graphical abstract and Figure 3 were created with Biorender.com.
Funding
This work was supported by the National Institutes of Health (EB025499) and National Science Foundation (CBET-2224610).
Author information
Authors and Affiliations
Contributions
Y.K.: Methodology; investigation; data curation; formal analysis; writing – original draft. M.L.: Methodology; investigation. J.H.: Investigation; data curation. Z.W.: Investigation. R.G.: Investigation. Y.L.: Investigation. T.G.: Methodology; investigation; writing – review and editing, J.P.B.: Methodology. S.S.: Funding acquisition; supervision; writing – review and editing. F.Q.: Conceptualization; methodology; funding acquisition; supervision; writing – review and editing. M.R.P.: Conceptualization; methodology; funding acquisition; supervision; writing – review and editing.
Corresponding author
Ethics declarations
Competing interests
Mark R. Prausnitz is an inventor of patents, a paid advisor, and a founder/shareholder of companies developing microneedle-based products. The associated conflict of interest has been disclosed and is managed by Georgia Institute of Technology. Srikanth Singamaneni is one of the inventors on a pending patent related to plasmonic fluor technology, and the technology has been licensed by the Office of Technology Management at Washington University in St Louis to Auragent Bioscience LLC, which is developing plasmonic fluor products. S.S. is one of the co-founders and shareholders of Auragent Bioscience LLC. These potential conflicts of interest have been disclosed and are being managed by Washington University in St Louis.The other authors do not declare any conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, Y., Lewis, M.B., Hwang, J. et al. Microneedle patch-based enzyme-linked immunosorbent assay to quantify protein biomarkers of tuberculosis. Biomed Microdevices 26, 15 (2024). https://doi.org/10.1007/s10544-024-00694-2
Accepted:
Published:
DOI: https://doi.org/10.1007/s10544-024-00694-2