Abstract
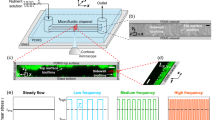
Biofilms are microbial colonies encased in an extracellular polymer matrix self-secreted through bacterial proliferation and differentiation. Biofilms exist almost everywhere such as sewers, rivers and oceans. In the fluid environment, the formation of biofilms is closely related to the relevant parameters of the flow field, such as the shear stress, the secondary flow, and the Reynolds number. In this paper, we use microfluidic channels made of polydimethylsiloxane to study the channel-geometry effect on Bacillus subtilis biofilms formation, such as the biofilm adhesion and structure. Our study shows that both the shear stress and the secondary flow play roles in the biofilm adhesion at the initial stage, the shear stress decides whether the biofilm adheres, if yes, then the secondary flow determines the adhesion rate. Our study further shows that after the biofilm forms, its structure evolves from loose to dense, with a concomitant 20-times rise in adhesion. Our study provides new insights into the adhesion of biofilms in natural and industrial fluid environments and helps understand the growth of biofilms.










Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
E. Banin, K.M. Brady, E.P. Greenberg, Chelator-Induced Dispersal and Killing of Pseudomonas aeruginosa Cells in a Biofilm. Appl. Environ. Microbiol. 72(3), 2064–2069 (2006). https://doi.org/10.1128/aem.72.3.2064-2069.2006
J.C. Conrad, R. Poling-Skutvik, Confined Flow: Consequences and Implications for Bacteria and Biofilms. Annu. Rev. Chem. Biomol. Eng. 9, 175–200 (2018). https://doi.org/10.1146/annurev-chembioeng-060817-084006
K. Drescher, Y. Shen, B.L. Bassler, H.A. Stone, Biofilm streamers cause catastrophic disruption of flow with consequences for environmental and medical systems. Proc. Natl. Acad. Sci. 110(11), 4345–4350 (2013). https://doi.org/10.1073/pnas.1300321110
R. Duddu, D.L. Chopp, B. Moran, A two-dimensional continuum model of biofilm growth incorporating fluid flow and shear stress based detachment. Biotechnol. Bioeng. 103(1), 92–104 (2009). https://doi.org/10.1002/bit.22233
E. Gjersing, S. Codd, J. Seymour, P. Stewart, Magnetic resonance microscopy analysis of advective transport in a biofilm reactor. Biotechnol. Bioeng. 89(7), 822–834 (2005). https://doi.org/10.1002/bit.20400
J. Kim, H.D. Park, S. Chung, Microfluidic approaches to bacterial biofilm formation. Molecules 17(8), 9818–9834 (2012). https://doi.org/10.3390/molecules17089818
M.K. Kim, K. Drescher, O.S. Pak, B.L. Bassler, H.A. Stone, Filaments in curved streamlines: Rapid formation of Staphylococcus aureus biofilm streamers. New. J. Phys. 16(6), 65024 (2014). https://doi.org/10.1088/1367-2630/16/6/065024
J. Klahre, H.C. Flemming, Monitoring of biofouling in papermill process waters. Water Res. 34(14), 3657–3665 (2000). https://doi.org/10.1016/S0043-1354(00)00094-4
R. Kolter, E.P. Greenberg, Microbial sciences: the superficial life of microbes. Nature 441(7091), 300–302 (2006). https://doi.org/10.1038/441300a
M. Krsmanovic, D. Biswas, H. Ali, A. Kumar, R. Ghosh, A.K. Dickerson, Hydrodynamics and surface properties influence biofilm proliferation. Adv. Colloid Interface Sci. 288, 102336 (2021). https://doi.org/10.1016/j.cis.2020.102336
N. Liu, T. Skauge, D. Landa-Marban, B. Hovland, B. Thorbjornsen, F.A. Radu, B.F. Vik, T. Baumann, G. Bodtker, Microfluidic study of effects of flow velocity and nutrient concentration on biofilm accumulation and adhesive strength in the flowing and no-flowing microchannels. J. Ind. Microbiol. Biotechnol. 46(6), 855–868 (2019). https://doi.org/10.1007/s10295-019-02161-x
B. Manz, F. Volke, D. Goll, H. Horn, Measuring local flow velocities and biofilm structure in biofilm systems with magnetic resonance imaging (MRI). Biotechnol. Bioeng. 84(4), 424–432 (2003). https://doi.org/10.1002/bit.10782
Y. Miura, Y. Watanabe, S. Okabe, Membrane biofouling in pilot-scale membrane bioreactors (MBRs) treating municipal wastewater: impact of biofilm formation. Environ. Sci. Technol. 41(2), 632–638 (2007). https://doi.org/10.1021/es0615371
M. Nemati, G.E. Jenneman, G. Voordouw, Mechanistic study of microbial control of hydrogen sulfide production in oil reservoirs. Biotechnol. Bioeng. 74(5), 424–434 (2001). https://doi.org/10.1002/bit.1133
G. O’Toole, H.B. Kaplan, R. Kolter, Biofilm formation as microbial development. Annu. Rev. Microbiol. 54, 49–79 (2000). https://doi.org/10.1146/annurev.micro.54.1.49
A. Park, H.-H. Jeong, J. Lee, K.P. Kim, C.-S. Lee, Effect of shear stress on the formation of bacterial biofilm in a microfluidic channel. BioChip J. 5(3), 236–241 (2011). https://doi.org/10.1007/s13206-011-5307-9
M. Parsek, P. Singh, Bacterial biofilms: an emerging link to disease pathogenesis. Annu. Rev. Microbiol. 57, 677–701 (2003). https://doi.org/10.1146/annurev.micro.57.030502.090720
R. Rusconi, S. Lecuyer, N. Autrusson, L. Guglielmini, H.A. Stone, Secondary flow as a mechanism for the formation of biofilm streamers. Biophys. J. 100(6), 1392–1399 (2011). https://doi.org/10.1016/j.bpj.2011.01.065
R. Rusconi, S. Lecuyer, L. Guglielmini, H.A. Stone, Laminar flow around corners triggers the formation of biofilm streamers. J. R. Soc. Interface 7(50), 1293–1299 (2010). https://doi.org/10.1098/rsif.2010.0096
M.M. Salek, S.M. Jones, R.J. Martinuzzi, The influence of flow cell geometry related shear stresses on the distribution, structure and susceptibility of Pseudomonas aeruginosa 01 biofilms. Biofouling 25(8), 711–725 (2009). https://doi.org/10.1080/08927010903114603
E Sherman, K Bayles, D Moormeier, J Endres, T Wei, Observations of Shear Stress Effects on Staphylococcus aureus Biofilm Formation. mSphere 4(4), (2019). https://doi.org/10.1128/mSphere.00372-19
R. Singh, D. Paul, R. Jain, Biofilms: implications in bioremediation. Trends Microbiol. 14(9), 389–397 (2006). https://doi.org/10.1016/j.tim.2006.07.001
P. Thomen, J. Robert, A. Monmeyran, A.F. Bitbol, C. Douarche, N. Henry, Bacterial biofilm under flow: First a physical struggle to stay, then a matter of breathing. PLoS One 12(4), e0175197 (2017). https://doi.org/10.1371/journal.pone.0175197
A Valiei, A Kumar, PP Mukherjee, Y Liu, T, Thundat A web of streamers: biofilm formation in a porous microfluidic device. Lab on a Chip 12(24), (2012). https://doi.org/10.1039/c2lc40815e
X. Wang, M. Hao, X. Du, G. Wang, J. Matsushita, The Mechanical Analysis of the Biofilm Streamer Nucleation and Geometry Characterization in Microfluidic Channels. Comput. Math. Methods Med. 2016, 7819403 (2016). https://doi.org/10.1155/2016/7819403
W.M. Weaver, V. Milisavljevic, J.F. Miller, D. Di Carlo, Fluid flow induces biofilm formation in Staphylococcus epidermidis polysaccharide intracellular adhesin-positive clinical isolates. Appl. Environ. Microbiol. 78(16), 5890–5896 (2012). https://doi.org/10.1128/AEM.01139-12
Z Yu, Fluid Mechanics - Revised edition, in chinese. (Meteorological press 1994), pp. 108–110
Y Zeng, Y Guo, Fluid mechanics,in chinese. (Hefei University of Technology Press 2008), pp. 78–82
W. Zimmerli, A. Trampuz, P. Ochsner, Prosthetic-joint infections. N. Engl. J. Med. 351(16), 1645–1654 (2004). https://doi.org/10.1056/NEJMra040181
Acknowledgements
The authors would like to thank Professor David A. Weitz and Professor Shmuel Rubinstein from Harvard University for their experimental support. The authors would like to thank the National Natural Science Foundation of China for funding support (11972074, 11772047 and 11620101001).
Funding
National Natural Science Foundation of China, 11972074, Xiaoling Wang, 11772047, Xiaoling Wang, 11620101001, Xiaoling Wang
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Author Xiaoling Wang, Author Song Liu, Author Fulin Dong, Author Duohuai Zhang, and Author Jinchang Zhang declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, S., Dong, F., Zhang, D. et al. Effect of microfluidic channel geometry on Bacillus subtilis biofilm formation. Biomed Microdevices 24, 11 (2022). https://doi.org/10.1007/s10544-022-00612-4
Accepted:
Published:
DOI: https://doi.org/10.1007/s10544-022-00612-4