Abstract
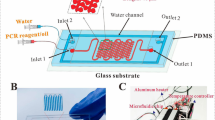
The reasons for restricting continuous flow polymerase chain reaction (CF-PCR) microfluidic chip from lab to application are that it is not portable and requires costly external precision pumps for sample injection. Herein, we employed water as the substitute for PCR solution, and investigated the effect of the cross-section, width-to-depth ratio, and the length ratio for three temperature zones of the micro channel on the thermal and flow distribution of fluid in micro tube by finite element analysis. Results show that the central velocity is uniform and stable velocity occupies the most if the cross-section is rectangular. The deviation between predefined temperature and theoretical temperature is slight and the fluid flux is the most if width-to-depth ratio is 1:1. It is suitable for the short DNA replication if the high temperature zone Wh is larger than the low temperature zone Wl, and vice versa. Then a portable CF-PCR microfluidic chip was fabricated and an automatic sample injection system was developed. As an application, we have successfully amplified the DNA of Treponema denticola in the chip within 8 min. Such a study may offer new insight into the design of CF-PCR microfluidic chip and promote it from lab-scale research to full-scale application.






Similar content being viewed by others
References
Q. Cao, M.C. Kim, C. Klapperich, Plastic microfluidic chip for continuous-flow polymerase chain reaction: Simulations and experiments. Biotechnol. J. 6, 177–184 (2011). https://doi.org/10.1002/biot.201000100
H. Chai, J. Xu, J. Xu, S. Ding, Y. Tang, P. Miao, Star trigon structure-aided DNA walker for amplified electrochemical detection of DNA. Electrochem. Commun. 99, 51–55 (2019). https://doi.org/10.1016/j.elecom.2019.01.003
P.-C. Chen, W. Fan, T.-K. Hoo, L.C.Z. Chan, Z. Wang, Simulation guided-design of a microfluidic thermal reactor for polymerase chain reaction. Chem. Eng. Res. Des. 90, 591–599 (2012). https://doi.org/10.1016/j.cherd.2011.09.008
P.J. Ezzo, C.W. Cutler, Microorganisms as risk indicators for periodontal disease. Periodontol 2000 32, 24–35 (2003). https://doi.org/10.1046/j.0906-6713.2003.03203.x
J.S. Farrar, C.T. Wittwer, Extreme PCR: Efficient and specific DNA amplification in 15–60 seconds. Clin. Chem. 61, 145–153 (2015). https://doi.org/10.1373/clinchem.2014.228304
L. Gui, C.L. Ren, Numeric simulation of heat transfer and electrokinetic flow in an electroosmosis-based continuous flow PCR chip. Anal. Chem. 78, 6215–6222 (2006). https://doi.org/10.1021/ac060553d
M. Hashimoto, P.-C. Chen, M.W. Mitchell, D.E. Nikitopoulos, S.A. Soper, M.C. Murphy, Rapid PCR in a continuous flow device. Lab. Chip. 4, 638–645 (2004). https://doi.org/10.1039/B406860B
B.J. Hindson et al., High-throughput droplet digital PCR system for absolute quantitation of DNA copy number. Anal. Chem. 83, 8604–8610 (2011). https://doi.org/10.1021/ac202028g
J.A. Kim, J.Y. Lee, S. Seong, S.H. Cha, S.H. Lee, J.J. Kim, T.H. Park, Fabrication and characterization of a PDMS–glass hybrid continuous-flow PCR chip. Biochem. Eng. J. 29, 91–97 (2006). https://doi.org/10.1016/j.bej.2005.02.032
M.U. Kopp, A.J. Mello, A. Manz, Chemical amplification: continuous-flow PCR on a chip. Science 280, 1046–1048 (1998). https://doi.org/10.1126/science.280.5366.1046
M. Krishnan, N. Agrawal, M.A. Burns, V.M. Ugaz, Reactions and fluidics in miniaturized natural convection systems. Anal. Chem. 76, 6254–6265 (2004). https://doi.org/10.1021/ac049323u
M. Krishnan, V.M. Ugaz, M.A. Burns, PCR in a Rayleigh-Bénard convection cell. Science 298, 793–793 (2002). https://doi.org/10.1126/science.298.5594.793
S. Kumar, M.A. Cartas-Ayala, T. Thorsen, Thermal modeling and design analysis of a continuous flow microfluidic chip. Int. J. Therm. Sci. 67, 72–86 (2013). https://doi.org/10.1016/j.ijthermalsci.2012.12.003
Z. Li, R. Ju, S. Sekine, D. Zhang, S. Zhuang, Y. Yamaguchi, All-in-one microfluidic device for on-site diagnosis of pathogens based on integrated continuous flow PCR and electrophoresis biochip. Lab Chip. 19, 2663–2668 (2019). https://doi.org/10.1039/C9LC00305C
Z. Li, B. Yang, S. Sekine, S. Zhuang, D. Zhang, Y. Yamaguchi, Alignment and Counting of Mitochondria based on Capillary. Electrophor. Sens. Actuat. B Chem. 265, 110–114 (2018). https://doi.org/10.1016/j.snb.2018.03.032
Z. Li, Y. Zhao, D. Zhang, S. Zhuang, Y. Yamaguchi, The development of a portable buoyancy-driven PCR system and its evaluation by capillary electrophoresis. Sens. Actuat. B Chem. 230, 779–784 (2016). https://doi.org/10.1016/j.snb.2016.02.143
G. Mohiuddin Mala, D. Li, Flow characteristics of water in microtubes. Int. J. Heat Fluid Flow 20, 142–148 (1999). https://doi.org/10.1016/S0142-727X(98)10043-7
T. Notomi, H. Okayama, H. Masubuchi, T. Yonekawa, K. Watanabe, N. Amino, T. Hase, Loop-mediated isothermal amplification of DNA. Nucl. Acids Res. 28, e63 (2000). https://doi.org/10.1093/nar/28.12.e63
X. Qiu et al., A smartphone-based point-of-care diagnosis of H1N1 with microfluidic convection PCR. Microsyst. Technol. 23, 2951–2956 (2017a). https://doi.org/10.1007/s00542-016-2979-z
X. Qiu et al., Real-time capillary convective PCR based on horizontal thermal convection. Microfluid. Nanofluid. 23, 39 (2019). https://doi.org/10.1007/s10404-019-2207-0
X. Qiu et al., Instrument-free point-of-care molecular diagnosis of H1N1 based on microfluidic convective PCR. Sens. Actuat. B Chem. 243, 738–744 (2017b). https://doi.org/10.1016/j.snb.2016.12.058
V.K. Rajendran, P. Bakthavathsalam, P.L. Bergquist, A. Sunna, A portable nucleic acid detection system using natural convection combined with a smartphone. Biosens. Bioelectron. 134, 68–75 (2019)
C. Schabmueller, M. Lee, A. Evans, A. Brunnschweiler, G. Ensell, D. Leslie, Closed chamber PCR chips for DNA amplification. Eng. Sci. Edu. J. 9, 259–264 (2000). https://doi.org/10.1049/esej:20000605
P.D. Schloss, D. Gevers, S.L. Westcott, Reducing the effects of PCR amplification and sequencing artifacts on 16S rRNA-based studies. PLoS One 6, e27310 (2011). https://doi.org/10.1371/journal.pone.0027310
S.L. Spurgeon, R.C. Jones, R. Ramakrishnan, High throughput gene expression measurement with real time PCR in a microfluidic dynamic array. PLoS One 3, e1662 (2008). https://doi.org/10.1371/journal.pone.0001662
H. Tachibana, M. Saito, K. Tsuji, K. Yamanaka, L.Q. Hoa, E. Tamiya, Self-propelled continuous-flow PCR in capillary-driven microfluidic device: Microfluidic behavior and DNA amplification. Sens. Actuat. B Chem. 206, 303–310 (2015). https://doi.org/10.1016/j.snb.2014.09.004
N.B. Trung, M. Saito, H. Takabayashi, P.H. Viet, E. Tamiya, Y. Takamura, Multi-chamber PCR chip with simple liquid introduction utilizing the gas permeability of polydimethylsiloxane. Sens. Actuat. B Chem. 149, 284–290 (2010). https://doi.org/10.1016/j.snb.2010.06.013
Acknowledgements
This work was supported by Science and Technology Commission of Shanghai Municipality (China) (No.18441900400) and National Natural Science Foundation of China (No. 61775140). We also gratefully acknowledge financial support from University of Shanghai for Science and Technology (No.2017KJFZ049).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, Z., Li, Y., Sekine, S. et al. Design and fabrication of portable continuous flow PCR microfluidic chip for DNA replication. Biomed Microdevices 22, 5 (2020). https://doi.org/10.1007/s10544-019-0457-y
Published:
DOI: https://doi.org/10.1007/s10544-019-0457-y