Abstract
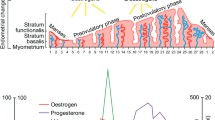
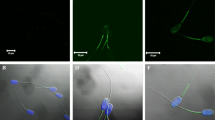
Lactoferrin (LF) is present in the oviduct, reduces in vitro gamete interaction, and affects sperm capacitation parameters in humans. Our aim was to investigate LF actions on further stages of the reproductive process in the Wistar rat model. Motile sperm were obtained from cauda epididymis to assess LF binding by direct immunofluorescence and LF effect on acrosome reaction (AR) using a Coomassie blue staining. After ovarian hyperstimulation of female rats, oocytes were surgically recovered and coincubated with motile sperm and different doses of LF to estimate the in vitro fertilization (IVF) rate. To evaluate the LF effect on pregnancy and embryo implantation, female rats (80 days old) were placed with males and received daily intraperitoneal injections of LF during one complete estrous cycle (pregnancy experiments) or during the first 8 gestational days (implantation experiments). The number of pregnant females and live born pups was recorded after labor. Moreover, the number of implantation sites was registered during the implantation period. LF was able to bind to the sperm head, midpiece, and tail. 10 and 100 μg/ml LF stimulated the AR but reduced the IVF rate. The administration of 100 and 200 mg/kg LF significantly decreased the number of implantation sites and the litter size, whereas 100 mg/kg LF declined the pregnancy rate. The results suggest that LF might interfere with the reproductive process, possibly interfering with gamete interaction or inducing a premature AR; nevertheless, the mechanisms involved are yet to be elucidated.





Similar content being viewed by others
Change history
20 June 2023
A Correction to this paper has been published: https://doi.org/10.1007/s10534-023-00515-8
References
Actor JK, Hwang S-A, Kruzel ML (2009) Lactoferrin as a natural immune modulator. Curr Pharm Des 15:1956–1973. https://doi.org/10.2174/138161209788453202
Aktuğ H, Bozok Çetintaş V, Uysal A et al (2013) Evaluation of the effects of STZ-induced diabetes on in vitro fertilization and early embryogenesis processes. J Diabetes Res 2013:1–9. https://doi.org/10.1155/2013/603813
Artym J, Zimecki M, Kruzel ML (2004) Effect of lactoferrin on the methotrexate-induced suppression of the cellular and humoral immune response in mice. Anticancer Res 24:3831–3836
Avrech O, Fisch B, Shalgi R (1997) Acrosomal status of human spermatozoa after follicular fluid or calcium ionophore challenge in relation to semen parameters and fertilizing capacity in vitro. Andrologia 29:97–101. https://doi.org/10.1111/j.1439-0272.1997.tb00470.x
Baveye S, Elass E, Mazurier J et al (1999) Lactoferrin: a multifunctional glycoprotein involved in the modulation of the inflammatory process. Clin Chem Lab Med 37(3):281–286. https://doi.org/10.1515/CCLM.1999.049
Bendahmane M, Zeng H-T, Tulsiani DRP (2002) Assessment of acrosomal status in rat spermatozoa: studies on carbohydrate and non-carbohydrate agonists. Arch Biochem Biophys 404:38–47. https://doi.org/10.1016/S0003-9861(02)00278-3
Berkestedt I, Herwald H, Ljunggren L et al (2010) Elevated plasma levels of antimicrobial polypeptides in patients with severe sepsis. J Innate Immun 2:478–482. https://doi.org/10.1159/000317036
Brock JH (2002) The physiology of lactoferrin. Biochem Cell Biol 80:1–6. https://doi.org/10.1139/o01-212
Chandra A, Martinez GM, Mosher WD et al (2005) Fertility, family planning, and reproductive health of U.S. women: data from the 2002 National Survey of Family Growth. Vital Heal Stat Ser 23:1–160
Chen Y, Wang B, Yang C et al (2021) Functional correlates and impact of dietary lactoferrin intervention and its concentration-dependence on neurodevelopment and cognition in neonatal piglets. Mol Nutr Food Res 65:e2001099. https://doi.org/10.1002/mnfr.202001099
de la Rosa G, Yang D, Tewary P et al (2008) Lactoferrin acts as an alarmin to promote the recruitment and activation of APCs and antigen-specific immune responses. J Immunol 180:6868–6876. https://doi.org/10.4049/jimmunol.180.10.6868
Elass-Rochard E, Legrand D, Salmon V et al (1998) Lactoferrin inhibits the endotoxin interaction with CD14 by competition with the lipopolysaccharide-binding protein. Infect Immun 66:486–491. https://doi.org/10.1128/IAI.66.2.486-491.1998
Gadella BM (2013) Dynamic regulation of sperm interactions with the zona pellucida prior to and after fertilisation. Reprod Fertil Dev 25:26–37. https://doi.org/10.1071/RD12277
García-Montoya IA, Cendón TS, Arévalo-Gallegos S, Rascón-Cruz Q (2012) Lactoferrin a multiple bioactive protein: an overview. Biochim Biophys Acta 1820:226–236. https://doi.org/10.1016/j.bbagen.2011.06.018
Ghersevich S, Massa E, Zumoffen C (2015) Oviductal secretion and gamete interaction. Reproduction. https://doi.org/10.1530/REP-14-0145
Ghersevich S, Filocco L, Massa E, Zumoffen C (2017) Chapter 8: Multiple effects of lactoferrin depend on diverse receptors for the protein in different cell types. In: Shalu V (ed) Lactoferrin: structure, biological functions, health benefits and clinical applications. NOVA Publisher, New York, pp 147–184
He Z, Xie M, Li QQ et al (2022) Research progress on the microregulatory mechanisms of fertilization: a review. In Vivo 36:2002–2013. https://doi.org/10.21873/invivo.12926
Hegazy R, Salama A, Mansour D, Hassan A (2016) Renoprotective effect of lactoferrin against chromium-induced acute kidney injury in rats: involvement of IL-18 and IGF-1 inhibition. PLoS ONE 11:e0151486. https://doi.org/10.1371/journal.pone.0151486
Hershlag A, Scholl GM, Jacob A et al (1998) Mannose ligand receptor assay as a test to predict fertilization in vitro: a prospective study. Fertil Steril 70:482–491. https://doi.org/10.1016/S0015-0282(98)00199-X
Hirohashi N, Yanagimachi R (2018) Sperm acrosome reaction: its site and role in fertilization. Biol Reprod 99:127–133. https://doi.org/10.1093/biolre/ioy045
Hung C-M, Yeh C-C, Chen H-L et al (2010) Porcine lactoferrin administration enhances peripheral lymphocyte proliferation and assists infectious bursal disease vaccination in native chickens. Vaccine 28:2895–2902. https://doi.org/10.1016/j.vaccine.2010.01.066
Ickowicz D, Finkelstein M, Breitbart H (2012) Mechanism of sperm capacitation and the acrosome reaction: role of protein kinases. Asian J Androl 14:816–821. https://doi.org/10.1038/aja.2012.81
Jin M, Fujiwara E, Kakiuchi Y et al (2011) Most fertilizing mouse spermatozoa begin their acrosome reaction before contact with the zona pellucida during in vitro fertilization. Proc Natl Acad Sci USA 108:4892–4896. https://doi.org/10.1073/pnas.1018202108
Kanwar JR, Roy K, Patel Y et al (2015) Multifunctional iron bound lactoferrin and nanomedicinal approaches to enhance its bioactive functions. Molecules. https://doi.org/10.3390/molecules20069703
Kell DB, Heyden EL, Pretorius E (2020) The biology of lactoferrin, an iron-binding protein that can help defend against viruses and bacteria. Front Immunol 11:1221. https://doi.org/10.3389/fimmu.2020.01221
Kim S-M, Kim J-S (2017) A review of mechanisms of implantation. Dev Reprod 21:351–359. https://doi.org/10.12717/dr.2017.21.4.351
Kobayashi J, Sasaki A, Watanabe A et al (2021) Effects of exogenous lactoferrin on characteristics and functions of bovine epididymal, ejaculated and frozen-thawed sperm. Anim Sci J 92:e13538. https://doi.org/10.1111/asj.13538
Kruzel ML, Zimecki M, Actor JK (2017) Lactoferrin in a context of inflammation-induced pathology. Front Immunol 8:1438. https://doi.org/10.3389/fimmu.2017.01438
Latorre D, Berlutti F, Valenti P et al (2012) LF immunomodulatory strategies: mastering bacterial endotoxin. Biochem Cell Biol 90:269–278. https://doi.org/10.1139/o11-059
Leaver RB (2016) Male infertility: an overview of causes and treatment options. Br J Nurs 25:S35–S40. https://doi.org/10.12968/bjon.2016.25.18.S35
Legrand D, Elass E, Pierce A, Mazurier J (2004) Lactoferrin and host defence: an overview of its immuno-modulating and anti-inflammatory properties. Biometals an Int J Role Met Ions Biol Biochem Med 17:225–229. https://doi.org/10.1023/b:biom.0000027696.48707.42
Legrand D, Elass E, Carpentier M, Mazurier J (2005) Lactoferrin: a modulator of immune and infl ammatory responses. Cell Mol Life Sci 62:2549–2559. https://doi.org/10.1007/s00018-005-5370-2
Legrand D (2012) Lactoferrin, a key molecule in immune and inflammatory processes. Biochem Cell Biol 90:252–268. https://doi.org/10.1139/o11-056
Legrand D (2016) Overview of lactoferrin as a natural immune modulator. J Pediatr 173(Suppl):S10–S15. https://doi.org/10.1016/j.jpeds.2016.02.071
Lepanto MS, Rosa L, Paesano R et al (2019) Lactoferrin in aseptic and septic inflammation. Molecules 24(7):1323. https://doi.org/10.3390/molecules24071323
Levay PF, Viljoen M (1995) Lactoferrin: a general review. Haematologica 80:252–267
Liu Y, Teng CT (1992) Estrogen response module of the mouse lactoferrin gene contains overlapping chicken ovalbumin upstream promoter transcription factor and estrogen receptor-binding elements. Mol Endocrinol 6:355–364. https://doi.org/10.1210/mend.6.3.1584212
Mahé C, Zlotkowska AM, Reynaud K et al (2021) Sperm migration, selection, survival, and fertilizing ability in the mammalian oviduct. Biol Reprod 105:317–331. https://doi.org/10.1093/biolre/ioab105
Marey MA, Yousef MS, Kowsar R et al (2016) Local immune system in oviduct physiology and pathophysiology: attack or tolerance? Domest Anim Endocrinol 56:S204–S211. https://doi.org/10.1016/j.domaniend.2016.02.005
Maritati M, Comar M, Zanotta N et al (2017) Influence of vaginal lactoferrin administration on amniotic fluid cytokines and its role against inflammatory complications of pregnancy. J Inflamm 14:5. https://doi.org/10.1186/s12950-017-0152-9
Massa E, Ghersevich S (2019) Milestones in the reproductive process: involvement of the oviduct environment. In: Berhardt L (ed) Advances in medicine and biology. Nova Science Publishers Inc, New York
Massa E, Pelusa F, Lo Celso A et al (2021) Lactoferrin levels in cervical fluid from in vitro fertilization (IVF) patients — correlation with IVF parameters. Biochem Cell Biol 99:91–96. https://doi.org/10.1139/bcb-2020-0098
Mrozikiewicz AE, Ożarowski M, Jędrzejczak P (2021) Biomolecular markers of recurrent implantation failure—A review. Int J Mol Sci 22:10082. https://doi.org/10.3390/ijms221810082
Onal A, Kayalioğlu G, Parlar A et al (2010) Effect of prolonged administration of bovine lactoferrin in neuropathic pain: involvement of opioid receptors, nitric oxide and TNF-alpha. Life Sci 86:251–259. https://doi.org/10.1016/j.lfs.2009.12.007
Paccola C, Resende C, Stumpp T et al (2013) The rat estrous cycle revisited: a quantitative and qualitative analysis. Anim Reprod 10:677–683
Pang T, Leach ST, Katz T et al (2014) Fecal biomarkers of intestinal health and disease in children. Front Pediatr 2:1–12. https://doi.org/10.3389/fped.2014.00006
Presti S, Manti S, Parisi GF et al (2021) Lactoferrin: cytokine modulation and application in clinical practice. J Clin Med 10(23):5482. https://doi.org/10.3390/jcm10235482
Sanches E, van de Looij Y, Sow S et al (2021) Dose-dependent neuroprotective effects of bovine lactoferrin following neonatal hypoxia-ischemia in the immature rat brain. Nutrients 13(11):3880. https://doi.org/10.3390/nu13113880
Sun H, Gong T-T, Jiang Y-T et al (2019) Global, regional, and national prevalence and disability-adjusted life-years for infertility in 195 countries and territories, 1990–2017: results from a global burden of disease study, 2017. Aging 11:10952–10991. https://doi.org/10.18632/aging.102497
Teng CT (2002) Differential expression and estrogen response of lactoferrin gene in the female reproductive tract of mouse, rat, and hamster. Biol Reprod 67:1439–1449. https://doi.org/10.1095/biolreprod.101.002089
Teng CT, Gladwell W, Beard C et al (2002) Lactoferrin gene expression is estrogen responsive in human and rhesus monkey endometrium. Mol Hum Reprod 8:58–67
Tsubota A, Yoshikawa T, Nariai K et al (2008) Bovine lactoferrin potently inhibits liver mitochondrial 8-OHdG levels and retrieves hepatic OGG1 activities in Long-Evans Cinnamon rats. J Hepatol 48:486–493. https://doi.org/10.1016/j.jhep.2007.11.013
Valenti P, Antonini G (2005) Lactoferrin: an important host defence against microbial and viral attack. Cell Mol Life Sci 62:2576–2587. https://doi.org/10.1007/s00018-005-5372-0
Varano G, Lombardi A, Cantini G et al (2008) Src activation triggers capacitation and acrosome reaction but not motility in human spermatozoa. Hum Reprod 23:2652–2662. https://doi.org/10.1093/humrep/den314
Vogel HJ (2012) Lactoferrin, a bird's eye view. Biochem Cell Biol 90:233–244. https://doi.org/10.1139/o2012-016
Wang P, Liu B, Wang Z et al (2011) Characterization of lactoferrin receptor on human spermatozoa. Reprod Biomed Online 22:155–161. https://doi.org/10.1016/j.rbmo.2010.10.003
Ward PP, Paz E, Conneely OM (2005) Multifunctional roles of lactoferrin: a critical overview. Cell Mol Life Sci 62:2540–2548. https://doi.org/10.1007/s00018-005-5369-8
Yanagimachi R (2022) Mysteries and unsolved problems of mammalian fertilization and related topics. Biol Reprod 106:644–675. https://doi.org/10.1093/biolre/ioac037
Zimecki M, Mazurier J, Machnicki M et al (1991) Immunostimulatory activity of lactotransferrin and maturation of CD4- CD8- murine thymocytes. Immunol Lett 30:119–123. https://doi.org/10.1016/0165-2478(91)90099-v
Zumoffen CM, Gil R, Caille AM et al (2013) A protein isolated from human oviductal tissue in vitro secretion, identified as human lactoferrin, interacts with spermatozoa and oocytes and modulates gamete interaction. Hum Reprod 28:1297–1308. https://doi.org/10.1093/humrep/det016
Zumoffen CM, Massa E, Caille AM et al (2015) Effects of lactoferrin, a protein present in the female reproductive tract, on parameters of human sperm capacitation and gamete interaction. Andrology 3:1068–1075. https://doi.org/10.1111/andr.12093
Acknowledgements
This study was partially supported by a grant from SECyT UNR (PID‐BIO581).
Funding
This study was partially supported by a grant from SECyT UNR (PID‐BIO581).
Author information
Authors and Affiliations
Contributions
EM, AG, MM, and MJM performed the experiments and analyzed the results. EM, FP and SG contributed to the experimental design, methodology, and analysis of the results. EM, AG, MM, and SG wrote the main manuscript and prepared the figures. FP and SG supervised the study and contributed to funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: all authors' given and family names were transposed.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Massa, E., Gola, A., Moriconi, M. et al. Lactoferrin affects in vitro and in vivo fertilization and implantation in rats. Biometals 36, 575–585 (2023). https://doi.org/10.1007/s10534-022-00460-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-022-00460-y