Abstract
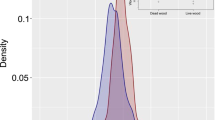
Deadwood, long recognized as playing an important role in storing carbon and releasing it as CO2 in forest ecosystems, is more recently drawing attention for its potential role in the cycling of other greenhouse trace gases. Across three Northeastern and Central US forests, mean methane (CH4) concentrations in deadwood were 23 times atmospheric levels (43.0 μL L−1 ± 12.3; mean ± SE), indicating a lower bound, mean radial wood surface area flux of ~6 × 10−4 μmol CH4 m−2 s−1. Site, decay class, log diameter, and species were all highly significant predictors of CH4 abundance in deadwood, and diameter and decay class interacted as important controls limiting CH4 concentrations in the smallest and most decayed logs. Nitrous oxide (N2O) concentrations were negatively correlated with CH4 (r2 = −0.20, p < 0.001) and on average ~25 % lower than ambient (276.9 nL L−1 ± 2.9; mean ± SE), indicating net consumption of nitrous oxide. Oxygen (O2) concentrations were uniformly near anaerobic (355.8 μL L−1 ±1.2; mean ± SE), and CO2 was elevated from atmospheric (9336.9 μL L−1 ± 600.6; mean ± SE). Most notably, our observations that CH4 concentrations were highest in the least decayed wood, may suggest that methanogenesis is not fuelled by structural wood decomposition but rather by consumption of more labile nonstructural carbohydrates.



Similar content being viewed by others
References
Adair EC, Parton WJ, Del Grosso SJ, Silver WL, Harmon ME, Hall SA, Burke IC, Hart SC (2008) Simple three-pool model accurately describes patterns of long-term litter decomposition in diverse climates. Glob Chang Biol 14(11):2636–2660
Aulakh M, Doran J, Mosier A (1992) Soil denitrification—significance, measurement, and effects of management. Advances in soil science. Springer, New York, pp 1–57
Barford CC, Wofsy SC, Goulden ML, Munger JW, Pyle EH, Urbanski SP, Hutyra L, Saleska SR, Fitzjarrald D, Moore K (2001) Factors controlling long- and short-term sequestration of atmospheric CO2 in a mid-latitude forest. Science 294(5547):1688–1691
Boddy L (2001) Fungal community ecology and wood decomposition processes in angiosperms: from standing tree to complete decay of coarse woody debris. Ecol Bull 49:43–56
Bruhn D, Møller IM, Mikkelsen TN, Ambus P (2012) Terrestrial plant methane production and emission. Physiol Plant 144(3):201–209
Carmichael MJ, Bernhardt ES, Bräuer SL, Smith WK (2014) The role of vegetation in methane flux to the atmosphere: should vegetation be included as a distinct category in the global methane budget? Biogeochemistry 119(1–3):1–24
Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE (2009) Towards a worldwide wood economics spectrum. Ecol Lett 12(4):351–366
Conrad R (1994) Compensation concentration as critical variable for regulating the flux of trace gases between soil and atmosphere. Biogeochemistry 27(3):155–170
Cornwell WK, Cornelissen JHC, Allison SD, Bauhus J, Eggleton P, Preston CM, Scarff F, Weedon JT, Wirth C, Zanne AE (2009) Plant traits and wood fates across the globe: rotted, burned, or consumed? Glob Chang Biol 15(10):2431–2449
Covey KR, Wood SA, Warren RJ II, Lee X, Bradford MA (2012) Elevated methane concentrations in trees of an upland forest. Geophys Res Lett 39(15):L15705
Cowling EB, Merrill W (1966) Nitrogen in wood and its role in wood deterioration. Can J Bot 44(11):1539–1554
Ettwig KF, Butler MK, Le Paslier D, Pelletier E, Mangenot S, Kuypers MMM, Schreiber F, Dutilh BE, Zedelius J, de Beer D, Gloerich J, Wessels HJCT, van Alen T, Luesken F, Wu ML, van de Pas-Schoonen KT, Op den Camp HJM, Janssen-Megens EM, Francoijs K-J, Stunnenberg H, Weissenbach J, Jetten MSM, Strous M (2010) Nitrite-driven anaerobic methane oxidation by oxygenic bacteria. Nature 464(7288):543–548
Ettwig KF, Shima S, Van De Pas-Schoonen KT, Kahnt J, Medema MH, Op Den Camp HJM, Jetten MSM, Strous M (2008) Denitrifying bacteria anaerobically oxidize methane in the absence of Archaea. Environ Microbiol 10(11):3164–3173
van Geffen KG, Poorter L, Sass-Klaassen U, van Logtestijn RSP, Cornelissen JHC (2010) The trait contribution to wood decomposition rates of 15 Neotropical tree species. Ecology 91(12):3686–3697
Heilmann-Clausen J, Christensen M (2004) Does size matter?: on the importance of various dead wood fractions for fungal diversity in Danish beech forests. For Ecol Manag 201(1):105–117
Hietala A, Dörsch P, Kvaalen H, Solheim H (2015) Carbon dioxide and methane formation in Norway spruce stems infected by white-rot fungi. Forests 6(9):3304
Hoch G, Richter A, KÖRner C (2003) Non-structural carbon compounds in temperate forest trees. Plant Cell Environ 26(7):1067–1081
Idol TW, Figler RA, Pope PE, Ponder F Jr (2001) Characterization of coarse woody debris across a 100 year chronosequence of upland oak–hickory forests. For Ecol Manag 149(1–3):153–161
Keppler F, Hamilton JTG, Braß M, Rockmann T (2006) Methane emissions from terrestrial plants under aerobic conditions. Nature 439(7073):187–191
Klüber HD, Conrad R (1998) Effects of nitrate, nitrite, NO and N2O on methanogenesis and other redox processes in anoxic rice field soil. FEMS Microbiol Ecol 25(3):301–318
Koike I, Hattori A (1975) Energy yield of denitrification: an estimate from growth yield in continuous cultures of Pseudomonas denitrificans under nitrate-, nitrite-and nitrous oxide-limited conditions. Microbiology 88(1):11–19
Krüger M, Beckmann S, Engelen B, Thielemann T, Cramer B, Schippers A, Cypionka H (2008) Microbial methane formation from hard coal and timber in an abandoned coal mine. Geomicrobiol J 25(6):315–321
Lenhart K, Bunge M, Ratering S, Neu TR, Schüttmann I, Greule M, Kammann C, Schnell S, Müller C, Zorn H, Keppler F (2012) Evidence for methane production by saprotrophic fungi. Nat Commun 3:1046
Luyssaert S, Schulze ED, Borner A, Knohl A, Hessenmoller D, Law BE, Ciais P, Grace J (2008) Old-growth forests as global carbon sinks. Nature 455(7210):213–215
Mackensen J, Bauhus J, Webber E (2003) Decomposition rates of coarse woody debris: a review with particular emphasis on Australian tree species. Aust J Bot 51(1):27–37
Massman WJ (1998) A review of the molecular diffusivities of H2O, CO2, CH4, CO, O3, SO2, NH3, N2O, NO, and NO2 in air, O2 and N2 near STP. Atmos Environ 32(6):1111–1127
Miller FR, McBee GG (1993) Genetics and management of physiologic systems of sorghum for biomass production. Biomass Bioenergy 5(1):41–49
Moorhead DL, Sinsabaugh RL (2006) A theoretical model of litter decay and microbial interaction. Ecol Monogr 76(2):151–174
Moorman TB, Parkin TB, Kaspar TC, Jaynes DB (2010) Denitrification activity, wood loss, and N2O emissions over 9 years from a wood chip bioreactor. Ecol Eng 36(11):1567–1574
Nord-Larsen T, Bergstedt A, Farver O, Heding N (2011) Drying of firewood: the effect of harvesting time, tree species and shelter of stacked wood. Biomass Bioenergy 35(7):2993–2998
Oberle B, Dunham K, Milo AM, Walton M, Young DF, Zanne AE (2014) Progressive, idiosyncratic changes in wood hardness during decay: implications for dead wood inventory and cycling. For Ecol Manag 323:1–9
Pacala SW, Hurtt GC, Baker D, Peylin P, Houghton RA, Birdsey RA, Heath L, Sundquist ET, Stallard RF, Ciais P, Moorcroft P, Caspersen JP, Shevliakova E, Moore B, Kohlmaier G, Holland E, Gloor M, Harmon ME, Fan S-M, Sarmiento JL, Goodale CL, Schimel D, Field CB (2001) Consistent land- and atmosphere-based US carbon sink estimates. Science 292(5525):2316–2320
Pan Y, Birdsey RA, Fang J, Houghton R, Kauppi PE, Kurz WA, Phillips OL, Shvidenko A, Lewis SL, Canadell JG, Ciais P, Jackson RB, Pacala SW, McGuire AD, Piao S, Rautiainen A, Sitch S, Hayes D (2011) A large and persistent carbon sink in the world’s forests. Science 333(6045):988–993
Panday U, Singh JS (1982) Leaf-litter decomposition in an oak-conifer forest in Himalaya: the effects of climate and chemical composition. Forestry 55(1):47–59
Pangala SR, Hornibrook ERC, Gowing DJ, Gauci V (2015) The contribution of trees to ecosystem methane emissions in a temperate forested wetland. Glob Chang Biol 21(7):2642–2654
Preston CM, Trofymow JA, Niu J, Fyfe CA (1998) 13CPMAS-NMR spectroscopy and chemical analysis of coarse woody debris in coastal forests of Vancouver Island. For Ecol Manag 111(1):51–68
R Development Core Team (2015) R: A language and environment for statistical computing. In: R Foundation for Statistical Computing, Vienna
Raghoebarsing AA, Pol A, van de Pas-Schoonen KT, Smolders AJP, Ettwig KF, Rijpstra WIC, Schouten S, Damste JSS, Op den Camp HJM, Jetten MSM, Strous M (2006) A microbial consortium couples anaerobic methane oxidation to denitrification. Nature 440(7086):918–921
Richardson AD, Carbone MS, Huggett BA, Furze ME, Czimczik CI, Walker JC, Xu X, Schaberg PG, Murakami P (2015) Distribution and mixing of old and new nonstructural carbon in two temperate trees. New Phytol 206(2):590–597
Richardson AD, Carbone MS, Keenan TF, Czimczik CI, Hollinger DY, Murakami P, Schaberg PG, Xu X (2013) Seasonal dynamics and age of stemwood nonstructural carbohydrates in temperate forest trees. New Phytol 197(3):850–861
Schwarze FWMR (2007) Wood decay under the microscope. Fungal Biol Rev 21(4):133–170
Stams A, Plugge C (2010) The microbiology of methanogenesis. In: Smith P, Reay D, Van Amstel A (eds) Methane and climate change. Taylor & Francis, New York, pp 14–26
Steppe K, Saveyn A, McGuire MA, Lemeur R, Teskey RO (2007) Resistance to radial CO2 diffusion contributes to between-tree variation in CO2 efflux of Populus deltoides stems. Funct Plant Biol 34(9):785–792
Teskey RO, Saveyn A, Steppe K, McGuire MA (2008) Origin, fate and significance of CO2 in tree stems. New Phytol 177(1):17–32
Tugtas AE, Pavlostathis SG (2007) Inhibitory effects of nitrogen oxides on a mixed methanogenic culture. Biotechnol Bioeng 96(3):444–455
Tugtas AE, Tezel U, Pavlostathis SG (2010) A comprehensive model of simultaneous denitrification and methanogenic fermentation processes. Biotechnol Bioeng 105(1):98–108
Vigano I, Röckmann T, Holzinger R, van Dijk A, Keppler F, Greule M, Brand WA, Geilmann H, van Weelden H (2009) The stable isotope signature of methane emitted from plant material under UV irradiation. Atmos Environ 43(35):5637–5646
Weedon JT, Cornwell WK, Cornelissen JH, Zanne AE, Wirth C, Coomes DA (2009) Global meta-analysis of wood decomposition rates: a role for trait variation among tree species? Ecol Lett 12(1):45–56
Wieder WR, Bonan GB, Allison SD (2013) Global soil carbon projections are improved by modelling microbial processes. Nat Clim Chang 3(10):909–912
Wofsy SC (2001) Where has all the carbon gone? Science 292(5525):2261–2263
Woodall CW, Monleon VJ (2008) Sampling protocol, estimation, and analysis procedures for the down woody materials indicator of the FIA program. General Technical Report NRS, vol 22. U.S. Department of Agriculture, Forest Service, Northern Research Station, Newtown Square, PA, 68 p
Woodcock D, Shier A (2002) Wood specific gravity and its radial variations: the many ways to make a tree. Trees 16(6):437–443
Würth MR, Peláez-Riedl S, Wright SJ, Körner C (2005) Non-structural carbohydrate pools in a tropical forest. Oecologia 143(1):11–24
Zanne AE, Oberle B, Dunham KM, Milo AM, Walton ML, Young DF (2015) A deteriorating state of affairs: how endogenous and exogenous factors determine plant decay rates. J Ecol 103(6):1421–1431
Zeikus J, Ward JC (1974) Methane formation in living trees: a microbial origin. Science 184(4142):1181
Zohoun S, Agoua E, Degan G, Perre P (2003) An experimental correction proposed for an accurate determination of mass diffusivity of wood in steady regime. Heat Mass Transf 39(2):147–155
Acknowledgments
The authors acknowledge the staff of the Yale Myers Forest, and the Tyson Research Center. Shannon Murray, Nora Hawkins, Jacqueline Kulig, and Joanna Parkman assisted with fieldwork at the Yale Myers Forest. This work was supported by National Science Foundation awards granted to KRC and MAB (NSF DGE-1405135), and to AEZ (NSF DEB-1302797). Additional funding was provided by the Yale Institute for Biospheric Studies, the Beck Fund, and the Middlebury Senior Research Project Supplement Award.
Author’s Contributions
KRC, CDO, MAB, XL, CPB, ML, and AEZ planned and designed the research. KRC, CPB, BO, DSM, TWC, and MCD conducted fieldwork. KRC, DSM, and CB analyzed data and designed graphics. KRC, MAB, BO, and BS wrote the manuscript. All authors contributed comments to earlier drafts.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Karsten Kalbitz.
Rights and permissions
About this article
Cite this article
Covey, K.R., de Mesquita, C.P.B., Oberle, B. et al. Greenhouse trace gases in deadwood. Biogeochemistry 130, 215–226 (2016). https://doi.org/10.1007/s10533-016-0253-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-016-0253-1