Abstract
Complementarity is crucial when prioritizing sites for biodiversity conservation. Networks of conservation corridors (CCs) can contribute to regional representativeness by complementing biodiversity features included in existing protected areas (PAs). We ask whether criteria important for CC management and design are effective at prioritizing complementary sites, and how the consideration of species represented in PAs influence criteria performance. We focused on species turnover of generalist and specialist dragonflies across 88 riverine sites. Criteria assessed included site-level estimates of dragonfly species richness, estimates of local habitat quality and corridor width. Measures of local habitat quality were based on either dragonfly indicator species or proportion of alien vegetation. Results showed that CCs complement dragonfly diversity in PAs by contributing unrepresented generalist species. Of the criteria, corridor width was the most efficient at prioritizing complementary sites, while prioritization based on dragonfly indicator species or species richness underperformed. When aiming to prioritize CC sites that also complement sites situated in established PAs, wide corridors with low levels of alien vegetation should be favoured.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Off-reserve areas are important for ongoing biodiversity conservation (Dahal et al. 2014; van Schalkwyk et al. 2019; Kane et al. 2022). Areas beyond protected area (PA) boundaries, such as conservation corridors (CCs), can add to regional connectivity and reduce the negative impacts of habitat loss and fragmentation by supporting dispersal among otherwise isolated habitats (Gilbert-Norton et al. 2010). Networks of CCs (also referred to as ecological networks) (Jongman and Pungetti 2004; Samways 2007) are especially beneficial when regionally employed alongside other ecological components such as stepping-stone habitats, core areas, and buffer zones. Together, these components enable ecosystems to exist in human-dominated landscapes (Forman 1995; Hess and Fischer 2001).
Corridors can further help maintain regional biodiversity by complementing biodiversity features (e.g., taxonomic diversity) already represented in established reserves. Important for identifying complementary sites is the concept of species turnover: the substitution (i.e., replacement) of species by other species across sites (Baselga and Orme 2012). For example, significant differences in species assemblages between networks of CCs and PAs suggest potential complementary value of CC sites (Pryke et al. 2015). Complementarity can aid in identifying areas for conservation investment, i.e. conservation prioritization (Wilson et al. 2009). Importantly, complementarity can improve representativeness in an efficient manner, e.g., enhance representativeness with minimum management cost (Kukkala and Moilanen 2013). Representativeness is related to the need for reserves to represent the full variety of biodiversity, and involves the extent to which biodiversity features occur within a set of sites (Margules and Sarkar 2007; Heink 2009). Improving representativeness can be invaluable in regions where PA establishment is biased (e.g., concentrated in economically unviable areas), and where existing PAs add to the conservation of certain features that are already well-represented at the expense of others.
Site-level estimates of diversity are routinely used to guide CC management and planning (Samways et al. 2010; van Schalkwyk et al. 2022). For example, wide corridors representing good quality habitat are associated with high species richness (Pryke and Samways 2003; van Schalkwyk et al. 2022). However, complementarity requires information on biodiversity content (rather than summary statistics such as species richness) as well as the joint consideration of multiple sites (Wilson et al. 2009). Dragonflies and damselflies (Odonata) (from here on referred to as dragonflies) have been extensively studied as indicators of ecosystem change, both terrestrial and riparian (Clausnitzer 2003; French and McCauley 2019; Vorster et al. 2020), and have also been incorporated in CC assessments (Kietzka et al. 2015; Mossman et al. 2015; Pryke et al. 2015). To date, however, not many assessments incorporate dragonfly species turnover (but see Pryke et al. (2015), and it is unknown whether promoting site-scale species diversity in CCs will encourage complementarity. In addition, conservation priority of potential sites will change as other priority areas are established and expanded, and so the process of conservation prioritization should be informed by what is already conserved (Wilson et al. 2009). The evaluation of CC site complementarity should therefore consider the influence of species represented in existing PAs.
We evaluate the efficiency of different criteria for prioritizing complementary corridors aimed at enhancing taxonomic representativeness of South African dragonflies. We focused on adult male dragonflies in streams of varying quality (in both corridors dissecting forestry plantations and PAs) (Kietzka et al. 2021a) and assess criteria that are often used at the site-scale to guide assessments, management, and planning of CC sites: species richness, local habitat quality, and corridor width (Hansen et al. 2018; van Schalkwyk et al. 2020; Kietzka et al. 2021a). Corridors were ranked according to these criteria, and we compared how different criteria influenced the gain in species representation with increasing number of selected sits. The efficiency of criteria for prioritizing complementary CC sites was assessed across two planning contexts: species represented across CC sites, and species represented across both CC and PA sites together.
Dragonfly species differ in their sensitivity to habitat degradation and disturbance. Some species are generalists that are tolerant and even thrive in transformed habitats, others are highly sensitive specialists that occur mostly in wild areas (Samways and Simaika 2016). We expect CC complementarity and criteria efficiency to differ between specialist and generalist species. By contributing to the decline of specialist species, landscape-scale land use change may contribute to assemblage homogenization and loss of turnover (Ball-Damerow et al. 2014). Corridors are situated in more transformed landscapes than PAs, and we therefore predict that CCs will complement the dragonfly species present in existing PAs through better representation of generalist species. Overall and irrespective of species sensitivity, we expect corridor width to be the most efficient criterion at selecting complementary sites, as wider corridors likely experience lower extinction rates, higher immigration rates and greater habitat heterogeneity than narrower corridors (MacArthur and Wilson 1967; Lees and Peres 2008). Furthermore, we predict that criteria representing changes to local habitat quality will be more efficient at prioritizing complementary CC sites for specialist species than generalist species.
Methods
Study area and sites
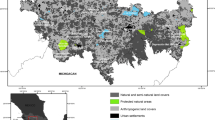
The study region is situated in the KwaZulu-Natal Midlands, South Africa (Fig. 1). Networks of CCs in this region consist of natural and semi-natural vegetation that intersect plantation forestry blocks of alien trees. The data used were collected for a previous study (Kietzka et al. 2021a). A methodological description is provided below, and additional details can be found in Kietzka et al. (2021a).
In total, 110 study sites were selected along rivers situated in CCs (55 sites) and PAs (55 sites). These sites varied in factors previously shown to be important for South African dragonfly diversity, including corridor width (in the case of CC sites) and invasive plant species cover (Samways and Sharratt 2010; Kietzka et al. 2015, 2017; Deacon et al. 2019). Not all 110 sites were considered in the present study. We removed six PA sites that were situated far from other sites so that inter-site geographical distances were more similar among CC and PA sites, so as to not artificially inflate species turnover between CCs and PAs. We removed six sites where no dragonflies were observed (see “Dragonfly sampling and species sensitivity“ section below for details on sampling methods) and 10 sites that were situated close to each other (< 500 m). This left 43 PA sites, and 45 CC sites (88 sites in total). Minimum distance between any two sites was ~ 574 m, the mean was ~ 46 km, and the maximum distance was 106 km.
Dragonfly sampling and species sensitivity
A site consisted of a 100 m transect parallel to a river and included the 5 m area to each side of the transect. Each site was visited twice between 09h00 and 15h00 on sunny, windless days during the summer months (between January 2018 and March 2018). During each visit, male dragonflies were surveyed by two observers for one hour using close-focus binoculars (total of four observation hours per site). Where specimens were difficult to identify in flight, they were captured using a hand-net and kept as voucher specimens, which are held at Stellenbosch University, South Africa.
Species were split into two groups that differ in sensitivity to changes in the natural environment. This classification was based on the Habitat Sensitivity Sub-Index (HSS) of the Dragonfly Biotic Index, which uses the presence of dragonfly species (mostly adults but also includes complementary data on other life stages where it is necessary to establish total local species composition) to assess freshwater conditions (Samways and Simaika 2016). The HSS relates to how sensitive a species is to changes in its natural environment and is based on historic occurrence records in natural versus disturbed sites. Tolerant generalist species include species that are more common in artificial waterbodies than natural environments, as well as species equally common across both natural and disturbed habitats (species that have a HSS score of either 0 or 1). Sensitive specialist species include species that are mostly observed in fully natural environments, as well as species highly sensitive to environmental change (species that have a HSS score of either 2 or 3).
Criteria for site prioritization
We compared four criteria for prioritizing CC sites: (1) dragonfly species richness, (2) local habitat quality based on dragonfly indicator taxa, (3) local habitat quality based on the percentage alien vegetation at a site, and (4) corridor width (Table 1).
Species richness was based on the observed species richness for each site (summed over both site surveys). A local estimate of habitat quality was based on the HSS (Samways and Simaika 2016). For each site, the HSS score for all observed species were summed and then divided by the number of species observed at that site. More details regarding the sub-index scores of the Dragonfly Biotic Index are provided by Samways and Simaika (2016).
Invasive alien vegetation remains an important threat to insect diversity (McGeoch 2002; Cardoso et al. 2020), woody species being especially problematic for dragonflies in South Africa (Samways and Sharratt 2010). A visual estimate of alien vegetation cover for each site was based on the most common species in the area, which included (in order of prevalence across sites): American bramble (Rubus cuneifolius), black wattle (Acacia mearnsii), bugweed (Solanum mauritianum), and scotch thistle (Cirsium vulgare). Each 100 m transect was subdivided into 10 plots. At each plot, two observers independently estimated alien vegetation cover, according to the aforementioned categories. Estimates recorded by the two observers were averaged and used to calculate the percentage of alien vegetation cover for each 100 m site.
Corridor width is an important design variable in conservation planning (Noss 1987; Beier 2018). Wider corridors are associated with higher terrestrial and aquatic arthropods species diversity (Pryke and Samways 2012; Kietzka et al. 2021a). We measured corridor width from aerial photographs as the linear minimum distance between two opposing corridor edges that intersects the centre of a transect.
None of the criteria showed spatial autocorrelation (Figure S1), nor were any correlated with each other (Figure S2).
Analyses
All analyses were conducted in R version 3.6.0 (R Core Team 2020).
Sampling efficiency
To determine dragonfly sampling efficiency, we used sampling completeness, which was calculated using the final slope of the species accumulation curve. This slope indicates the expected gain in diversity when sampling a new individual, and was calculated with the following formula:
Here, ns denotes the total number of individuals, and ns−1 represents the number of individuals at the point in the curve where the last single species was added (Cardoso et al. 2011). This final slope was calculated using the R package BAT (Cardoso et al. 2021).
Criteria efficiency for representation
The efficiency of criteria to prioritize complementary CC sites was assessed using a pattern-based approach based on the Environmental Diversity strategy of Faith and Walker (1996a). Sites were projected in a two-dimensional space (or ‘ordination space’) where the inter-site distances represent species turnover (i.e., sites situated further apart have greater species turnover) (Fig. 2). These distances were used to infer the relative complementary value of a site (e.g., sites that have unique species compositions would separate out further from the other sites and so have higher complementary value) and measure representativeness with increasing number of selected sites.
General workflow for assessing the relative efficiency of different criteria prioritizing complementary conservation corridor (CC) sites. With increasing number of selected sites, forgone biodiversity is calculated iteratively as the distance between each hypothetical demand point and the nearest site. PA, protected area
We consider two scenarios when prioritizing CC sites: (1) representativeness of only networks of CCs and (2) representativeness of both CC and PA sites together. In the first scenario, we assess whether criteria can prioritize complementary sites across CCs. Here the ordination space represents only CCs (45 sites). In the second, we assess whether criteria prioritizing CC sites improve efficiency of existing PAs. Here criteria efficiency is assessed in reference to representativeness of CCs and PAs combined, and the ordination space represents both CCs and PAs (88 sites).
As input to ordination, species turnover was measured as between-site dissimilarities due to species replacement (Simpson dissimilarity calculated from species presence/absence data) (Baselga 2010). To define the two-dimensional space, we used multidimensional scaling (with 1000 iterations and 20 minimum random starts). The borders of this ordination space (or polytope) was defined using a convex hull (Faith and Walker 1996b). Within this polytope space, we placed hypothetical continuously spaced locations (or ‘demand points’). The distance between all these hypothetical demand points and their nearest site was used to infer site complementarity. We placed twice as many demand points as sites included in the ordination (90 when ordination considers only CC sites, 176 when ordination considers both CC and PA sites).
To maximize representation, site selection should aim to minimise the distance between all hypothetical demand points and their nearest site, which represents the amount of ‘forgone biodiversity’ (Faith 1995). Sites were prioritized for inclusion in the selection process according to their criteria ranking (species richness, HSS, alien vegetation cover, or corridor width). As each site was added to the selection, we calculated the sum of the distance between each demand point and its nearest site. This distance was plotted against the number of selected sites.
To assess the complementary value of CCs to existing PAs, we considered the ordination space representing all sites (CCs and PAs), and compared random selection of CCs sites, PA sites, as well as CC and PA sites combined. Here we do not consider random site selection a worst-case scenario, but rather better than what would be expected under real world conditions, where conservation sites are rarely selected at random (Ferrier 2002; Delso et al. 2021). We also use random site selection as a benchmark to compare the ability of the four different criteria to select complementary sites. If a criterion performs better than random site selection, we consider this an indication of high efficiency. Where representativeness only considers CCs, random site selection of CC sites was the reference. Where representativeness considers both CCs and PAs, random selection of CC and PA sites combined was the reference. To this last scenario, we also considered random selection of only PA sites to assess criteria efficiency in reference to existing PAs.
For each of the two scenarios, the procedure described above was repeated 10 times, each time reprojecting the between-site dissimilarities in ordination space. Where criteria had similar values across different sites, we randomized the order of these same-value sites in each repetition. These analyses were repeated for all species, as well as subsets representing specialist and generalist species. Between-site dissimilarities in species replacement were calculated using the R package betapart (Baselga et al. 2021) and multidimensional scaling conducted using the vegan R package (Oksanen et al. 2019).
Results
Sampling efficiency
In total, 36 species and 1175 individuals of adult male dragonflies were observed across the 88 sites. Table S1 provides a species abundance list. The species accumulation curve reached an asymptote (Figure S3). Based on the final slope, the expected gain in diversity was low (0.003). The slope suggests that an additional ~ 333 dragonfly specimens would need to be recorded to increase the number of observed species.
We discarded from the data species with less than 10 observed individuals. These sparse observations could be due to low species detectability and we therefore consider them unreliable. Their removal also discouraged convergence issues during multidimensional scaling. Removal of these species did not significantly alter the ranking of sites according to species richness or the HSS. This reduced the number of species used in analyses to 20. Of these, nine were classified as specialists and 11 as generalists (Table S1).
Complementary value of CCs
Figure 3 shows the curves tracing successive increase in species representation as more CC sites are included in the selection and compares random site selection of CCs, PAs, as well as CCs and PAs combined. In all cases, the ordination space (representativeness) considers species sampled from all sites (CCs and PAs). The graphs show that CCs complement dragonfly diversity in PAs by contributing to the representation of generalist species, indicated by the more rapid decline in forgone biodiversity when CCs and PAs are considered together, compared to PA sites alone.
Forgone biodiversity with increasing number of randomly selected sites. Representation considered both conservation corridor (CC) and protected area (PA) sites. All—all species. Specialist—Specialist species. Generalist—Generalist species. The curve represents the gain in representation that corresponds to a decrease in forgone biodiversity as more sites are selected
Criteria efficiency
Figure 4 compares efficiency of site prioritization according to the four criteria (species richness, HSS, alien vegetation cover, or corridor width) across two contexts: species present across CCs, and species present across both CCs and PAs.
Values of forgone biodiversity with increasing number of selected conservation corridor (CC) sites, for all species (All), specialist species (Specialist), and generalist species (Generalist). Two contexts are considered: species present across CCs, and species present across both CCs and protected areas (PAs). The curve represents the gain in representation that corresponds to a decrease in forgone biodiversity as more sites are selected according to four criteria: species richness (Spp), habitat quality based on the Habitat Sensitivity Sub-Index (HSS), habitat quality based on alien vegetation cover (AlienVeg), and corridor width (Width)
For all species, the efficiency of criteria for prioritizing complementary sites was generally comparable to random site selection (Figures S4 and S5). However, prioritization based on the HSS did not perform as well as random selection at representing species across CCs, and prioritization based on species richness performed more poorly than random selection at representing species across CCs and PAs combined.
For subsets of the species assemblage representing specialist and generalist species, the efficiency of criteria for prioritizing complementary sites was generally comparable to random site selection (Figures S6 and S7). For sensitive species across CCs, site prioritization based on species richness or the HSS did not perform as well as random site selection at the start of the selection procedure.
Of the four criteria, corridor width was the best performer in prioritizing complementary CC sites (for all species, as well as specialist and generalist species separately), and occasionally performed better than random site selection. This was especially the case for generalist species when considering representation across both CC and PA sites.
Discussion
We found CCs complement dragonfly assemblages in existing PAs through contributing to the representation of generalist species (i.e., species that are less responsive to habitat degradation). Of the criteria assessed, we found corridor width to be the most effective at prioritizing complementary sites, across either CCs or CCs and PAs combined. Furthermore, we did not find habitat quality to be more efficient at selecting complementary CC sites for specialist species compared to generalist species.
Studies have reported a global decrease in specialist species, which has been related to their higher sensitivity to degraded and fragmented habitats (Korkeamäki and Suhonen 2002; Colles et al. 2009; Morelli et al. 2020). These specialist species therefore tend to be located in less fragmented and less disturbed landscapes (Devictor et al. 2008). At the same time, specialist species can be more competitive in these natural environments (Büchi and Vuilleumier 2014), which could explain why generalist species are better represented across networks of CCs. The results show that by contributing to the representation of generalist species, CCs are aiding in the coexistence of sensitive and tolerant species across the region, thereby maximizing total species richness.
Of the criteria assessed, prioritization based on corridor width was the most efficient at selecting complementary CC sites, especially for generalist species. Research suggests that generalist species can benefit from habitat destruction, but they will perform better in landscapes of intermediate quality than landscapes of poor quality (Ramiadantsoa et al. 2018). Compared to narrow corridors, wide corridors experience less edge effects (van Schalkwyk et al. 2020), are better at maintaining connectivity (Hilty et al. 2006), contain better quality habitats (Pryke and Samways 2001), and experience less shading from alien trees, which is especially important for dragonflies in these CCs (Remsburg et al. 2008; Samways and Sharratt 2010). The higher conservation value of these wide corridors aligns with previous assessments based on site-level estimates of insect diversity (Pryke and Samways 2012; van Schalkwyk et al. 2017; Schalkwyk et al. 2020, 2021). This is the first work, however, to show the complementary value of wide corridors, both in relation to diversity represented in CCs and in relation to diversity represented in existing PAs.
In general, prioritization criteria selected complementary sites that were comparable to random site selection, indicating unbiased species representation. Conservation site selection in the real-world is often biased and influenced by socio-political, economical, or aesthetic values (Kukkala and Moilanen 2013). Random site selection can therefore be expected to provide a good representation of a region’s biodiversity and we did not consider it a worst-case scenario (Ferrier 2002).
Prioritization based on the HSS was less effective than random site selection when representing species across CCs, and prioritization based on species richness was less effective when representing species across both CCs and PAs. We only focused on the sensitivity sub-index of the Dragonfly Biotic Index as our focus was on habitat quality, but our results reflect other studies that also incorporated information on species distribution and threat status when calculating site scores, which found high levels of redundancy (the same species were repeatedly represented) (Simaika and Samways 2009). Species richness is often used for prioritizing conservation actions (Simaika et al. 2013) but it is also known to underperform at improving representativeness (Veach et al. 2017). Alternative ways of calculating species richness that shift the focus to rare or specialist species have been proposed (Astudillo-Scalia and de Albuquerque 2019; Jalkanen et al. 2020b). However, as we have shown, the value of CCs for increasing regional species richness lies in contributing to the representation of generalist species.
Variation in species detectability can distort results. We tried to control for this by removing the very rarest of species (< 10 observed individuals). The fact remains that rare species make up a large number of species in a community and could be rare for reasons other than low detectability. Future assessments should consider the influence of these rare species, while controlling for detection bias. Although our assessment suggests relatively good sampling completeness, we must bear in mind that this is only a temporal snapshot of the assemblages present across the whole region. Some sites were excluded from the analyses, as there were no dragonflies sampled at these sites. This implies that these sites have no value, and this is likely not true should more sampling be done over a longer time (Kietzka et al. 2021b) or if we also incorporated larval stages into our sampling scheme (Khelifa 2019; Kietzka et al. 2021c). We also did not consider the influence of inter-site geographical distance on criteria performance, which could be important considering the positive association between species turnover and geographical distance (i.e., distance decay) (Nekola and White 1999). Furthermore, species-level representation can be only one among many different prioritization goals (Lehtomäki and Moilanen 2013). Successful conservation plans need to go beyond only representing biodiversity to also ensuring biodiversity persistence by considering important processes, such as dispersal, species interactions, migration, and distributional range shifts in response to climate change (Sarkar et al. 2006; Economo 2011; Chase et al. 2020).
Conclusion and management implementations
Protected areas are often the focus of conservation assessments (Cantú-Salazar et al. 2013), but areas outside of PAs can contribute significantly to conservation. The potential of CCs for adding unrepresented features are increasingly being recognised in conservation plans, e.g., Schmid and Alonso (2005) and Jalkanen et al. (2020). Our results show that networks of CCs enhance regional representativeness by complementing dragonfly diversity represented in existing PAs. Wide conservation corridors with little alien vegetation cover provide the most complementarity for maximizing regional representativeness.
Data Availability
These data will be made available with the publication of this manuscript and will be accessible via the Figshare repository.
References
Astudillo-Scalia Y, de Albuquerque FS (2019) Evaluating the performance of rarity as a surrogate in site prioritization for biodiversity conservation. Glob Ecol Conserv 18:e00639. https://doi.org/10.1016/j.gecco.2019.e00639
Ball-Damerow JE, M’Gonigle LKM, Vincent HR et al (2014) Local and regional factors influencing assemblages of dragonflies and damselflies (Odonata) in California and Nevada. J Insect Conserv 18:1027–1036. https://doi.org/10.1007/s10841-014-9709-6
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19:134–143. https://doi.org/10.1111/j.1466-8238.2009.00490.x
Baselga A, Orme CDL (2012) Betapart: an R package for the study of beta diversity. Methods Ecol Evol 3:808–812. https://doi.org/10.1111/j.2041-210X.2012.00224.x
Baselga A, Orme D, Sebastien V et al (2021) betapart: Partitioning Beta Diversity into Turnover and Nestedness Components. R package version 1.5.4
Beier P (2018) A rule of thumb for widths of conservation corridors. Conserv Biol. https://doi.org/10.1111/cobi.13256
Büchi L, Vuilleumier S (2014) Coexistence of specialist and generalist species is shaped by dispersal and environmental factors. Am Nat 183. https://doi.org/10.1086/675756
Cantú-Salazar L, Orme CDL, Rasmussen PC et al (2013) The performance of the global protected area system in capturing vertebrate geographic ranges. Biodivers Conserv 22:1033–1047. https://doi.org/10.1007/s10531-013-0467-7
Cardoso P, Pekár S, Jocqué R, Coddington JA (2011) Global patterns of guild composition and functional diversity of spiders. PLoS ONE 6. https://doi.org/10.1371/journal.pone.0021710
Cardoso P, Barton PS, Birkhofer K et al (2020) Scientists’ warning to humanity on insect extinctions. Biol Conserv 108426. https://doi.org/10.1016/j.biocon.2020.108426
Cardoso P, Mammola S, Rigal F, Carvalho J (2021) BAT: Biodiversity Assessment Tools. R package version 2.7.1
Chase JM, Jeliazkov A, Ladouceur E, Viana DS (2020) Biodiversity conservation through the lens of metacommunity ecology. Ann N Y Acad Sci 1469:86–104. https://doi.org/10.1111/nyas.14378
Clausnitzer V (2003) Dragonfly communities in coastal habitats of Kenya: indication of biotope quality and the need of conservation measures. Biodivers Conserv 12:333–356. https://doi.org/10.1023/A:1021920402913
Colles A, Liow LH, Prinzing A (2009) Are specialists at risk under environmental change? Neoecological, paleoecological and phylogenetic approaches. Ecol Lett 12:849–863. https://doi.org/10.1111/j.1461-0248.2009.01336.x
Dahal BR, McAlpine CA, Maron M (2014) Bird conservation values of off-reseve forests in lowland Nepal. For Ecol Manage 323:28–38. https://doi.org/10.1016/j.foreco.2014.03.033
Deacon C, Samways MJ, Pryke JS (2019) Aquatic insects decline in abundance and occupy low-quality artificial habitats to survive hydrological droughts. Freshw Biol 64:1643–1654. https://doi.org/10.1111/fwb.13360
Delso Á, Fajardo J, Muñoz J (2021) Protected area networks do not represent unseen biodiversity. Sci Rep 11:12275. https://doi.org/10.1038/s41598-021-91651-z
Devictor V, Julliard R, Jiguet F (2008) Distribution of specialist and generalist species along spatial gradients of habitat disturbance and fragmentation. Oikos 117:507–514. https://doi.org/10.1111/j.0030-1299.2008.16215.x
Economo EP (2011) Biodiversity conservation in metacommunity networks: linking pattern and persistence. Am Nat 177:E167–E180. https://doi.org/10.1086/659946
Faith DP (1995) Biodiversity and regional sustainability analysis. CSIRO, Canberra
Faith DP, Walker PA (1996a) Environmental diversity: on the best-possible use of surrogate data for assessing the relative biodiversity of sets of areas. Biodivers Conserv 5:399–415. https://doi.org/10.1007/BF00056387
Faith DP, Walker PA (1996b) DIVERSITY: a Software Package for Sampling phylogenetic and environmental diversity. Reference and user’s guide. V. 2.1. CSIRO Division of Wildlife and Ecology, Canberra
Ferrier S (2002) Mapping spatial pattern in biodiversity for regional conservation planning: where to from here? Syst Biol 51:331–363
Forman RTT (1995) Land mosaics: the Ecology of Landscapes and Regions. Harvard University, Massachusetts
French SK, McCauley SJ (2019) The movement responses of three libellulid dragonfly species to open and closed landscape cover. Insect Conserv Divers 12:437–447. https://doi.org/10.1111/icad.12355
Gilbert-Norton L, Wilson R, Stevens JR, Beard KH (2010) A meta-analytic review of corridor effectiveness. Conserv Biol 24:660–688. https://doi.org/10.1111/j.1523-1739.2010.01450.x
Hansen S, Roets F, Seymour CL et al (2018) Alien plants have greater impact than habitat fragmentation on native insect flower visitation networks. Divers Distrib 24:58–68
Heink U (2009) Representativeness - an appropriate criterion for evaluation in nature conservation. Gaia-Ecological Perspect Sci Soc 18:322–330
Hess GR, Fischer RA (2001) Communicating clearly about conservation corridors. Landsc Urban Plan 55:195–208. https://doi.org/10.1016/S0169-2046(01)00155-4
Hilty JA, Lidicker WZJ, Merenlender AM (2006) Corridor Ecology: the Science and Practice of linking landscapes for Biodiversity Conservation. Island Press, Washington
Jalkanen J, Toivonen T, Moilanen A (2020a) Identification of ecological networks for land-use planning with spatial conservation prioritization. Landsc Ecol 35:353–371. https://doi.org/10.1007/s10980-019-00950-4
Jalkanen J, Vierikko K, Moilanen A (2020b) Spatial prioritization for urban Biodiversity Quality using biotope maps and expert opinion. Urban For Urban Green 49:126586. https://doi.org/10.1016/j.ufug.2020.126586
Jongman RHG, Pungetti G (2004) Ecological networks and greenways. Concept, Design, implementation. Cambridge University Press, Cambridge
Kane A, Monadjem A, Aschenborn HKO et al (2022) Understanding continent-wide variation in vulture ranging behavior to assess feasibility of vulture safe zones in Africa: challenges and possibilities. Biol Conserv 109516. https://doi.org/10.1016/j.biocon.2022.109516
Khelifa R (2019) Sensitivity of biodiversity indices to life history stage, habitat type and landscape in Odonata community. Biol Conserv 237:63–69. https://doi.org/10.1016/j.biocon.2019.06.010
Kietzka GJ, Pryke JS, Samways MJ (2015) Landscape ecological networks are successful in supporting a diverse dragonfly assemblage. Insect Conserv Divers 8:229–237. https://doi.org/10.1111/icad.12099
Kietzka GJ, Pryke JS, Samways MJ (2017) Aerial adult dragonflies are highly sensitive to in-water conditions across an ancient landscape. Divers Distrib 23:14–26. https://doi.org/10.1111/ddi.12493
Kietzka GJ, Pryke JS, Gaigher R, Samways MJ (2021a) Webs of well-designed conservation corridors maintain river ecosystem integrity and biodiversity in plantation mosaics. Biol Conserv 254:108965. https://doi.org/10.1016/j.biocon.2021.108965
Kietzka GJ, Pryke JS, Gaigher R, Samways MJ (2021b) 32 years of essential management to retain value of an urban dragonfly awareness pond. Urban Ecosyst 1–10. https://doi.org/10.1007/s11252-021-01115-5
Kietzka GJ, Pryke JS, Gaigher R, Samways MJ (2021c) Congruency between adult male dragonflies and their larvae in river systems is relative to spatial grain. Ecol Indic 124:107390. https://doi.org/10.1016/j.ecolind.2021.107390
Korkeamäki E, Suhonen J (2002) Distribution and habitat specialization of species affect local extinction in dragonfly Odonata populations. Ecography (Cop) 25:459–465. https://doi.org/10.1034/j.1600-0587.2002.250408.x
Kukkala AS, Moilanen A (2013) Core concepts of spatial prioritisation in systematic conservation planning. Biol Rev 88:443–464. https://doi.org/10.1111/brv.12008
Lees AC, Peres CA (2008) Conservation value of remnant riparian forest corridors of varying quality for amazonian birds and mammals. Conserv Biol 22:439–449. https://doi.org/10.1111/j.1523-1739.2007.00870.x
Lehtomäki J, Moilanen A (2013) Methods and workflow for spatial conservation prioritization using Zonation. Environ Model Softw 47:128–137. https://doi.org/10.1016/j.envsoft.2013.05.001
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton
Margules CR, Sarkar S (2007) Systematic Conservation Planning, First edit. Cambridge University Press, Cambridge
McGeoch MA (2002) Insect conservation in South Africa: an overview. Afr Entomol 10:1–10
Morelli F, Benedetti Y, Callaghan CT (2020) Ecological specialization and population trends in european breeding birds. Glob Ecol Conserv 22:e00996. https://doi.org/10.1016/j.gecco.2020.e00996
Mossman HL, Panter CJ, Dolman PM (2015) Modelling biodiversity distribution in agricultural landscapes to support ecological network planning. Landsc Urban Plan 141:59–67. https://doi.org/10.1016/j.landurbplan.2015.04.010
Nekola JC, White PS (1999) The distance decay of similarity in biogeography and ecology. J Biogeogr 26:867–878. https://doi.org/10.1046/j.1365-2699.1999.00305.x
Noss RF (1987) Corridors in real landscapes: a reply to Simberloff and Cox. Conserv Biol 1:159–164. https://doi.org/10.1111/j.1523-1739.1987.tb00024.x
Oksanen J, Blanchet FG, Friendly M et al (2019) vegan: Community Ecology Package. R package version 2.5-6
Pryke SR, Samways MJ (2001) Width of grassland linkages for the conservation of butterflies in south african afforested areas. Biol Conserv 101:85–96. https://doi.org/10.1016/S0006-3207(01)00042-8
Pryke SR, Samways MJ (2003) Quality of remnant indigenous grassland linkages for adult butterflies (Lepidoptera) in an afforested african landscape. Biodivers Conserv 12:1985–2004. https://doi.org/10.1023/A:1024103527611
Pryke JS, Samways MJ (2012) Ecological networks act as extensions of protected areas for arthropod biodiversity conservation. J Appl Ecol 49:591–600. https://doi.org/10.1111/j.1365-2664.2012.02142.x
Pryke JS, Samways MJ, De Saedeleer K (2015) An ecological network is as good as a major protected area for conserving dragonflies. Biol Conserv 191:537–545. https://doi.org/10.1016/j.biocon.2015.07.036
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ramiadantsoa T, Hanski I, Ovaskainen O (2018) Responses of generalist and specialist species to fragmented landscapes. Theor Popul Biol 124:31–40. https://doi.org/10.1016/j.tpb.2018.08.001
Remsburg AJ, Olson AC, Samways MJ (2008) Shade alone reduces adult Dragonfly (Odonata: Libellulidae) abundance. J Insect Behav 21:460–468. https://doi.org/10.1007/s10905-008-9138-z
Samways MJ (2007) Implementing ecological networks for conserving insect and other biodiversity. In: Stewart AJA, New TR, Lewis OT (eds) Insect Conservation Biology. Springer Netherlands, pp 127–143
Samways MJ, Sharratt NJ (2010) Recovery of endemic dragonflies after removal of invasive alien trees. Conserv Biol 24:267–277. https://doi.org/10.1111/j.1523-1739.2009.01427.x
Samways MJ, Simaika JP (2016) Manual for Freshwater Assessment for South Africa: Dragonfly Biotic Index. South African National Biodiversity Institute, Pretoria
Samways MJ, Bazelet CS, Pryke JS (2010) Provision of ecosystem services by large scale corridors and ecological networks. Biodivers Conserv 19:2949–2962. https://doi.org/10.1007/s10531-009-9715-2
Sarkar S, Pressey RL, Faith DP et al (2006) Biodiversity conservation planning tools: present status and challenges for the future. Annu Rev Environ Resour 31:123–159. https://doi.org/10.1146/annurev.energy.31.042606.085844
Schmid J, Alonso LE (eds) (2005) A rapid biological assessment of the Mantadia-Zahamena Corridor, Madagascar. RAP Bulletin of Biological Assessment No. 32. Conservation International, Washington
Simaika JP, Samways MJ (2009) Reserve selection using red listed taxa in three global biodiversity hotspots: dragonflies in South Africa. Biol Conserv 142:638–651. https://doi.org/10.1016/j.biocon.2008.11.012
Simaika JP, Samways MJ, Kipping J et al (2013) Continental-scale conservation prioritization of african dragonflies. Biol Conserv 157:245–254. https://doi.org/10.1016/j.biocon.2012.08.039
van Schalkwyk J, Pryke JS, Samways MJ (2017) Wide corridors with much environmental heterogeneity best conserve high dung beetle and ant diversity. Biodivers Conserv 26:1243–1256. https://doi.org/10.1007/s10531-017-1299-7
van Schalkwyk J, Pryke JS, Samways MJ, Gaigher R (2019) Complementary and protection value of a Biosphere Reserve buffer zone for increasing local representativeness of ground-living arthropods. Biol Conserv 239:108292. https://doi.org/10.1016/j.biocon.2019.108292
van Schalkwyk J, Pryke JS, Samways MJ, Gaigher R (2020) Corridor width determines strength of edge influence on arthropods in conservation corridors. Landsc Ecol 6:1175–1185. https://doi.org/10.1007/s10980-020-01008-6
van Schalkwyk J, Gaigher R, Pryke JS, Samways MJ (2021) Within-corridor heterogeneity is more important than corridor design for maintaining butterfly functional and taxonomic diversity. J Appl Ecol 1–13. https://doi.org/10.1111/1365-2664.14006
van Schalkwyk J, Pryke JS, Samways MJ, Gaigher R (2022) Corridor width and orientation are complementary design variables for butterflies in conservation corridors. Landsc Ecol 37:2535–2549
Veach V, Minin ED, Pouzols FM, Moilanen A (2017) Species richness as criterion for global conservation area placement leads to large losses in coverage of biodiversity. Divers Distrib Distrib 23:715–726. https://doi.org/10.1111/ddi.12571
Vorster C, Samways MJ, Simaika JP et al (2020) Development of a new continental-scale index for freshwater assessment based on dragonfly assemblages. Ecol Indic 109:105819. https://doi.org/10.1016/j.ecolind.2019.105819
Wilson KA, Cabeza M, Klein CJ (2009) Fundamental concepts of spatial conservation prioritization. In: Moilanen A, Wilson KA, Possingham H (eds) Spatial conservation prioritization: quantitative methods and computational tools. Oxford University Press, Oxford, pp 16–27
Acknowledgements
We thank Mondi Group for financial support and providing accommodation, as well as access to their land. We thank Ezemvelo KZN Wildlife for access to field sites. We are also grateful to A. Hallett, L. Kets, M. Eckert, and S. Beirne for field assistance.
Funding
This work received financial support from Mondi Group.
Open access funding provided by Stellenbosch University.
Author information
Authors and Affiliations
Contributions
G.J.K., R.G., J.S.P, and M.J.S. conceived and designed the study. G.J.K. collected the data. J.v.S conducted the analysis and is the primary author of the manuscript. All authors contributed critically to draft manuscripts and approved the final manuscript for publication.
Corresponding author
Ethics declarations
Ethical approval
All appropriate ethics and other approvals were obtained for the research.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by David Hawksworth.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
van Schalkwyk, J., Kietzka, G.J., Pryke, J.S. et al. Enhancing semi-aquatic species representativeness beyond protected areas: dragonflies in networks of conservation corridors. Biodivers Conserv 32, 3991–4005 (2023). https://doi.org/10.1007/s10531-023-02678-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02678-9