Abstract
Objectives
Diversity in backbone polymer composition makes hydrogel-based resources open to broad spectrum of applications. Biomacromolecules which have reactive functional groups in their structural frame and can also exhibit hydrogel properties could be utilized in biomedical, pharmaceutical and drug delivery applications after some chemical modifications.
Results
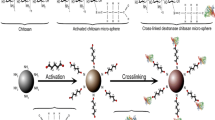
Present study aims towards development of chitosan-based hydrogel system crosslinked together with glucosyltransferase. Hydrogel structure worked as an immobilization matrix and as a microreactor system to catalyze the cleavage of a disaccharide. Uniform chitosan hydrogel beads were prepared and dextransucrase was attached using multipoint covalent interaction approach. Strong interaction was developed by linking polymeric hydrogel with the biocatalyst utilizing glutaraldehyde as spacer arms. This bifunctional crosslinking agent performed two important tasks that includes functionalization of hydrogel beads and crosslinking of this activated matrix system with enzyme fragments. Hydrogel beads required 18.0 h crosslinking time with enzyme (6.5 mg ml−1, 189.9 DSU) under specific environment (4 °C, 100 rpm) to saturate all available ends. Enzyme fragments were observed bound with hydrogel beads when screened for surface topology indicating successful crosslinking. Steady state kinetics of crosslinked dextransucrase was studied in detail and it was revealed that it can catalyse sucrose in 30.0 min at 35 °C (pH 5.5) with an energy of activation around 15.23 kJ mol−1 with increased Vmax (785 DSU ml−1) and Km (256 mM) values as compared to soluble enzyme version. Thermal stability of the crosslinked dextransucrase also particularly improved 2.5 fold at 45 °C in comparison with soluble enzyme. Improved catalytic performance suggests that multipoint covalent immobilization protocol adapted using hydrogel system could be tailored as microreactor for catalysis of profitable macromolecules.






Similar content being viewed by others
Code availability
Not applicable.
References
Aman A, Siddiqui NN, Qader SAU (2012) Characterization and potential applications of high molecular weight dextran produced by Leuconostoc mesenteroides AA1. Carbohydr Polym 87:910–915. https://doi.org/10.1016/j.carbpol.2011.08.094
Aranaz I, Mengíbar M, Harris R, Paños I, Miralles B, Acosta N, Galed G, Heras A (2009) Functional characterization of chitin and chitosan. Curr Chem Biol 3:203–230. https://doi.org/10.2174/187231309788166415
Bashar MM, Khan MA (2013) An overview on surface modification of cotton fiber for apparel use. J Polym Environ 21:181–190. https://doi.org/10.1007/s10924-012-0476-8
Berensmeier S, Ergezinger M, Bohnet M, Buchholz K (2004) Design of immobilised dextransucrase for fluidised bed application. J Biotechnol 114:255–267. https://doi.org/10.1016/j.jbiotec.2004.04.009
Bilal M, Asgher M, Cheng H, Yan Y, Iqbal HMN (2019) Multi-point enzyme immobilization, surface chemistry, and novel platforms: a paradigm shift in biocatalyst design. Crit Rev Biotechnol 39:202–219. https://doi.org/10.1080/07388551.2018.1531822
Bisswanger H (2014) Enzyme assays. Perspect Sci 1:41–55. https://doi.org/10.1016/j.pisc.2014.02.005
Casadey R, Broglia M, Barbero C, Criado S, Rivarola C (2020) Controlled release systems of natural phenolic antioxidants encapsulated inside biocompatible hydrogels. React Funct Polym 156:104729. https://doi.org/10.1016/j.reactfunctpolym.2020.104729
Côté GL, Skory CD (2016) Effect of a single point mutation on the interaction of glucans with a glucansucrase from Leuconostoc mesenteroides NRRL B-1118. Carbohydr Res 428:57–61. https://doi.org/10.1016/j.carres.2016.04.010
da Silva RM, Gonçalves LRB, Rodrigues S (2020) Different strategies to co-immobilize dextransucrase and dextranase on to agarose based supports: operational stability study. Int J Biol Macromol 156:411–419. https://doi.org/10.1016/j.ijbiomac.2020.04.077
de Segura AG, Alcalde M, Yates M, Cervantes MLR, Cortés NL, Ballesteros A, Plou FJ (2004) Immobilization of dextransucrase from Leuconostoc mesenteroides NRRL B-512F on Eupergit C supports. Biotechnol Prog 20:1414–1420. https://doi.org/10.1021/bp0400083
Dols M, Remaud-Simeon M, Willemot RM, Vignon M, Monsan PF (1997) Characterization of dextransucrases from Leuconostoc mesenteroides NRRL B-1299. Appl Biochem Biotechnol 62:47–59. https://doi.org/10.1007/BF02787983
Dragan ES, Dinu MV (2020) Advances in porous chitosan-based composite hydrogels: synthesis and applications. React Funct Polym 146:104372. https://doi.org/10.1016/j.reactfunctpolym.2019.104372
Dwamena AK, Woo SH, Kim CS (2020) Enzyme immobilization on porous chitosan hydrogel capsules formed by anionic surfactant gelation. Biotechnol Lett 42:845–852. https://doi.org/10.1007/s10529-020-02829-w
Graebin NG, de Andrades D, Bonin MC, Rodrigues RC, Ayub MA (2016) Dextransucrase immobilized on activated-chitosan particles as a novel biocatalyst. J Mol Catal B 133:S143–S149. https://doi.org/10.1016/j.molcatb.2016.12.007
Hashem AM, Gamal AA, Hassan ME, Hassanein NM, Esawy MA (2016) Covalent immobilization of Enterococcus faecalis Esawy dextransucrase and dextran synthesis. Int J Biol Macromol 82:905–912. https://doi.org/10.1016/j.ijbiomac.2015.09.076
Juanga RS, Wub FC, Tseng RL (2001) Solute adsorption and enzyme immobilization on chitosan beads prepared from shrimp shell wastes. Bioresour Technol 80:187–193. https://doi.org/10.1016/S0960-8524(01)00090-6
Karpova N, Shagdarova B, Lunkov A, Il’ina A, Varlamov V (2021) Antifungal action of chitosan in combination with fungicides in vitro and chitosan conjugate with gallic acid on tomatoes against Botrytis cinerea. Biotechnol Lett 43:1565–1574. https://doi.org/10.1007/s10529-021-03138-6
Kim D, Day DF (1994) A new process for the production of clinical dextran by mixed culture fermentation of Lipomyces starkeyi and Leuconostoc mesenteroides. Enzyme Microb Technol 16:844–848. https://doi.org/10.1016/0141-0229(94)90058-2
Kothari D, Baruah R, Goyal A (2012) Immobilization of glucansucrase for the production of gluco-oligosaccharides from Leuconostoc mesenteroides. Biotechnol Lett 34:2101–2106. https://doi.org/10.1007/s10529-012-1014-4
Krajewska B (2004) Application of chitin-and chitosan-based materials for enzyme immobilizations: a review. Enzyme Microb Technol 35:126–139. https://doi.org/10.1016/j.enzmictec.2003.12.013
Leathere TD, Hayman GT, Cote GL (1995) Rapid screening of Leuconostoc mesenteroides mutants for elevated proportions of alternan to dextran. Curr Microbiol 31:19–22. https://doi.org/10.1007/BF00294628
Lopez A, Monsan P (1980) Dextran synthesis by immobilized dextransucrase. Biochimie 62:323–329. https://doi.org/10.1016/S0300-9084(80)80161-1
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein Measurement with the Folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/S0021-9258(19)52451-6
Monteiro OAC Jr, Airoldi C (1999) Some studies of crosslinking chitosan-glutaraldehyde interaction in a homogeneous system. Int J Biol Macromol 26:119–128. https://doi.org/10.1016/-8130(S014199)00068-9
Ngah WW, Fatinathan S (2008) Adsorption of Cu (II) ions in aqueous solution using chitosan beads, chitosan–GLA beads and chitosan–alginate beads. Chem Eng J 143:62–72. https://doi.org/10.1016/j.cej.2007.12.006
Nguyen TTH, Seo YS, Cho JY, Lee S, Kim GJ, Yoon JW, Ahn SH, Hwang KH, Park JS, Jang TS, Kim D (2015) Synthesis of oligosaccharide-containing orange juice using glucansucrase. Biotechnol Bioprocess E 20:447–452. https://doi.org/10.1007/s12257-014-0741-x
Parlak M, Ustek D, Tanriseven A (2013) A novel method for covalent immobilization of dextransucrase. J Mol Catal B 89:52–60. https://doi.org/10.1016/j.molcatb.2012.12.013
Qu B, Luo Y (2020) Chitosan-based hydrogel beads: preparations, modifications and applications in food and agriculture sectors–a review. Int J Biol Macromol 152:437–448. https://doi.org/10.1016/j.ijbiomac.2020.02.240
Reischwitz A, Reh KD, Buchholz K (1995) Unconventional immobilization of dextransucrase with alginate. Enzyme Microb Technol 17:457–461. https://doi.org/10.1016/0141-0229(94)00091-5
Rinaudo M (2006) Chitin and chitosan: properties and applications. Prog Polym Sci 31:603–632. https://doi.org/10.1016/j.progpolymsci.2006.06.001
Shamala TR, Prasad MS (1995) Preliminary studies on the production of high and low viscosity dextran by Leuconostoc spp. Process Biochem 30:237–241. https://doi.org/10.1016/0032-9592(95)85004-X
Sutherland IW, Reh HJ, Reed G, Puhler A, Stadler P (1996) Biotechnology, 2nd edn. VCH, New York
Vakili M, Rafatullah M, Salamatinia B, Abdullah AZ, Ibrahim MH, Tan KB, Gholami Z, Amouzgar P (2014) Application of chitosan and its derivatives as adsorbents for dye removal from water and wastewater: a review. Carbohydr Polym 113:115–130. https://doi.org/10.1016/j.carbpol.2014.07.007
Wahab RA, Elias N, Abdullah F, Ghoshal SK (2020) On the taught new tricks of enzymes immobilization: an all-inclusive overview. React Funct Polym. https://doi.org/10.1016/j.reactfunctpolym.2020.104613
Wang J, Zhuang S (2017) Removal of various pollutants from water and wastewater by modified chitosan adsorbents. Crit Rev Environ Sci Technol 47:2331–2386. https://doi.org/10.1080/10643389.2017.1421845
Wang D, Yan L, Ma X, Wang W, Zou M, Zhong J, Ding T, Ye X, Liu D (2018) Ultrasound promotes enzymatic reactions by acting on different targets: enzymes, substrates and enzymatic reaction systems. Int J Biol Macromol 119:453–461. https://doi.org/10.1016/j.ijbiomac.2018.07.133
Xie JH, Jin ML, Morris GA, Zha XQ, Chen HQ, Yi Y, Li JE, Wang ZJ, Gao J, Nie SP, Shang P, Xie MY (2016) Advances on bioactive polysaccharides from medicinal plants. Crit Rev Food Sci Nutr 56:S60–S84. https://doi.org/10.1080/10408398.2015.1069255
Yagar H, Balkan U (2017) Entrapment of laurel lipase in chitosan hydrogel beads. Artif Cells Nanomed Biotechnol 45:864–870. https://doi.org/10.1080/21691401.2016.1182920
Zhang Y, Guo L, Xu D, Li D, Yang N, Chen F, Jin Z, Xu X (2018) Effects of dextran with different molecular weights on the quality of wheat sourdough breads. Food Chem 256:373–379. https://doi.org/10.1016/j.foodchem.2018.02.146
Funding
This research work was indigenously supported by The Karachi Institute of Biotechnology and Genetic Engineering (KIBGE), University of Karachi, Pakistan.
Author information
Authors and Affiliations
Contributions
AA contributed in conceptualization, validation, formal analysis, investigation, writing- review & editing of research data, arranged resources, performed data curation and performed project administration. FS assisted in methodology, formal analysis and helped in writing the original first draft of the manuscript. SP contributed in validation of results and assisted in writing and editing of the final manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aman, A., Shahid, F. & Pervez, S. Exploration of a three-dimensional matrix as micro-reactor in the form of reactive polyaminosaccharide hydrogel beads using multipoint covalent interaction approach. Biotechnol Lett 44, 299–319 (2022). https://doi.org/10.1007/s10529-022-03223-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-022-03223-4