Abstract
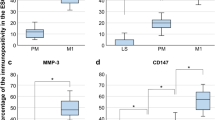
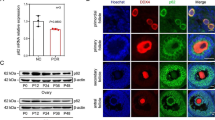
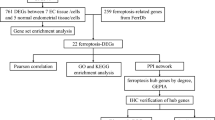
Endometriosis (EMS) is a common gynecological condition with apparent heterogeneity, lack of diagnostic markers, and unclear pathogenesis. A series of bioinformatics methods were employed to explore EMS’s pathological mechanisms and potential biomarkers by analyzing the combined datasets of EMS (GSE7305, GSE7307, GSE58198, E-MTAB-694), which included 34 normal, 127 eutopic, and 46 ectopic endometrium samples. Then, wet-laboratory experiments (including Western blot, qRT-PCR, and Immunohistochemistry, Immunofluorescence, CCK-8, EdU, Wound healing, Transwell, and Adhesion assays) were applied to examine the biomarkers’ expression and function in primary endometrial stromal cells. Bioinformatic analysis indicated that the core pathogenesis of EMS was dysregulated immune-inflammation and tissue remolding processes. Among the upregulated DEGs, BST2 was screened as a potential diagnostic biomarker in EMS, which associated with the revised American Fertility Society (r-AFS) stage and immune-inflammation processes of EMS. Moreover, BST2’s overexpression was affirmed in the RNA and protein levels in EMS tissues. In vitro experiments demonstrated that TNF-α promoted the expression of BST2 in ESCs. And BST2 knockdown inhibited migration, invasion, adhesion, and inflammation except for the proliferation of ESCs, probably via the TNF-α/NF-κB pathway. Through a combination of wet and dry studies, we concluded that the core pathogenesis of endometriosis was dysregulated immune-inflammation and tissue remolding, and BST2 might be a potential diagnostic and therapeutic target in endometriosis.







Similar content being viewed by others
Data Availability
The datasets GSE7305, GSE7307, E-MTAB-694, GSE51981, GSE105764, GSE47360, and GSE25628, used in this study can be found in the NCBI GEO database (https://www.ncbi.nlm.nih.gov/geo/).
References
Bf B, Gw B, Mm L et al (2004) Efficacy of anti-tumor necrosis factor therapy in the treatment of spontaneous endometriosis in baboons. Fertil Steril. https://doi.org/10.1016/j.fertnstert.2003.09.034
Bulun SE, Wan Y, Matei D (2019a) Epithelial mutations in endometriosis: link to ovarian cancer. Endocrinology 160:626–638. https://doi.org/10.1210/en.2018-00794
Bulun SE, Yilmaz BD, Sison C et al (2019b) Endometriosis. Endocr Rev 40:1048–1079. https://doi.org/10.1210/er.2018-00242
Chen C, Shi H, Wang B et al (2018) Aberrant expression of the innate restriction factor bone marrow stromal antigen-2 in primary Sjögren’s syndrome. J Craniomaxillofac Surg 46:1899–1904. https://doi.org/10.1016/j.jcms.2018.08.008
Clayton RD, Duffy SR, Wilkinson N et al (2004) Anti-proliferative effect of mycobacteria, IFN-γ and TNF-α on primary cultures derived from endometrial stroma: possible relevance to endometriosis?: Mycobacteria; a role in endometriosis therapy? Am J Reprod Immunol 51:63–70. https://doi.org/10.1046/j.8755-8920.2003.00114.x
Crispi S, Piccolo MT, D’Avino A et al (2013) Transcriptional profiling of endometriosis tissues identifies genes related to organogenesis defects. J Cell Physiol 228:1927–1934. https://doi.org/10.1002/jcp.24358
D’Antonio M, Martelli F, Peano S et al (2000) Ability of recombinant human TNF binding protein-1 (r-hTBP-1) to inhibit the development of experimentally-induced endometriosis in rats. J Reprod Immunol. https://doi.org/10.1016/s0165-0378(00)00073-5
D’Hooghe TM, Nugent NP, Cuneo S et al (2006) Recombinant human TNFRSF1A (r-hTBP1) inhibits the development of endometriosis in baboons: a prospective, randomized, placebo- and drug-controlled study. Biol Reprod 74:131–136. https://doi.org/10.1095/biolreprod.105.043349
Dinarello CA (2011) A clinical perspective of IL-1β as the gatekeeper of inflammation. Eur J Immunol 41:1203–1217. https://doi.org/10.1002/eji.201141550
Dw B (2003) Endometriosis is sustained by tumour necrosis factor-alpha. Med Hypotheses. https://doi.org/10.1016/s0306-9877(02)00336-5
Gu G, Zhao D, Yin Z, Liu P (2012) BST-2 binding with cellular MT1-MMP blocks cell growth and migration via decreasing MMP2 activity. J Cell Biochem 113:1013–1021. https://doi.org/10.1002/jcb.23433
Hänzelmann S, Castelo R, Guinney J (2013) GSVA: gene set variation analysis for microarray and RNA-Seq data. BMC Bioinform 14:7. https://doi.org/10.1186/1471-2105-14-7
Hever A, Roth RB, Hevezi P et al (2007) Human endometriosis is associated with plasma cells and overexpression of B lymphocyte stimulator. Proc Natl Acad Sci USA 104:12451–12456. https://doi.org/10.1073/pnas.0703451104
Houshdaran S, Nezhat CR, Vo KC et al (2016) Aberrant endometrial DNA methylome and associated gene expression in women with endometriosis. Biol Reprod. https://doi.org/10.1095/biolreprod.116.140434
Iwabu Y, Fujita H, Kinomoto M et al (2009) HIV-1 Accessory protein Vpu internalizes cell-surface BST-2/tetherin through transmembrane interactions leading to lysosomes. J Biol Chem 284:35060. https://doi.org/10.1074/jbc.M109.058305
Jeffries MA, Dozmorov M, Tang Y et al (2011) Genome-wide DNA methylation patterns in CD4+ T cells from patients with systemic lupus erythematosus. Epigenetics 6:593–601. https://doi.org/10.4161/epi.6.5.15374
Jiang L, Zhang M, Wang S et al (2020a) Common and specific gene signatures among three different endometriosis subtypes. PeerJ 8:e8730. https://doi.org/10.7717/peerj.8730
Jiang L, Zhang M, Wu J et al (2020b) Exploring diagnostic m6A regulators in endometriosis. Aging (Albany NY) 12:25916–25938. https://doi.org/10.18632/aging.202163
Jin H, Zhang L, Wang S, Qian L (2021) BST2 promotes growth and induces gefitinib resistance in oral squamous cell carcinoma via regulating the EGFR pathway. Arch Med Sci 17:1772–1782. https://doi.org/10.5114/aoms.2019.86183
Jividen K, Movassagh MJ, Jazaeri A, Li H (2014) Two methods for establishing primary human endometrial stromal cells from hysterectomy specimens. J Vis Exp. https://doi.org/10.3791/51513
Kupzig S, Korolchuk V, Rollason R et al (2003) Bst-2/HM1.24 is a raft-associated apical membrane protein with an unusual topology. Traffic 4:694–709. https://doi.org/10.1034/j.1600-0854.2003.00129.x
Lee BNR, Chang H, Son YS et al (2018) IFN-γ enhances the wound healing effect of late EPCs (LEPCs) via BST2-mediated adhesion to endothelial cells. FEBS Lett 592:1705–1715. https://doi.org/10.1002/1873-3468.13078
Li S, Zhou Y, Huang Q et al (2021) Iron overload in endometriosis peritoneal fluid induces early embryo ferroptosis mediated by HMOX1. Cell Death Discov 7:1–12. https://doi.org/10.1038/s41420-021-00751-2
Liu G, Du X, Xiao L et al (2021) Activation of FGD5-AS1 promotes progression of cervical cancer through regulating BST2 to inhibit macrophage M1 polarization. J Immunol Res 2021:5857214. https://doi.org/10.1155/2021/5857214
Liu W, Cao Y, Guan Y, Zheng C (2018) BST2 promotes cell proliferation, migration and induces NF-κB activation in gastric cancer. Biotechnol Lett 40:1015–1027. https://doi.org/10.1007/s10529-018-2562-z
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550. https://doi.org/10.1186/s13059-014-0550-8
Lu D, Song H, Shi G (2013) Anti-TNF-α treatment for pelvic pain associated with endometriosis. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD008088.pub3
Mahauad-Fernandez WD, DeMali KA, Olivier AK, Okeoma CM (2014) Bone marrow stromal antigen 2 expressed in cancer cells promotes mammary tumor growth and metastasis. Breast Cancer Res 16:493. https://doi.org/10.1186/s13058-014-0493-8
Mahauad-Fernandez WD, Okeoma CM (2015) The role of BST-2/Tetherin in host protection and disease manifestation. Immun Inflamm Dis 4:4–23. https://doi.org/10.1002/iid3.92
Mansouri M, Viswanathan K, Douglas JL et al (2009) Molecular mechanism of BST2/tetherin downregulation by K5/MIR2 of Kaposi’s sarcoma-associated herpesvirus. J Virol 83:9672–9681. https://doi.org/10.1128/JVI.00597-09
Mukai S, Oue N, Oshima T et al (2017) Overexpression of transmembrane protein BST2 is associated with poor survival of patients with esophageal, gastric, or colorectal cancer. Ann Surg Oncol 24:594–602. https://doi.org/10.1245/s10434-016-5100-z
Nabha SM, dos Santos EB, Yamamoto HA et al (2008) Bone marrow stromal cells enhance prostate cancer cell invasion through type I collagen in an MMP-12 dependent manner. Int J Cancer 122:2482–2490. https://doi.org/10.1002/ijc.23431
Neil SJD, Zang T, Bieniasz PD (2008) Tetherin inhibits retrovirus release and is antagonized by HIV-1 Vpu. Nature 451:425–430. https://doi.org/10.1038/nature06553
Nisolle M, Donnez J (1997) Peritoneal endometriosis, ovarian endometriosis, and adenomyotic nodules of the rectovaginal septum are three different entities. Fertil Steril 68:585–596. https://doi.org/10.1016/S0015-0282(97)00191-X
Padariya M, Sznarkowska A, Kote S et al (2021) Functional interfaces, biological pathways, and regulations of interferon-related DNA damage resistance signature (IRDS) genes. Biomolecules 11:622. https://doi.org/10.3390/biom11050622
Palladino MA, Bahjat FR, Theodorakis EA, Moldawer LL (2003) Anti-TNF-α therapies: the next generation. Nat Rev Drug Discov 2:736–746. https://doi.org/10.1038/nrd1175
Ritchie ME, Phipson B, Wu D et al (2015) limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43:e47–e47. https://doi.org/10.1093/nar/gkv007
Sachs MC (2017) plotROC: A tool for plotting ROC curves. J Stat Softw 79:2. https://doi.org/10.18637/jss.v079.c02
Sampson JA (1927) Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am J Obstet Gynecol 14:422–469. https://doi.org/10.1016/S0002-9378(15)30003-X
Saunders PTK, Horne AW (2021) Endometriosis: etiology, pathobiology, and therapeutic prospects. Cell 184:2807–2824. https://doi.org/10.1016/j.cell.2021.04.041
Sauter D, Hotter D, Engelhart S et al (2013) A rare missense variant abrogates the signaling activity of tetherin/BST-2 without affecting its effect on virus release. Retrovirology 10:85. https://doi.org/10.1186/1742-4690-10-85
Sayeed A, Luciani-Torres G, Meng Z et al (2013) Aberrant regulation of the BST2 (Tetherin) promoter enhances cell proliferation and apoptosis evasion in high grade breast cancer cells. PLoS ONE 8:e67191. https://doi.org/10.1371/journal.pone.0067191
Schubert HL, Zhai Q, Sandrin V et al (2010) Structural and functional studies on the extracellular domain of BST2/tetherin in reduced and oxidized conformations. Proc Natl Acad Sci U S A 107:17951–17956. https://doi.org/10.1073/pnas.1008206107
Shi L, Xue X, Tian H et al (2021) WEE1 promotes endometriosis via the Wnt/β-catenin signaling pathway. Reprod Biol Endocrinol 19:161. https://doi.org/10.1186/s12958-021-00844-8
Sohler F, Sommer A, Wachter DL et al (2013) Tissue remodeling and nonendometrium-like menstrual cycling are hallmarks of peritoneal endometriosis lesions. Reprod Sci 20:85–102. https://doi.org/10.1177/1933719112451147
Stilley JAW, Birt JA, Nagel SC et al (2010) Neutralizing TIMP1 restores fecundity in a rat model of endometriosis and treating control rats with TIMP1 causes anomalies in ovarian function and embryo development. Biol Reprod 83:185–194. https://doi.org/10.1095/biolreprod.109.083287
Subramanian A, Tamayo P, Mootha VK et al (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci 102:15545–15550. https://doi.org/10.1073/pnas.0506580102
Tanaka T, Narazaki M, Kishimoto T (2014) IL-6 in inflammation, immunity, and disease. Cold Spring Harb Perspect Biol. https://doi.org/10.1101/cshperspect.a016295
Tamaresis JS, Irwin JC, Goldfien GA et al (2014) Molecular classification of endometriosis and disease stage using high-dimensional genomic data. Endocrinology 155:4986–4999. https://doi.org/10.1210/en.2014-1490
Tokarev A, Suarez M, Kwan W et al (2013) Stimulation of NF-κB Activity by the HIV restriction factor BST2. J Virol 87:2046–2057. https://doi.org/10.1128/JVI.02272-12
Tosti C, Pinzauti S, Santulli P et al (2015) Pathogenetic mechanisms of deep infiltrating endometriosis. Reprod Sci 22:1053–1059. https://doi.org/10.1177/1933719115592713
Vallvé-Juanico J, Houshdaran S, Giudice LC (2019) The endometrial immune environment of women with endometriosis. Hum Reprod Update 25:564–591. https://doi.org/10.1093/humupd/dmz018
Vestergaard AL, Knudsen UB, Munk T et al (2011) Transcriptional expression of type-I interferon response genes and stability of housekeeping genes in the human endometrium and endometriosis. MHR: Basic Sci Reprod Med 17:243–254. https://doi.org/10.1093/molehr/gaq100
Wang S, Yi M, Zhang X et al (2021) LncRNA CDKN2B-AS1 promotes epithelial-mesenchymal transition by targeting the miR-424–5p/AKT3 axis in ovarian endometriosis. Reprod BioMed Online. https://doi.org/10.1016/j.rbmo.2021.02.004
Wang X-M, Ma Z-Y, Song N (2018) Inflammatory cytokines IL-6, IL-10, IL-13, TNF-α and peritoneal fluid flora were associated with infertility in patients with endometriosis. Eur Rev Med Pharmacol Sci 22:2513–2518. https://doi.org/10.26355/eurrev_201805_14899
Xavier P, Belo L, Beires J et al (2006) Serum levels of VEGF and TNF-alpha and their association with C-reactive protein in patients with endometriosis. Arch Gynecol Obstet 273:227–231. https://doi.org/10.1007/s00404-005-0080-4
Yamagata Y, Nishino K, Takaki E et al (2014) Genome-wide DNA methylation profiling in cultured eutopic and ectopic endometrial stromal cells. PLoS ONE 9:e83612. https://doi.org/10.1371/journal.pone.0083612
Yang D, Yang Z, Chen L et al (2020) Dihydromyricetin increases endothelial nitric oxide production and inhibits atherosclerosis through microRNA-21 in apolipoprotein E-deficient mice. J Cell Mol Med 24:5911–5925. https://doi.org/10.1111/jcmm.15278
Yang L-L, Wu L, Yu G-T et al (2018) CD317 Signature in head and neck cancer indicates poor prognosis. J Dent Res 97:787–794. https://doi.org/10.1177/0022034518758604
Yj C, Hh W, Wt L et al (2013) A tumor necrosis factor-α inhibitor reduces the embryotoxic effects of endometriotic peritoneal fluid. Fertil Steril. https://doi.org/10.1016/j.fertnstert.2013.07.1985
Yokoyama T, Enomoto T, Serada S et al (2013) Plasma membrane proteomics identifies bone marrow stromal antigen 2 as a potential therapeutic target in endometrial cancer. Int J Cancer 132:472–484. https://doi.org/10.1002/ijc.27679
Yoo H, Park S-H, Ye S-K, Kim M (2011) IFN-γ-induced BST2 mediates monocyte adhesion to human endothelial cells. Cell Immunol 267:23–29. https://doi.org/10.1016/j.cellimm.2010.10.011
Yu G, Wang L-G, Han Y, He Q-Y (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16:284–287. https://doi.org/10.1089/omi.2011.0118
Zhao L, Gu C, Ye M et al (2018) Integration analysis of microRNA and mRNA paired expression profiling identifies deregulated microRNA-transcription factor-gene regulatory networks in ovarian endometriosis. Reprod Biol Endocrinol 16:4. https://doi.org/10.1186/s12958-017-0319-5
Zondervan KT, Becker CM, Missmer SA (2020) Endometriosis. N Engl J Med 382:1244–1256. https://doi.org/10.1056/NEJMra1810764
Acknowledgements
The authors would like to thank the GEO training course of Helix-Life as well as Biotrainee for the training of bioinformatics analysis, and Dr. Jianming Zeng (CEO of Biotrainee) and Dr. Guozi (Chongqing Medical University) for generously sharing their experience and codes.
Funding
This work was supported by the National Natural Science Foundation of China (Grant number 81671437 to X.F., Grant number 81771558 to X.X., Grant number 81801425 to T.Z.), Natural Science Foundation of Hunan Province (Grant number 2023JJ40892 to L.J.), and Natural Science Foundation of Changsha City (Grant number kq2208352 to L.J.). The funders had no role in the study design, data collection, analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
L.J. and X.F. conceived and designed the study. L.J., S.W., X.W., and T.Z. acquired and analyzed data. L.J., X.X., F.Z., and J.M. helped interpret data and prepare figures and tables. L.J. drafted the manuscript. X.F. and X.X. revised the manuscript. All authors reviewed the manuscript. L.J and X.F. gave the final approval of the version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiang, L., Wang, S., Xia, X. et al. Novel Diagnostic Biomarker BST2 Identified by Integrated Transcriptomics Promotes the Development of Endometriosis via the TNF-α/NF-κB Signaling Pathway. Biochem Genet (2024). https://doi.org/10.1007/s10528-024-10666-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10528-024-10666-z