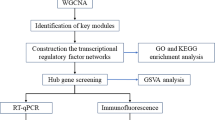
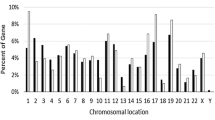
Abstract
Osteoporosis is a systemic bone disease characterized by low bone mineral density and bone microstructure damage, resulting in increased bone fragility and fracture risk. The present study aimed to identify key genes and functionally enriched pathways in osteoporotic patients. Weighted Gene Co-expression Network Analysis (WGCNA) was applied to microarray datasets of blood samples of osteoporotic patients from the Sao Paulo Ageing & Health [SPAH] study (26 osteoporotic samples and 31 normal samples) to construct co-expression networks and identify hub gene. The results showed that HDGF, AP2M1, DNAJC6, TMEM183B, MFSD2B, IGKV1-5, IGKV1-8, IGKV3-7, IGKV3D-11, and IGKV1D-42 are genes which were associated with the disease status of osteoporosis. Differentially expressed genes are enriched in proteasomal protein catabolic process, ubiquitin ligase complex, and ubiquitin-like protein transferase activity. Functional enrichment analysis demonstrated that genes in the tan module were enriched in immune-related functions, indicating that the immune system plays a critical role in osteoporosis. Validation assay demonstrated that the HDGF, AP2M1, TMEM183B, and MFSD2B levels were decreased in osteoporosis samples compared with healthy controls, while the levels of IGKV1-5, IGKV1-8, and IGKV1D-42 were increased in osteoporosis samples compared with healthy controls. In conclusion, our data identified and validated the association of HDGF, AP2M1, TMEM183B, MFSD2B, IGKV1-5, IGKV1-8, and IGKV1D-42 with osteoporosis in elderly women. These results suggest that these transcripts have potential clinical significance and may help to explain the molecular mechanisms and biological functions of osteoporosis.





Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Bridges SL Jr, Lee SK, Johnson ML, Lavelle JC, Fowler PG, Koopman WJ, Schroeder HW Jr (1995) Somatic mutation and CDR3 lengths of immunoglobulin kappa light chains expressed in patients with rheumatoid arthritis and in normal individuals. J Clin Invest 96:831–841. https://doi.org/10.1172/jci118129
Cao J, Ding H, Shang J, Ma L, Wang Q, Feng S (2021) Weighted gene co-expression network analysis reveals specific modules and hub genes related to immune infiltration of osteoarthritis. Ann Transl Med 9:1525. https://doi.org/10.21037/atm-21-4566
Cheng C, Wentworth K, Shoback DM (2020a) New frontiers in osteoporosis therapy. Annu Rev Med 71:277–288. https://doi.org/10.1146/annurev-med-052218-020620
Cheng CI, Chang HR, Tai MH, Chou MH, Chen GT, Chen PH, Kao YH (2020b) Hepatoma-derived growth factor enhances osteoblastic transformation of rat aortic vascular smooth muscle cells in vitro. Life Sci 256:117964. https://doi.org/10.1016/j.lfs.2020.117964
Compston JE, McClung MR, Leslie WD (2019) Osteoporosis. Lancet 393:364–376. https://doi.org/10.1016/s0140-6736(18)32112-3
Deng L, Huang L, Sun Y, Heath JM, Wu H, Chen Y (2015) Inhibition of FOXO1/3 promotes vascular calcification. Arterioscler Thromb Vasc Biol 35:175–183. https://doi.org/10.1161/atvbaha.114.304786
Duan JY, Lin X, Xu F, Shan SK, Guo B, Li FX, Wang Y, Zheng MH, Xu QS, Lei LM et al (2021) Ferroptosis and its potential role in metabolic diseases: a curse or revitalization? Front Cell Dev Biol 9:701788. https://doi.org/10.3389/fcell.2021.701788
Everett AD, Lobe DR, Matsumura ME, Nakamura H, McNamara CA (2000) Hepatoma-derived growth factor stimulates smooth muscle cell growth and is expressed in vascular development. J Clin Invest 105:567–575. https://doi.org/10.1172/jci7497
Everett AD, Stoops T, McNamara CA (2001) Nuclear targeting is required for hepatoma-derived growth factor-stimulated mitogenesis in vascular smooth muscle cells. J Biol Chem 276:37564–37568. https://doi.org/10.1074/jbc.M105109200
Faienza MF, Ventura A, Marzano F, Cavallo L (2013) Postmenopausal osteoporosis: the role of immune system cells. Clin Dev Immunol 2013:575936. https://doi.org/10.1155/2013/575936
Fischer V, Haffner-Luntzer M (2022) Interaction between bone and immune cells: Implications for postmenopausal osteoporosis. Semin Cell Dev Biol 123:14–21. https://doi.org/10.1016/j.semcdb.2021.05.014
Fujita H, Ochi M, Ono M, Aoyama E, Ogino T, Kondo Y, Ohuchi H (2019) Glutathione accelerates osteoclast differentiation and inflammatory bone destruction. Free Radic Res 53:226–236. https://doi.org/10.1080/10715762.2018.1563782
Gates BJ, Das S (2012) Risedronate’s role in reducing hip fracture in postmenopausal women with established osteoporosis. Clin Med Insights Arthritis Musculoskelet Disord 5:1–14. https://doi.org/10.4137/cmamd.S4092
Gong R, Xiao HM, Zhang YH, Zhao Q, Su KJ, Lin X, Mo CL, Zhang Q, Du YT, Lyu FY et al (2021) Identification and functional characterization of metabolites for bone mass in peri- and postmenopausal chinese women. J Clin Endocrinol Metab 106:e3159–e3177. https://doi.org/10.1210/clinem/dgab146
Hao ML, Zuo XQ, Qiu Y, Li J (2021) WGCNA identification of genes and pathways involved in the pathogenesis of postmenopausal osteoporosis. Int J Gen Med 14:8341–8353. https://doi.org/10.2147/ijgm.S336310
Hu TH, Huang CC, Liu LF, Lin PR, Liu SY, Chang HW, Changchien CS, Lee CM, Chuang JH, Tai MH (2003) Expression of hepatoma-derived growth factor in hepatocellular carcinoma. Cancer 98:1444–1456. https://doi.org/10.1002/cncr.11653
Kao YH, Chen CL, Jawan B, Chung YH, Sun CK, Kuo SM, Hu TH, Lin YC, Chan HH, Cheng KH et al (2010) Upregulation of hepatoma-derived growth factor is involved in murine hepatic fibrogenesis. J Hepatol 52:96–105. https://doi.org/10.1016/j.jhep.2009.10.002
Keppie SJ, Mansfield JC, Tang X, Philp CJ, Graham HK, Önnerfjord P, Wall A, McLean C, Winlove CP, Sherratt MJ et al (2021) Matrix-bound growth factors are released upon cartilage compression by an aggrecan-dependent sodium flux that is lost in osteoarthritis. Function (oxf). https://doi.org/10.1093/function/zqab037
Langfelder P, Horvath S (2008) WGCNA: an R package for weighted correlation network analysis. BMC Bioinform 9:559. https://doi.org/10.1186/1471-2105-9-559
Lee SK, Bridges SL Jr, Koopman WJ, Schroeder HW Jr (1992) The immunoglobulin kappa light chain repertoire expressed in the synovium of a patient with rheumatoid arthritis. Arthritis Rheum 35:905–913. https://doi.org/10.1002/art.1780350809
Li X, Yang Y, Sun G, Dai W, Jie X, Du Y, Huang R, Zhang J (2020) Promising targets and drugs in rheumatoid arthritis: a module-based and cumulatively scoring approach. Bone Joint Res 9:501–514. https://doi.org/10.1302/2046-3758.98.Bjr-2019-0301.R1
Li S, Chen B, Chen H, Hua Z, Shao Y, Yin H, Wang J (2021) Analysis of potential genetic biomarkers and molecular mechanism of smoking-related postmenopausal osteoporosis using weighted gene co-expression network analysis and machine learning. PLoS ONE 16:e0257343. https://doi.org/10.1371/journal.pone.0257343
Masaike Y, Takagi T, Hirota M, Yamada J, Ishihara S, Yung TM, Inoue T, Sawa C, Sagara H, Sakamoto S et al (2010) Identification of dynamin-2-mediated endocytosis as a new target of osteoporosis drugs, bisphosphonates. Mol Pharmacol 77:262–269. https://doi.org/10.1124/mol.109.059006
McHeyzer-Williams M, Okitsu S, Wang N, McHeyzer-Williams L (2011) Molecular programming of B cell memory. Nat Rev Immunol 12:24–34. https://doi.org/10.1038/nri3128
Morbach H, Richl P, Faber C, Singh SK, Girschick HJ (2008) The kappa immunoglobulin light chain repertoire of peripheral blood B cells in patients with juvenile rheumatoid arthritis. Mol Immunol 45:3840–3846. https://doi.org/10.1016/j.molimm.2008.05.011
Moxley G (1992) Variable-constant segment genotype of immunoglobulin kappa is associated with increased risk for rheumatoid arthritis. Arthritis Rheum 35:19–25. https://doi.org/10.1002/art.1780350104
Noh JY, Yang Y, Jung H (2020) Molecular mechanisms and emerging therapeutics for osteoporosis. Int J Mol Sci. https://doi.org/10.3390/ijms21207623
Okuda Y, Nakamura H, Yoshida K, Enomoto H, Uyama H, Hirotani T, Funamoto M, Ito H, Everett AD, Hada T et al (2003) Hepatoma-derived growth factor induces tumorigenesis in vivo through both direct angiogenic activity and induction of vascular endothelial growth factor. Cancer Sci 94:1034–1041. https://doi.org/10.1111/j.1349-7006.2003.tb01397.x
Panach L, Pertusa C, Martínez-Rojas B, Acebrón Á, Mifsut D, Tarín JJ, Cano A, García-Pérez M (2020) Comparative transcriptome analysis identifies CARM1 and DNMT3A as genes associated with osteoporosis. Sci Rep 10:16298. https://doi.org/10.1038/s41598-020-72870-2
Pietschmann P, Mechtcheriakova D, Meshcheryakova A, Föger-Samwald U, Ellinger I (2016) Immunology of osteoporosis: a mini-review. Gerontology 62:128–137. https://doi.org/10.1159/000431091
Qian GF, Yuan LS, Chen M, Ye D, Chen GP, Zhang Z, Li CJ, Vijayan V, Xiao Y (2019) PPWD1 is associated with the occurrence of postmenopausal osteoporosis as determined by weighted gene co-expression network analysis. Mol Med Rep 20:3202–3214. https://doi.org/10.3892/mmr.2019.10570
Schroeder HW Jr, Cavacini L (2010) Structure and function of immunoglobulins. J Allergy Clin Immunol 125:S41-52. https://doi.org/10.1016/j.jaci.2009.09.046
Sharma S, Mahajan A, Mittal A, Gohil R, Sachdeva S, Khan S, Dhillon M (2020) Epigenetic and transcriptional regulation of osteoclastogenesis in the pathogenesis of skeletal diseases: a systematic review. Bone 138:115507. https://doi.org/10.1016/j.bone.2020.115507
Wang T, He C (2020) TNF-α and IL-6: the link between immune and bone system. Curr Drug Targets 21:213–227. https://doi.org/10.2174/1389450120666190821161259
Weitzmann MN (2017) Bone and the immune system. Toxicol Pathol 45:911–924. https://doi.org/10.1177/0192623317735316
Xiao J, Wang R, Cai X, Ye Z (2021) Coupling of co-expression network analysis and machine learning validation unearthed potential key genes involved in rheumatoid arthritis. Front Genet 12:604714. https://doi.org/10.3389/fgene.2021.604714
Yang TL, Shen H, Liu A, Dong SS, Zhang L, Deng FY, Zhao Q, Deng HW (2020) A road map for understanding molecular and genetic determinants of osteoporosis. Nat Rev Endocrinol 16:91–103. https://doi.org/10.1038/s41574-019-0282-7
Zhang X, Chen K, Chen X, Kourkoumelis N, Li G, Wang B, Zhu C (2020) Integrative analysis of genomics and transcriptome data to identify regulation networks in female osteoporosis. Front Genet 11:600097. https://doi.org/10.3389/fgene.2020.600097
Zhao C (2021) Identifying the hub gene and immune infiltration of osteoarthritis by bioinformatical methods. Clin Rheumatol 40:1027–1037. https://doi.org/10.1007/s10067-020-05311-0
Zhu N, Zhang P, Du L, Hou J, Xu B (2020) Identification of key genes and expression profiles in osteoarthritis by co-expressed network analysis. Comput Biol Chem 85:107225. https://doi.org/10.1016/j.compbiolchem.2020.107225
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
Conceptualization and design were performed by PF, XF; Methodology and Software were performed by PF, NH, DP, LH; Writing—original draft preparation were performed by PF, XF.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fan, P., Feng, X., Hu, N. et al. Identifying Key Genes and Functionally Enriched Pathways in Osteoporotic Patients by Weighted Gene Co-Expression Network Analysis. Biochem Genet 62, 436–451 (2024). https://doi.org/10.1007/s10528-023-10425-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-023-10425-6