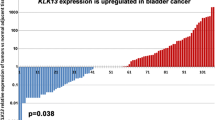
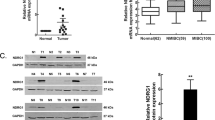
Abstract
Bladder cancer is one of the most predominant tumors of the genitourinary tract. In addition to pathological findings, the molecular modifications that might affect tumorigenesis and tumor outcome should be considered when treating bladder cancer. Accordingly, we aimed to investigate the expression levels of both the ASPM and TEF genes in bladder cancer tissues and their value in disease prognosis. The expression levels of the ASPM and TEF genes were analyzed by quantitative real-time PCR (qRT-PCR) in 90 bladder cancer tissue specimens and 90 specimens of normal urinary bladder tissue taken away from the tumor site. The upregulation of ASPM expression and the downregulation of TEF expression were observed in bladder cancer tissues compared to adjacent normal tissues, and these levels were correlated with high-grade tumors, advanced stage disease and the presence of metastasis. Both genes had the ability to predict metastatic association with sensitivity (84.62%) and specificity (68.42%; *P < 0.001) for the ASPM gene and for the TEF gene with sensitivity (80.77%) and specificity (78.95%; *P < 0.001). Additionally, Kaplan–Meier survival analysis indicated that elevated ASPM expression levels and reduced TEF expression levels significantly correlated with decreased overall survival and progression-free survival. The current analysis concludes that ASPM and TEF expressions might be used as potential biomarkers in bladder cancer patients.




Similar content being viewed by others
References
Amin MB, McKenney JK, Paner GP et al (2013) ICUD-EAU International Consultation on Bladder Cancer 2012: pathology. Eur Urol 63:16–35. https://doi.org/10.1016/j.eururo.2012.09.063
Amin MB, Smith SC, Reuter VE et al (2015) Update for the practicing pathologist: the International Consultation On Urologic Disease-European association of urology consultation on bladder cancer. Mod Pathol 28:612–630. https://doi.org/10.1038/modpathol.2014.158
Antoni S, Ferlay J, Soerjomataram I et al (2017) Bladder cancer incidence and mortality: a global overview and recent trends. Eur Urol 71(1):96–108
Bikeye SNN, Colin C, Marie Y et al (2010) ASPM-associated stem cell proliferation is involved in malignant progression of gliomas and constitutes an attractive therapeutic target. Cancer Cell Int 10:1–9. https://doi.org/10.1186/1475-2867-10-1
Brown AK, Webb AE (2018) Regulation of FOXO factors in mammalian cells. Current topics in developmental biology. Academic Press Inc, Cambridge, pp 165–192
Buchman JJ, Durak O, Tsai LH (2011) ASPM regulates Wnt signaling pathway activity in the developing brain. Genes Dev 25:1909–1914. https://doi.org/10.1101/gad.16830211
Cao J, Zhao X, Zhong Z et al (2016) Prognostic value of pre-operative renal insufficiency in urothelial carcinoma: a systematic review and meta-analysis. Sci Rep. https://doi.org/10.1038/srep35214
Capecchi MR, Pozner A (2015) ASPM regulates symmetric stem cell division by tuning Cyclin E ubiquitination. Nat Commun 6:8763. https://doi.org/10.1038/ncomms9763
Chen C, Hu L, Li X, Hou J (2017) Preoperative anemia as a simple prognostic factor in patients with urinary bladder cancer. Med Sci Monit 23:3528–3535. https://doi.org/10.12659/MSM.902855
Doenhoff MJ, Chiodini PL, Hamilton JV (2004) Specific and sensitive diagnosis of schistosome infection: can it be done with antibodies? Trends Parasitol 20:35–39
Dunphy KM, Garino MC, Shaw NM et al (2017) When the gold standard proves to be fool’s gold-blue-light cystoscopy in a case of high-risk non-muscle-invasive bladder cancer. Urology 110:27–30. https://doi.org/10.1016/j.urology.2017.05.032
Gutierrez O, Berciano MT, Lafarga M, Fernandez-Luna JL (2011) A novel pathway of TEF regulation mediated by microRNA-125b contributes to the control of actin distribution and cell shape in fibroblasts. PLoS ONE 6:e17169. https://doi.org/10.1371/journal.pone.0017169
Hua P, Liu W, Kuo SH et al (2012) Association of Tef polymorphism with depression in Parkinson disease. Mov Disord 27:1694–1697. https://doi.org/10.1002/mds.25195
Inukai T, Inaba T, Dang J et al (2005) TEF, an antiapoptotic bZIP transcription factor related to the oncogenic E2A-HLF chimera, inhibits cell growth by down-regulating expression of the common β chain of cytokine receptors. Blood 105:4437–4444. https://doi.org/10.1182/blood-2004-08-2976
Jiang F, Qi W, Wang Y et al (2019) lncRNA PEG10 promotes cell survival, invasion and migration by sponging miR-134 in human bladder cancer. Biomed Pharmacother 114:108814. https://doi.org/10.1016/j.biopha.2019.108814
Kaufman DS, Shipley WU, Feldman AS (2009) Bladder cancer. Lancet 374:239–249. https://doi.org/10.1016/S0140-6736(09)60491-8
Kouprina N, Pavlicek A, Collins NK et al (2005) The microcephaly ASPM gene is expressed in proliferating tissues and encodes for a mitotic spindle protein. Hum Mol Genet 14:2155–2165. https://doi.org/10.1093/hmg/ddi220
Kwan ML, Garren B, Nielsen ME, Tang L (2019) Lifestyle and nutritional modifiable factors in the prevention and treatment of bladder cancer. Urol Oncol Semin Orig Investig 37:380–386. https://doi.org/10.1016/j.urolonc.2018.03.019
Lavender S, Hilton PJ, Jones NF (1969) The measurement of glomerular filtration-rate in renal disease. Lancet (Lond, Engl) 2:1216–1218. https://doi.org/10.1016/s0140-6736(69)90752-1
Lin SY, Pan HW, Liu SH et al (2008) ASPM\s a novel marker for vascular invasion, early recurrence, and poor prognosis of hepatocellular carcinoma. Clin Cancer Res 14:4814. https://doi.org/10.1158/1078-0432.CCR-07-5262
Maia ARR, De Man J, Boon U et al (2015) Inhibition of the spindle assembly checkpoint kinase TTK enhances the efficacy of docetaxel in a triple-negative breast cancer model. Ann Oncol 26:2180–2192. https://doi.org/10.1093/annonc/mdv293
Maire V, Baldeyron C, Richardson M et al (2013) TTK/hMPS1 Is an attractive therapeutic target for triple-negative breast cancer. PLoS ONE 8:e63712. https://doi.org/10.1371/journal.pone.0063712
Moch H, Humphrey PA, Ulbright TM et al (2016) Tumor of urinary tract. In: Moch H, Humphrey PA, Ulbright TM, Reuter V (eds) WHO Classification of Tumours of the Urinary System and Male Genital Organs, 4th edn. International Agency for Research on Cancer, Lyon, pp 78–133
Pai VC, Hsu CC, Chan TS et al (2019) ASPM promotes prostate cancer stemness and progression by augmenting Wnt−Dvl-3−β-catenin signaling. Oncogene 38:1340–1353. https://doi.org/10.1038/s41388-018-0497-4
Paik J, Ding Z, Narurkar R et al (2009) FoxOs cooperatively regulate diverse pathways governing neural stem cell homeostasis. Cell Stem Cell 5:540–553. https://doi.org/10.1016/j.stem.2009.09.013
Rivlin N, Brosh R, Oren M, Rotter V (2011) Mutations in the p53 tumor suppressor gene: important milestones at the various steps of tumorigenesis. Genes Cancer 2:466–474
Rosenkrantz AB, Ego-Osuala IO, Khalef V et al (2016) Investigation of multisequence magnetic resonance imaging for detection of recurrent tumor after transurethral resection for bladder cancer. J Comput Assist Tomogr 40:201–205. https://doi.org/10.1097/RCT.0000000000000363
Siegel RL, Miller KD, Jemal A (2018) Cancer statistics, 2018. CA Cancer J Clin 68:7–30. https://doi.org/10.3322/caac.21442
Tang J, Lu M, Cui Q et al (2019) Overexpression of ASPM, CDC20, and TTK confer a poorer prognosis in breast cancer identified by gene co-expression network analysis. Front Oncol 9:1–14. https://doi.org/10.3389/fonc.2019.00310
Torre LA, Bray F, Siegel RL et al (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108. https://doi.org/10.3322/caac.21262
Wang E, Miller LD, Ohnmacht GA et al (2000) High-fidelity mRNA amplification for gene profiling. Nat Biotechnol 18:457–459. https://doi.org/10.1038/74546
Wang WY, Hsu CC, Wang TY et al (2013) A gene expression signature of epithelial tubulogenesis and a role for ASPM in pancreatic tumor progression. Gastroenterology 145:1110–1120. https://doi.org/10.1053/j.gastro.2013.07.040
Xie J-J, Zhuo Y-J, Zheng Y et al (2017) High expression of ASPM correlates with tumor progression and predicts poor outcome in patients with prostate cancer. Int Urol Nephrol 49:817–823. https://doi.org/10.1007/s11255-017-1545-7
Xu Z, Zhang QI, Luh F et al (2019) Overexpression of the ASPM gene is associated with aggressiveness and poor outcome in bladder cancer. Oncol Lett 17:1865–1876. https://doi.org/10.3892/ol.2018.9762
Yang J, Wang B, Chen H et al (2019) Thyrotroph embryonic factor is downregulated in bladder cancer and suppresses proliferation and tumorigenesis via the AKT/FOXOs signalling pathway. Cell Prolif 52:1–13. https://doi.org/10.1111/cpr.12560
Funding
There was no funding for this analysis.
Author information
Authors and Affiliations
Contributions
All the authors have contributed to the study to an adequate degree to be named as authors. AAS and SES performed the lab investigation and the molecular analysis and selected the study design. SG and ME were responsible for samples and data collections and evaluation of the involved patients. AS Performed the pathological examinations and all authors participate in writing and revision of the paper and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflict of interest.
Ethical Approval
This work was performed based on the Declaration of Helsinki and the principles of the “Ethical Committee of Medical Research”, Faculty of Medicine, Menoufia University. A written consent was provided by all participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Saleh, A.A., Gohar, S.F., Hemida, A.S. et al. Evaluation of ASPM and TEF Gene Expressions as Potential Biomarkers for Bladder Cancer. Biochem Genet 58, 490–507 (2020). https://doi.org/10.1007/s10528-020-09962-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-020-09962-1