Abstract
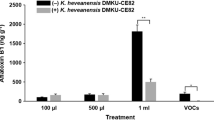
Aspergillus flavus infects stored grains and produces aflatoxins with highly toxic and carcinogenic properties, which are a threat to food safety and human health. Microbes and their bio-active compounds are considered effective agents to control A. flavus and aflatoxins during the pre- and post-harvest stages. Here, we reveal that Bacillus flexus strain TR-1 isolated from tea plant rhizosphere soil could efficiently inhibit the growth of A. flavus in dual culture tests through the production of antifungal volatiles. Additionally, these volatiles showed great inhibitory effects against A. flavus infection and aflatoxin production in maize and peanut with high water activity levels during storage. Scanning electron microscope analysis further proved that the conidia of A. flavus on the peanut surface treated with TR-1 volatiles could not germinate and were severely dehydrated and shrunken. Gas chromatography-tandem mass spectrometry revealed that five antifungal compounds were identified in the volatile profiles, including benzene, methyl thiolacetate, dimethyl disulfide, butanethioic acid S-methyl ester analogue, dimethyl trisulfide (DMTS), and S-methyl 3-methylbutanethioate. DMTS, with the best antagonistic effect, completely inhibited the growth of A. flavus at 10 μl−1 (compound volume/airspace volume). TR-1 also exhibited efficient and broad inhibitory effects against six other fungal pathogens. Overall, B. flexus strain TR-1 and its produced volatiles showed valid antifungal activity against phytopathogens. This study provides novel materials for the production of bioactive agents to control plant pathogens in stored grains.





Similar content being viewed by others
References
Abdallah MF, Girgin G, Baydar T (2019) Mycotoxin detection in maize, commercial feed, and raw dairy milk samples from Assiut City. Egypt Vet Sci 6(2):57
Alamri SA (2012) Biodegradation of microcystin-RR by Bacillus flexus isolated from a Saudi freshwater lake. Saudi J Biol Sci 19(4):435–440
Boukaew S, Prasertsan P (2020) Efficacy of volatile compounds from Streptomyces philanthi RL-1-178 as a biofumigant for controlling growth and aflatoxin production of the two aflatoxin-producing fungi on stored soybean seeds. J Appl Microbiol 129(3):652–664
Cao HY, Jiao Y, Yin N, Li Y, Ling J, Mao ZC, Yang YH, Xie BY (2019) Analysis of the activity and biological control efficacy of the Bacillus subtilis strain Bs-1 against Meloidogyne incognita. Crop Prot 122:125–135
Curto G, Dongiovanni C, Sasanelli N, Santori A, Myrta A (2014) Efficacy of dimethyl disulfide (DMDS) in the control of the root-knot nematode Meloidogyne incognita and the cyst nematode Heterodera carotae on carrot in field condition in Italy. Acta Hortic 1044:405–410
Dugravot S, Thibout E, Abdo-Ghalia A, Huignard H (2004) How a specialist and a non-specialist insect cope with dimethyl disulfide produced by Allium porrum. Entomol Exp Appl 113:173–179
Egmond HP, Jonker MA (2003) Worldwide regulations for mycotoxins in food and feed in 2003. FAO Food Nutr Pap 81
Giacomucci L, Raddadi N, Soccio M, Lotti N, Fava F (2019) Polyvinyl chloride biodegradation by Pseudomonas citronellolis and Bacillus flexus. N Biotechnol 52:35–41
Gong AD, Li HP, Shen L, Zhang JB, Wu AB, He WJ, Yuan QS, He JD, Liao YC (2015) The Shewanella algae strain YM8 produces volatiles with strong inhibition activity against Aspergillus pathogens and aflatoxins. Front Microbiol 6:1091
Gong AD, Dong FY, Hu MJ, Kong XW, Wei FF, Gong SJ, Zhang YM, Zhang JB, Wu AB, Liao YC (2019a) Antifungal activity of volatile emitted from Enterobacter asburiae Vt-7 against Aspergillus flavus and aflatoxins in peanuts during storage. Food Control 106:106718
Gong AD, Wu NN, Kong XW, Zhang YM, Hu MJ, Gong SJ, Dong FY, Wang JH, Zhao ZY, Liao YC (2019b) Inhibitory effect of volatiles emitted from Alcaligenes faecalis N1–4 on Aspergillus flavus and aflatoxins in storage. Front Microbiol 10:1419
Gong AD, Sun GJ, Zhao ZY, Liao YC, Zhang JB (2020) Staphylococcus saprophyticus L-38 produces volatile 3,3-dimethyl-1,2-epoxybutane with strong inhibitory activity against Aspergillus flavus germination and aflatoxin production. World Mycotoxin J 13(2):247–258
Gong AD, Wang GZ, Sun YK, Song MG, Dimuna C, Gao Z, Wang HL, Yang P (2022) Dual activity of Serratia marcescens Pt-3 in phosphate-solubilizing and production of antifungal volatiles. BMC Microbiol 2:26
Hassan ZU, Thani RA, Alnaimi H, Migheli Q, Jaoua S (2019) Investigation and application of Bacillus licheniformis volatile compounds for the biological control of toxigenic Aspergillus and Penicillium spp. ACS Omega 4(17):17186–17193
Jaibangyang S, Nasanit R, Limtong S (2020) Biological control of aflatoxin-producing Aspergillus flavus by volatile organic compound-producing antagonistic yeasts. BioControl 65(3):377–386
Lee HJ, Ryu D (2017) Worldwide occurrence of mycotoxins in cereals and cereal-derived food products: Public health perspectives of their co-occurrence. J Agric Food Chem 65(33):7034–7051
Lyu A, Yang L, Wu MD, Zhang J, Li GQ (2020) High efficacy of the volatile organic compounds of Streptomyces yanglinensis 3–10 in suppression of Aspergillus contamination on peanut kernels. Front Microbiol 11:142
Ma F, Cai XF, Mao J, Yu L, Li PW (2021) Adsorptive removal of aflatoxin B1 from vegetable oils via novel adsorbents derived from a metal-organic framework. J Hazard Mater 412:125170
Mannaa M, Oh JY, Kim KD (2017) Biocontrol activity of volatile-producing Bacillus megaterium and Pseudomonas protegens against Aspergillus flavus and aflatoxin production on stored rice grains. Mycobiology 45(3):213–219
Maurya A, Kumar S, Singh BK, Chaudhari AK, Dwivedy AK, Prakash B, Dubey NK (2021) Mechanistic investigations on antifungal and antiaflatoxigenic activities of chemically characterised Carum carvi L. essential oil against fungal infestation and aflatoxin contamination of herbal raw materials. Nat Prod Res 36(7):4563–4568
Medina A, Gilbert MK, Mack BM, Obrian G, Rodriguez A, Bhatnager D, Magan N (2017) Interactions between water activity and temperature on the Aspergillus flavus transcriptome and aflatoxin B1 production. Int J Food Microbiol 256:36–44
Mitchell NJ, Bowers E, Hurburgh C, Wu F (2016) Potential economic losses to the USA corn industry from aflatoxin contamination. Food Addit Contam Part A 33(3):540–550
Moradi M, Rohani M, Fani SR, Mosavian MTH, Probst C, Khodaygan P (2020) Biocontrol potential of native yeast strains against Aspergillus flavus and aflatoxin production in pistachio. Food Addit Contam Part A 37(11):1963–1973
Muhialdin BJ, Algboory HL, Kadum H, Mohammed NK, Saari N, Hassan Z, Hussin ASM (2020) Antifungal activity determination for the peptides generated by Lactobacillus plantarum TE10 against Aspergillus flavus in maize seeds. Food Control 109:106898
Mulla SI, Ameen F, Tallur PN, Bharagava RN, Bangeppagari M, Eqani SAMAS, Bagewadi ZK, Mahadevan GD, Yu CP, Ninnekar HZ (2017) Aerobic degradation of fenvalerate by a Gram-positive bacterium, Bacillus flexus strain XJU-4. 3 Biotech 7(5):320
Papazlatani C, Rousidou C, Katsoula A, Kolyvas M, Genitsaris S, Papadopoulou KK, Karpouzas DG (2016) Assessment of the impact of the fumigant dimethyl disulfide on the dynamics of major fungal plant pathogens in greenhouse soils. Eur J Plant Pathol 146:391–400
Saleh AE, Ul-Hassan Z, Zeidan R, Al-Shamary N, Al-Yafei T, Alnaimi H, Higazy NS, Migheli Q, Jaoua S (2021) Biocontrol activity of Bacillus megaterium BM344-1 against toxigenic fungi. ACS Omega 6(16):10984–10990
Tian F, Lee SY, Woo SY, Choi HY, Park SB, Chun HS (2021) Effect of plant-based compounds on the antifungal and antiaflatoxigenic efficiency of strobilurins against Aspergillus flavus. J Hazard Mater 415:125663
Warth B, Sulyok M, Fruhmann P, Mikula H, Berthiller F, Schuhmacher R, Hametner C, Abia WA, Adam G, Frohlich J, Krska R (2012) Development and validation of a rapid multi-biomarker liquid chromatography/tandem mass spectrometry method to assess human exposure to mycotoxins. Rapid Commun Mass Spectrom 26(13):1533–1540
Xiong YW, Li XW, Wang TT, Gong Y, Zhang CM, Xing K, Qin S (2020) Root exudates-driven rhizosphere recruitment of the plant growth-promoting rhizobacterium Bacillus flexus KLBMP 4941 and its growth-promoting effect on the coastal halophyte Limonium sinense under salt stress. Ecotoxicol Environ Saf 194:110374
Yan CL, Wang Q, Yang QL, Wu W (2020) Recent advances in aflatoxins detection based on nanomaterials. Nanomater 10(9):1626
Yang MG, Lu LF, Pang J, Hu YL, Guo QB, Li ZJ, Wu SF, Liu HH, Wang CL (2019) Biocontrol activity of volatile organic compounds from Streptomyces alboflavus TD-1 against Aspergillus flavus growth and aflatoxin production. J Microbiol 57(5):396–404
Zhu YL, Xu YN, Yang QL (2021) Antifungal properties and AFB1 detoxification activity of a new strain of Lactobacillus plantarum. J Hazard Mater 414(7):125569
Funding
This work was supported by the National Natural Science Foundation of China [grant numbers 31701740], the National Natural Science Foundation of Henan [grant number 222300420519, 222301420111], Scientific and Technological Project in Henan Province [grant number 212102110447], and Nanhu Scholars Program for Young Scholars of XYNU. The authors would like to thank TopEdit (https://www.topeditsci.com) for its linguistic assistance during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Handling Editor: Jane Debode
Supplementary Information
Below is the link to the electronic supplementary material.
10526_2023_10187_MOESM1_ESM.tif
Supplementary file1 (TIF 338 KB)—Mass spectrogram comparison between VOCs in TR-1 (RT=3.042 min) (up histogram) and benzene in the NIST 17 MS spectral database (down histogram). Horizontal axis indicates mass charge ratios, and peaks the relative abundance
10526_2023_10187_MOESM2_ESM.tif
Supplementary file2 (TIF 475 KB)—Mass spectrogram comparison between VOCs in TR-1 (RT=3.388 min) (up histogram) and methyl thiolacetate in the NIST 17 MS spectral database (down histogram). Horizontal axis indicates mass charge ratios, and peaks the relative abundance
10526_2023_10187_MOESM3_ESM.tif
Supplementary file3 (TIF 1109 KB)—Mass spectrogram comparison between VOCs in TR-1 (RT=4.389 min) (up histogram) and DMDS in the NIST 17 MS spectral database (down histogram). Horizontal axis indicates mass charge ratios, and peaks the relative abundance
10526_2023_10187_MOESM4_ESM.tif
Supplementary file4 (TIF 696 KB)—Mass spectrogram comparison between VOCs in TR-1 (RT=7.581 min) (up histogram) and butanethioic acid S-methyl ester in the NIST 17 MS spectral database (down histogram). Horizontal axis indicates mass charge ratios, and peaks the relative abundance
10526_2023_10187_MOESM5_ESM.tif
Supplementary file5 (TIF 1329 KB)—Mass spectrogram comparison between VOCs in TR-1 (RT=12.113 min) (up histogram) and DMTS in the NIST 17 MS spectral database (down histogram). Horizontal axis indicates mass charge ratios, and peaks the relative abundance
10526_2023_10187_MOESM6_ESM.tif
Supplementary file6 (TIF 1588 KB)—Broad spectrum antifungal activity of TR-1 against six important fungal pathogens. The fungi were inoculated in the center of a PDA plate and challenged with (control+TR-1) or without (control) TR-1, respectively. Fg: F. graminearum PH1, Fv: F. verticillioides 3056, Cg: C. graminicola M2, Cf: C. fructicola MG3, Aa: A. alternata TA133, and Bc: B. cinerea Y25
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gong, AD., Song, MG., Wang, HL. et al. Inhibitory effect of volatile organic compounds from Bacillus flexus TR-1 against Aspergillus flavus and aflatoxins in grains during storage. BioControl 68, 181–190 (2023). https://doi.org/10.1007/s10526-023-10187-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-023-10187-5